All published articles of this journal are available on ScienceDirect.
The Evolution of Anatomic Anterior Cruciate Ligament Reconstruction
Abstract
Anterior cruciate ligament reconstruction has evolved significantly since the early 1900’s, back when an emphasis was placed on repair and not reconstruction. Over the past century, the technique has evolved from intra-articular non anatomic reconstruction, to extra articular reconstruction, back to intra articular (performed arthroscopically), to now, the advent of anatomic insertion site restoration. This review will aim to illustrate the changes that have occurred, describing the rational for this process, based upon anatomical, radiological, biomechanical and clinical studies, all of which have aimed to improve patient function following ACL injury.
INTRODUCTION
Since the first reported case by Robert Adams in 1837, the treatment of anterior cruciate ligament rupture has significantly evolved over the past 175 years [1]. Although the biomechanical advantages of having an intact ACL were known then, it wasn’t until 1895 when Mayo Robson of Leeds Royal Infirmary performed the first ACL repair [1]. Since then, the treatment of ACL injuries has occupied many orthopaedic minds, with debates raging between extra-articular versus intra-articular fixation, open versus arthroscopic technique, optimal graft selection, graft fixation, rehabilitation, and more recently, anatomical reconstruction. In a recent article, Fu and Karlsson described anatomic ACL reconstruction as ‘the functional restoration of the ACL to its native dimensions, collagen orientation and insertion sites’ [2].
This review will focus on how we have evolved to the philosophy of anatomic reconstruction, what was the impetus for change, and what potential benefits may be found.
THE FORGOTTEN YEARS OF ANATOMIC ACL RECONSTRUCTION
The early pioneers of operative treatment of ACL rupture mostly focused on direct repair. It wasn’t until 1916 when Hey Groves of Bristol performed the first reconstruction [1]. He recognized the anatomy of the ACL, and the need for an intra-articular, obliquely orientated graft to control anterior translation; in his case, using the entire fascia lata, routed into the joint via a laterally placed bone tunnel. In fact, he and a number of surgeons at that time were the early adopters of anatomic ACL reconstruction, but their philosophy was largely forgotten, until resurrected more recently by Fu and Yasuda in the early 2000’s [3, 4].
Anatomic techniques were soon to be replaced by extra-articular reconstruction, pioneered by Strickler (1937), Lemaire (1960) and MacIntosh (1970’s), utilizing a lateral extra-articular tenodesis to control anterolateral tibial subluxation, and by Slocum who described the pes anserinus transfer, to help control anteromedial rotational laxity [1]. Although these techniques were shown to control external rotational laxity, they were soon found to stretch out and not control the knee to the desired manner. This then led the way to combined intra- and extra-articular reconstruction. Interestingly, although the combined procedure by in large went out of fashion in the late 1990’s, extra-articular tenodesis is making somewhat of a resurgence, with a number of authors using the procedure to aid control of rotational laxity, particularly in revision scenarios [5].
THE 80’S AND 90’S - WHICH GRAFT AND WHICH TECHNIQUE?
The 1980’s and 90’s were dominated by which technique intra-articular reconstruction should be performed. David Dandy of Cambridge performed the first arthroscopic assisted intra-articular ACL reconstruction in 1980 [1]. He combined a carbon fibre intra-articular graft with a Macintosh lateral extra-articular tenodesis, reporting satisfactory results. However, it was later noted that the synthetic ligament soon failed.
With improved instrumentation and technological advances in fibre optics, allowing improved arthroscopic visualization, the traditional two-incision open technique gave way to the one incision, arthroscopic ACL reconstruction. Graft choices varied, predominantly between patella tendon and hamstring autograft, and allograft. A phase of synthetic graft utilization occurred including among others, carbon fibre, dacron and polyester, almost all of which encountered early failure rates, and hence a move away from this technology was established [6-8]. Like the extra-articular tenodesis, synthetic ligaments are also increasing in popularity. The LARS ligament has been marketed as a graft which can allow early return to sport, a choice which has been taken up by many older elite sportsmen [9].
Debate regarding graft placement, particularly femoral tunnel position, centered on the clock face position (Fig. 1). A clock face would be superimposed on the intercondylar notch and the femoral tunnel drilled at the 11 o’clock position on the right, and the 1 o’clock position on the left, with an offset guide referenced from the deep aspect of the notch with the knee at 90 degrees. A number of techniques of drilling the femoral tunnel were described. The two incision technique involved a laterally based incision centered over the lateral femoral condyle, allowing an outside-in guide to be placed on the femoral footprint, from a posterolaterally based approach. Although considered to be more anatomic, it was deemed too challenging by many, therefore instrumentation was developed to allow for transtibial femoral tunnel drilling. For example, Howell developed a guide which used the roof of the intercondylar notch as a guide to allow satisfactory position of the tibial tunnel, avoiding graft impingement in extension [10]. At the same time, many surgeons in the southern hemisphere were popularizing anteromedial portal drilling of the femoral tunnel, a significant evolution in terms of what would come in later years.
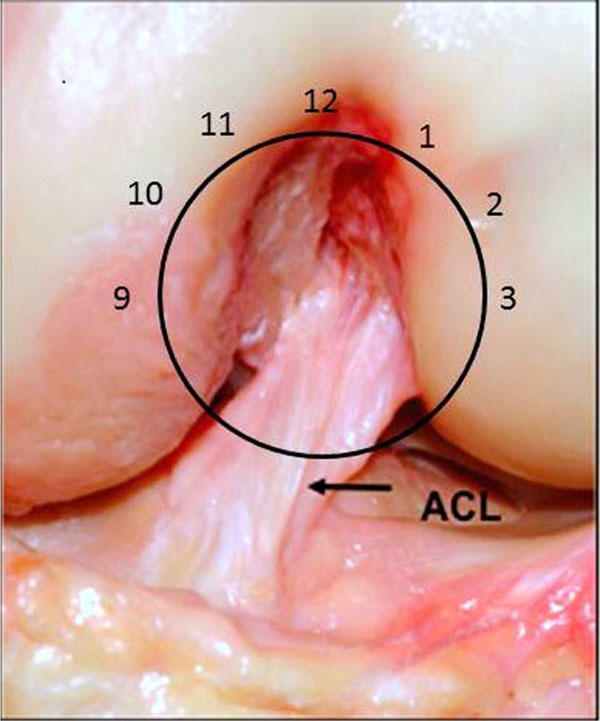
An anterior view of the intercondylar notch in a cadaver with a clock face superimposed. The native ACL can be seen to attach to the femur between the 1 and 3 o’clock positions.
Through the publication of a number of case series of ACL reconstructions, satisfactory results were reported [11-13]. However, many surgeons commented that by using a transtibial drilling technique, the correct position of the femoral tunnel was not possible and therefore a vertically placed graft resulted [14]. This concept was later supported by Bach and colleagues who, again focused on tibial tunnel position initially to enable proper placement of the femoral tunnel [15].
IF IT’S NOT BROKEN, WHY FIX IT?
Considering all of the potential options available, the results of arthroscopic ACL reconstruction achieved fairly uniform, reproducible results. As previously described, most of the literature consisted of case series reporting satisfactory results. However, more detailed analysis of many of the studies revealed many issues.
Yunes et al. performed a meta-analysis of controlled trials comparing patella tendon and hamstring tendon graft ACL reconstructions [16]. Only four studies were deemed of satisfactory scientific rigor to be included. Although there was a significant heterogeneity in the surgical procedure and reported outcome measures, some interesting finding were reported. Between 16-25% of patients had a positive pivot shift test, 27-40% had greater than 3mm side to side difference on maximum manual KT1000 testing, and only 64-75% of patients returned to their pre-injury level of sport.
In 2005, Prodromos et al. performed a further meta-analysis comparing hamstring and patella tendon reconstructions [17]. Again, suboptimal results were found with only 77% of the hamstring reconstructions, and 66% of patella tendon reconstructions, achieving normal stability rates. In a similar study, the same group analysed the difference in rates of stability between autograft and allograft [18]. Autograft stability was reported as normal in 72% of cases with only 59% of allograft cases being normal.
Other studies included biomechanical assessment of traditional ACL reconstructions, which were noted to not restore normal tibial rotation compared to the contralateral knee. Ristansis et al. showed that abnormal tibial rotation occurred when descending stairs and pivoting [19], later confirming that a traditionally placed hamstring graft would not restore the normal knee joint kinematics [20]. Tashman et al. showed similar findings when investigating running in an ACL reconstructed knee [21]. The combination of abnormal kinematics, clinical studies indicating a significant proportion of patients exhibiting poor clinical rotational stability, and studies showing a relationship between ACL injury, reconstruction and the development of osteoarthritis [22], suggested that further investigational work was required to re-establish normal knee joint kinematics and hopefully a more optimal physiological joint environment.
One of many subsequent alterations to the technique to address the above findings included the femoral tunnel position being placed ‘further around the clock face’, to give a more oblique graft orientation and potentially improve rotational control. Loh et al. demonstrated that improved rotational control could be established in cadaveric knees when the femoral tunnel position was moved from 11 o’clock to the 10 o’clock position in the intercondylar notch [3]. Lee et al. supported this finding in a clinical study, demonstrating that lower subjective outcome scores and reduced rotational control were noted in patents that had a more vertically oriented graft secondary to vertical femoral tunnel placement in the axial plane [23]. These studies, amongst others, supported the establishment of anteromedial (AM) portal femoral tunnel drilling, versus the more accepted trans-tibial technique, allowing a more obliquely orientated femoral tunnel and graft to be fashioned. Numerous authors have since shown that satisfactory anatomic positioning of the femoral tunnel is not possible, [24, 25] without compromising the tibial tunnel in some manner [26, 27] when drilling the femoral tunnel using a trans-tibial technique. Hemming showed that the trans-tibial technique could produce an anatomically centered femoral tunnel, however the tibial tunnel length was compromised due to the oblique nature and proximal starting position, which in turn would result in tibial graft fixation being compromised [27]. Bedi et al. also demonstrated that the central position in the femoral footprint could not be reached without compromising the tibial tunnel aperture, on this occasion utilizing an eccentrically placed guide pin within the tibial tunnel to drill the femur, resulting in an oval hole which in turn could affect graft fixation and incorporation [26]. ‘Potentially reducing anterior control of the tibia on femur.
‘DOUBLE BUNDLE OR DOUBLE TROUBLE’ [28]
The major revolution in ACL surgery came with the emergence of double bundle reconstruction. Although not a new concept, Radford et al. having studied the procedure in sheep in 1994 [29], it wasn’t until the early 2000’s when Yasuda [4] and Fu [30] took it through to clinical practice that the idea really gained in popularity. A number of other groups also investigated the use of two grafts reconstructing both the anteromedial (AM) and posterolateral (PL) bundles of the ACL (Fig. 2). Each bundle was recognized to have differing kinematic roles, the AM being more taught in flexion, and the PL more taught in extension. This led to concepts of differential tensioning [31], different types of graft for different bundles, and newer surgical techniques to allow reproducible placement of grafts without intra-operative complications. However, concern was raised with regards to the need for 4 tunnels being drilled in the knee, the potential complications intra-operatively which could be associated with this, as well as technical difficulty associated with potential later revision. In an editorial by Harner and Poehling in 2004 [28], these concerns were voiced, along with a recommendation of caution as to the general acceptance of double bundle ACL reconstruction prior to level 1 randomised comparative studies being performed demonstrating clinical superiority over traditional single bundle techniques.
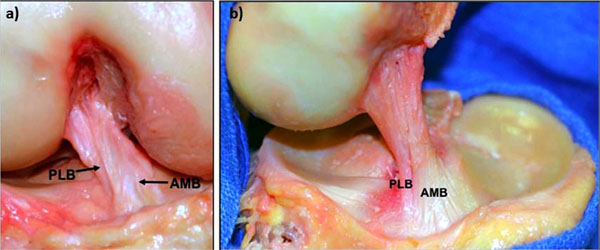
a) A cadaveric dissection demonstrating the two bundles of the ACL; the anteromedial (AM) and posterolateral (PL) bundles, and b) the fan-like insertion on the femoral condyle, viewed from the medial side. Courtesy of Dr. Charles Brown.
In 2008, Kondo et al. published the results of a cohort study, in which double bundle reconstruction was compared to single bundle reconstruction [32]. They found that an improvement in anterior and rotational stability was found, however, no subjective clinical differences were found between the groups in terms of patient reported outcome measures. Similar results were found in randomized comparative studies by Muneta et al. [33] and Jarvela et al. [34]. In a meta-analysis by Meredick et al., double bundle ACL reconstruction was not found to produce clinically superior control of anterior and rotational laxity over single bundle reconstruction [35]. Additionally, concerns have since been raised of tunnel widening associated with double bundle reconstruction [36] and potentially greater risk of re-rupture.
UNDERSTANDING ANATOMY
The most significant result of double bundle ACL reconstruction was the further investigation and understanding of ACL insertion anatomy.
Although Bernard and Hertel had actually previously described the centre of the ACL femoral footprint radiologically in 1997 (Fig. 3) [37], it was the renewed interest from the pioneers of double bundle reconstruction which redefined the femoral footprint anatomy. In 2006, Colombet et al. described the anatomy of both ACL bundles on fresh frozen cadaveric specimens and documented insertion sites of femoral and tibial footprints both macroscopically and radiologically [38]. One year later, Ferretti et al. published their work on the femoral footprint insertion site anatomy, again using fresh frozen cadavers, as well as arthroscopic evaluation of ACL reconstructions [39]. They described distinct anatomical boney ridges, the ‘lateral intercondylar ridge’ and the ‘lateral bifurcate ridge’, which were present in 100% and between 81-86% (depending on the nature of the specimen) of specimens respectively. This led to a number of other papers defining the insertion site anatomy, from anatomical dissection methods [40, 41], radiologically [42-44], histologically [45] and arthroscopic landmarks (Fig. 4a, b) [46].
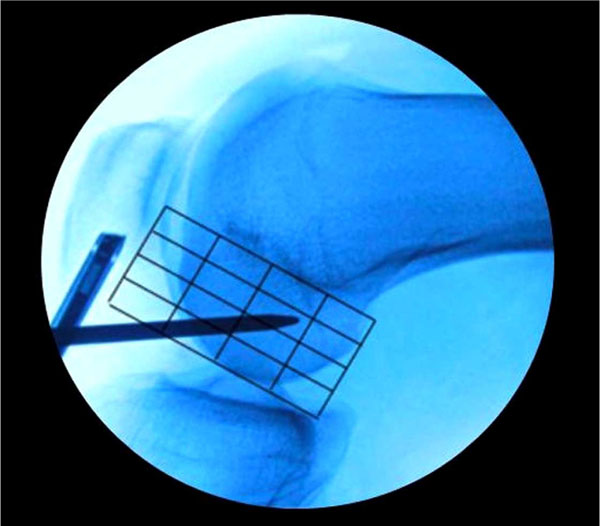
An intra-operative fluoroscopic lateral image of the knee with the Bernard and Hertel grid laid over the lateral wall of the intercondylar notch. The microfracture awl is placed in the mid bundle position of the femoral insertion footprint.
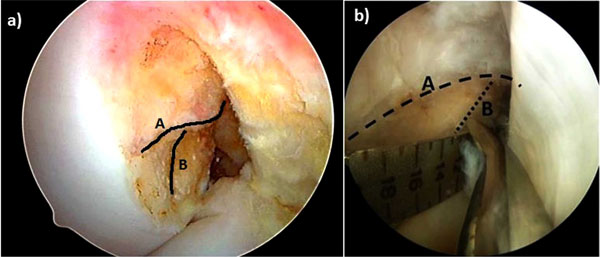
Arthroscopic view of the lateral wall of the intercondylar notch from the anteromedial portal. a) the lateral intercondylar ridge (‘residents ridge) (A) and the lateral bifurcate ridge (B) are shown. b) the mid bundle position being marked with a microfracture awl, placed on the lateral bifurcate ridge.
Interestingly, the lateral intercondylar ridge as it is now known was previously recognized by Clancy, who named it Residents ridge, as it was here that inexperienced surgeons often made the mistake of placing offset guides along it, believing it to be the posterior wall of the intercondylar notch. This invariably led to a shallow position on the femur and a poorly functioning graft.
This new understanding of anatomy led some investigators to believe that the ‘clock face’ description of the femoral insertion was not accurate enough [47], hence newer terminology has evolved to describe the arthroscopic view of anatomical placement (Fig. 5) [48].
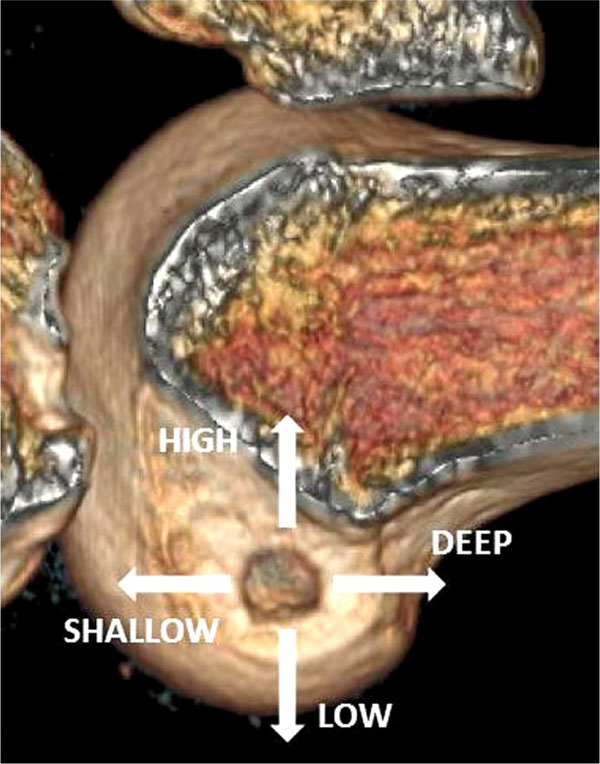
The arthroscopic description of tunnel position on the femur.
Mechanical testing followed these new concepts, allowing investigators to elicit differences in tunnel position. Kato et al. used porcine knees to determine the difference in a number of combinations of femoral and tibial footprint positions [49]. They described a traditional ACL reconstruction as one which runs from an anteromedial (AM) position on the femur to a posterolateral (PL) position on the tibia (to allow femoral drilling via transtibial approach). They compared this combination to AM-AM position and mid bundle to mid bundle position. Kinematic data confirmed that during simulated anterior and rotational loads, the mid-mid combination most closely restored the normal kinematics of the knee. Kondo et al. performed a similar study in human cadaveric specimens [50]. Comparing traditional single bundle to double bundle and anatomic single bundle reconstructions, simulated anterior translation and pivot shift revealed significant improvements in rotational control with the double bundle over traditional single bundle; however no differences were noted between double bundle and anatomic single bundle reconstructions.
These findings were similar to an earlier paper in which anatomic single bundle and double bundle reconstructions provided similar levels of restoration of rotational control in cadaveric specimens [51]. However, a more recent study by Cross et al. have shown that AM position reconstruction can equally restore the anterior and rotational control similar to mid bundle reconstruction [52]. They concluded that either position will provide satisfactory time zero biomechanical results.
Further evidence of the potential benefit of anatomic single bundle reconstruction has been provided by Claes et al. who performed in-vivo gait analysis following ACL reconstruction, comparing anatomic single to double bundle reconstructions [53]. They found that tibial rotation was equally restored by both procedures during performance of low and high demand activities including pivoting.
ANATOMIC RECONSTRUCTION
Anatomical and biomechanical studies have provided compelling evidence and persuasive arguments to support anatomic ACL reconstruction. However, similar to the double bundle argument, it is important that these new procedures are safe, and ideally improve clinical outcome. At present there are very few clinical studies documenting the outcome following anatomic single bundle reconstruction. A number of authors have described a variety of techniques to aid the placement of the femoral and tibial tunnels [54-56]. However, no clinical data was provided to show that these techniques were efficacious. Seibold published his philosophy on how to restore the complete femoral and tibial footprints, using a combination of footprint measurement, tunnel placement and drill diameter to establish a more optimal coverage of the insertion site [57]. However, again no clinical data accompanied this description, therefore it is the authors’ belief that this method is not scientifically proven and should therefore be followed with caution.
In 2010, van Eck et al. published a systematic review on surgical techniques relating to anatomical reconstruction [58]. They concluded that significant under-reporting of surgical data and operative techniques made comparative analysis difficult. They encouraged investigators to report their techniques in a more specific and standardized fashion.
A number of papers have been published using CT to validate the position of the femoral and tibial tunnels during anatomic reconstruction. Forsythe et al. reported the 3D CT scan analysis of tunnel position of double bundle ACL reconstruction. Using similar methods, Bird et al. demonstrated that the anatomic mid bundle position could be found intra-operatively using a mark 50% of the way along a line measured from the deep aspect of the lateral wall of the intercondylar notch/femoral condyle to the most anterior aspect of the articular cartilage margin [59]. This was validated using 3D CT measurement referencing the Bernard and Hertel grid radiographic landmarks [37].
In 2012, Hussein et al. published the results of the first randomized comparative study, comparing patient reported outcome and objective clinical testing in patients undergoing one of either traditional single bundle reconstruction, double bundle reconstruction or anatomic single bundle reconstruction [60]. At a mean follow up of 51 months, double bundle reconstructions showed a statistically significant improvement in anterior and rotational stability compared to both of the other groups. Anatomic single bundle was shown to be better than the conventional single bundle approach, but these findings were not significant. However, minimal clinically relevant differences were found in the subjective patient reported outcome between all three groups.
On face value, it would be tempting to conclude based on this study, that double bundle reconstruction is superior to the other techniques. However, reports of increasing failure rate associated with double bundle reconstruction have been published [61, 62]. This, combined with a more difficult revision, would suggest that double bundle techniques should be left for well-defined clinical cases, such as augmentation of single bundle ruptures or some revision scenarios [63, 64].
FUTURE PERSPECTIVES
ACL reconstruction techniques will continue to evolve. At present, most investigators are working toward an anatomical approach to restoring ACL function and normal knee joint kinematics. Interestingly, recent results from the Danish ACL registry have suggested a higher re-rupture rate associated with AM portal drilling of the femoral tunnel (unpublished data – personal communication). Although one cannot assume a direct relationship between these two variables, a number of hypotheses have been suggested. By producing a more obliquely orientated, anatomical graft, the likelihood is that the graft will experience greater load during high demand activities, thereby causing failure. Equally, the grafts that we currently use are stiffer constructs than the native ACL [65]. It is possible that these stiff constructs are unable to absorb the load placed on it, transmitting significant loads to other parts of the joint which do not normally see such high stress. Also, by a process of stress shielding, the normal cellular constituents of the graft may not experience the required degree of mechanobiological stimulus to produce extra-cellular matrix and produce a mature collagen network. These theories may therefore form the basis of research in future years, with the emphasis placed on developing more appropriate grafts, placed in their correct anatomical locations.
Rehabilitation will continue to play a major factor in achieving optimal surgical outcomes. Arden et al. have shown that as many as two thirds of patients will not have returned to their pre-injury level of sport by 12 months following reconstruction [66]. Furthermore, in a study evaluating return to sporting activity in a cohort of ACL reconstructions 2-7 years out from surgery, the same authors concluded that the inability to reach sports participation at the pre-injury level within 12 months from surgery, was not predictive of a return to pre-injury level sports participation at a later date [67]. This data, combined with biological hypotheses detailed above, may suggest that a slower return to sport may be optimal to allow satisfactory graft maturation and neuromuscular control of the knee.
Lastly, a combination of improved biomechanical control with biological augmentation will likely be required to improve long term outcome in ACL reconstruction, particularly with respect to the development of osteoarthritis. Potter et al. have recently shown the consequences of impaction injury of the articular surface following ACL injury [68]. At 7 to 11 years following injury, a bone bruise detected by MRI on the lateral femoral condyle of the knee was associated with a 50 times baseline risk of cartilage loss, compared to 30 times baseline in the patella, and 19 times in the medial femoral condyle. Impaction injury of articular cartilage has been shown to have detrimental effects on extracellular matrix constituents, and chondrocyte death [69-71]. A number of early studies have shown potential benefit in adding biological adjuvants to injured joints, to help modulate the damage repair response, and hopefully alter the catabolic cascade which the articular surface invariably follows [72-74]. It remains to be seen whether biological augmentation of a biomechanical procedure, such as ACL reconstruction, will have a significant effect on long term patient function.
CONCLUSION
Anterior cruciate ligament reconstruction has gone through an evolutionary process over the last century. Initial anatomical reconstructions initially gave way to non-anatomical procedures; however, over the past decade anatomic reconstruction has enjoyed a resurgence in popularity (Fig. 6). Anatomical and biomechanical studies provide compelling clinical evidence that anatomic insertion site restoration is the future of ACL reconstruction; however, more comparative controlled clinical studies, with greater numbers of patients and longer term follow up are awaited to ascertain whether anatomic single bundle reconstruction will provide any clinical benefit over conventional techniques.

Arthroscopic view of two ACL reconstructions: a) Pre anatomic position, with the graft placed higher in the notch resulting in more vertical collagen fibre orientation, and b) anatomic ACL reconstruction with a low placed graft resulting in a more oblique graft position. Courtesy of Dr. Charles Brown.
ACKNOWLEDGEMENT
Authors like to acknowledge Dr Charles Brown for providing some of the images.
CONFLICT OF INTEREST
Neither of the authors have a conflict of interest to declare in relation to the production of this manuscript.


