All published articles of this journal are available on ScienceDirect.
Preliminary Results of Expandable Transforaminal Lumbar Interbody Fusion Cages
Abstract
Background:
Compared with static cages, expandable cages for Transforaminal Lumbar Interbody Fusion (TLIF), are thought to require less posterior bony removal and nerve root retraction. They may allow the creation of a greater lordotic angle and lordosis restoration.
Objective:
This study investigated whether TLIF using an expandable lordotic interbody cage facilitates an improvement in both segmental lordosis and the restoration of intradiscal height.
Methods:
A total of 32 patients with 40 operated segments underwent TLIF surgery for lumbar degenerative disc disease and were consecutively included in this prospective observational study. Of those patients, 25 received monosegmental treatment, six were treated bisegmentally, and one was treated trisegmentally. All patients were assessed clinically and radiographically preoperatively, as well as one week, six months, and two years postoperatively.
Results:
Two patients required revision for screw loosening and pseudarthrosis. In four patients, the endplate was violated intraoperatively due to cage placement. Postoperatively, cage subsidence was observed in four patients. Significant improvement in the mean degree of spondylolisthesis was noted at the two-year mark. Mean segmental lordosis improved postoperatively. A significant increase in mean disc height of the treated segment was also found. Overall, with the exception of pain, no significant clinical or radiographic changes were reported between the first postoperative week and the two-year year follow-up mark. The mean pain, functional, and quality of life outcomes improved significantly from the preoperative to postoperative period, with no deterioration between six months and two years.
Conclusion:
This study demonstrates that favorable outcomes can be achieved by using an expandable titanium cage in TLIF procedures.
1. INTRODUCTION
Degenerative lumbar disc disease is a common cause of symptomatic low back pain and lumbar spine pathologies, such as spinal stenosis. Neural decompression and spinal column reconstruction are the foundations of surgical treatments for lumbar pathologies. Lumbar interbody fusion is a widely accepted technique for restoring spinal stability and alignment, as well as disc height [1, 2]. Sagittal alignment is of paramount importance in spinal arthrodesis [3]. Lumbar lordosis is an integral part of sagittal balance, and current evidence indicates that surgical correction leads to a secondary correction of the thoracic curve and sacral slope [4].
Transforaminal Lumbar Interbody Fusion (TLIF) is globally a popular surgical method, but it has not been conclusively shown to reconstruct lumbar lordosis [5]. The static intervertebral fusion cage is the device of choice in TLIF procedures, but TLIF cages must be large enough to achieve initial stability [6]. The facets, which provide extension and torsional stability as well as the support of the vertical load, must therefore be removed unilaterally to create sufficient space for an adequate implant insertion [6, 7].
Surgical access to the disc space during conventional TLIF surgery is limited and may be challenging due to the anatomy of the neural structures. TLIF is therefore associated with an increased risk of dural injury or nerve root violation during implantation. The narrow neural corridor may result in undersizing of the cage, while the small footprint of the cage may increase the risk of subsidence [8]. The expandable cage is a recent innovation that is thought to require less posterior bony removal and nerve root retraction compared with static cages [5]. The major advantages of expandable cages are that they are expanded after passing the neural structures and being placed inside the disc space. Due to the particular distraction mechanism, they may allow the creation of a greater lordotic angle and a greater lordosis restoration [8].
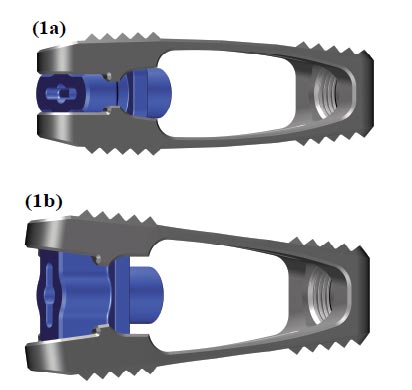
Several expandable lordotic interbody cages can be used in TLIF. In this study, we used the VERTACONNECT (SIGNUS Medizintechnik GmbH, Alzenau, Germany) implant, which is a TLIF cage made of biocompatible titanium alloy (Fig. 1). The aim of this study was to assess the effectiveness of this cage in a clinical setting. We hypothesized that the design of this cage would facilitate an improvement in both segmental lordosis and the restoration of the intradiscal height.
2. MATERIALS AND METHODS
The local institutional ethics committee approved this prospective study, and all patients provided informed consent prior to the study commencement. Eligible patients were those between 18 and 90 years of age with lumbar degenerative disc disease and with an indication for mono- or multisegmental spondylodesis of the lumbar spine.
Absolute contraindications included: existing fusion of the lumbar spine; post-traumatic deformities; osteoporotic fractures and resulting deformities; and osteolysis as a result of tumors, metastasis, or inflammation. Patients who met the eligibility requirements and gave their informed consent were consecutively enrolled in the study.
Scores for the Oswestry Disability Index (ODI) [9], Euro Qol 5D (EQ-5D) [10], and a back and leg pain scale (Nume- ric Rating Scale [NRS], with scale graduation of 0 to 10) were recorded preoperatively and at one week, six months, and 24 months postoperatively.
After six months, a radiological follow-up was performed with a thin-layer Computed Tomography (CT) scan (1 mm cuts) to assess fusion. The presence of fusion was based on Bridwell’s criteria [11]. The presence or absence of bony bridging and the determination of fusion were based on the consensus of two reviewers (two senior spine surgeons).
Standing plain radiographs were taken one week postoperatively as well as during the six- and 24-month follow-up visits. Disc height was determined radiographically and calculated as the mean of the anterior, middle, and posterior disc heights. Spondylolisthesis was determined in accordance with a measuring method advocated by Danielson [12]. Cage subsidence was defined as a disc space height loss ≥ 1 mm alongside a visible fracture of the vertebral body endplate [13].
The implant used was an expandable TLIF cage with a rectangular geometry (Fig. 1). The cage is available in different lengths and heights and with lordotic angles varying from 3 to 15°. Anterior height varies from 9 to 19 mm.
Extra attention was paid during the surgery to insert a sufficient amount of bone substitute material into the disc space inside and around the cage. Autologous bone obtained during decompression was preferred.
The cage was positioned horizontally across the anterior column, followed by an expansion in height and in lordosis. The cages were also designed in such a way that they were placed on the ventral and dorsal part of the cortical ring of the vertebral body when the length was optimal and the implantation was correct. The design of the cages resulted in stable bi-apophyseal support, which potentially prevents subsidence.
The surgical approach was carried out using a standard technique [14, 15]. A unilateral facetectomy was performed and the disc was exposed via a standard TLIF approach. After incision and cleaning of the disc space, the implant was inserted in a non-expanded state in an oblique manner. The dura and nerve roots were protected with retractors while inserting the cage. The contralateral facet joint was not directly altered during surgery, but it was decorticated to place bone graft material and induce a posterior fusion at the contralateral side. All patients were treated using a standard supplemental screw-rod system.
2.1. Statistical Analysis
Categorical variables are presented as frequencies and percentages, while continuous data are presented as mean and standard deviation. Analyses of the longitudinal data were based on mixed linear models. Wald tests were used to evaluate differences in the outcomes for each single time point. Two-sided p-values < 0.05 were considered statistically significant. Stata/SE 15.1 (StataCorp, College Station, TX, USA) was used for the statistical analysis.
3. RESULTS
A total of 32 patients with 40 operated segments underwent surgery between March 2014 and July 2015 and were included in this prospective observational study. Of those patients, 25 received monosegmental treatment, six patients were treated bisegmentally, and one was treated trisegmentally. The segments treated were the intervertebral L4–L5 disc spaces with a total of 23 applications, L3–L4 with 15 applications, and L2–L3 with two applications. The baseline characteristics of the population are presented in Table 1. The number of patients re-examined at each follow-up is presented in Fig. (2).
3.1. Reoperations
One patient was revised for screw loosening and one for pseudarthrosis, at five and six months after index surgery, respectively. One patient required surgical removal of a hematoma, while another patient required a wound revision. One patient underwent reoperation with an extension of the fusion due to a fracture in an adjacent segment.
3.2. Radiographic Results
Pseudarthrosis was observed in a single segment in one patient. All other patients showed the beginning of or complete fusion. Overall, the fusion rate across all study patients and all operated segments was 97.5% (Fig. 3). In four segments (four patients), an endplate violation was noted by intraoperative fluoroscopy during the placement of the cage. Further radiographic follow-up also revealed cage subsidence in four segments (four patients).
Radiographic analysis showed significant improvement in the degree of spondylolisthesis from the preoperative to postoperative period (p < 0.001 for one week and two years) (Table 2). No significant differences were found in the global lordotic angle between the preoperative and postoperative periods. Compared with the preoperative measurement, seg- mental lordosis was improved one week postoperatively, with no significant changes noted between the first postoperative week and the two-year follow-up. A significant increase in the disc height was reported, with no significant changes between the first postoperative week and the two-year follow-up.
| Variable | Value |
|---|---|
| Age (Years) | 69.0 ± 10.6 (42.9–85.3) |
| Sex (M : F) | 18 : 14 |
| No. of Segments | - |
| - 1 | 25 (78.1) |
| - 2 | 6 (18.8) |
| - 3 | 1 (3.1) |
| Total number of segments | 40 |
| Segments | - |
| - L2-L3 | 2 (5.0) |
| - L3-L4 | 15 (37.5) |
| - L4-L5 | 23 (57.5) |
| Timepoint | Item | Spondylolisthesis | Lordotic Angle | Segmental Lordosis | Disc Height, Ventral [mm] | Disc Height, Ventral [mm] |
|---|---|---|---|---|---|---|
| Preoperative (n = 40) | Mean (95% CI) | 18.0 (14.8–21.2) | 46.6 (42.2–51.0) | 14.5 (12.1–16.9) | 4.0 (3.4–4.6) | 6.3 (5.4–7.2) |
| 1 week (n = 40) | Mean (95% CI) | 2.5 (-0.7–5.8) | 46.9 (42.5–51.4) | 16.7 (14.3–19.1) | 9.6 (9.0–10.3) | 14.4 (13.5–15.3) |
| p-value | < 0.001 | 0.883 | 0.016 | < 0.001 | < 0.001 | |
| 6 months (n = 39) | Mean (95% CI) | - | 42.7 (34.6–63.8) | 15.1 (11.8–18.4) | 9.8 (8.8–10.8) | 14.0 (12.5–15.6) |
| p-value | 0.137 | 0.288 | 0.743 | 0.661 | ||
| 2 years (n = 19) | Mean (95% CI) | 3.7 (0.6–6.8) | 41.4 (36.1–46.8) | 16.7 (13.9–19.6) | 9.0 (8.2–9.8) | 13.3 (12.1–14.6) |
| p-value | 0.478 | 0.673 | 0.321 | 0.141 | 0.438 |
| Timepoint | Item | NRS Back Pain | NRS Leg Pain | ODI | EQ-5D | EQ-5D (VAS) |
|---|---|---|---|---|---|---|
| Preoperative (n = 32) | Mean (95% CI) | 6.5 (5.6–7.4) | 6.2 (5.2–7.1) | 40.4 (33.9–46.9) | 0.47 (0.37–0.57) | 52 (43–60) |
| 1 week (n = 32) | Mean (95% CI) | 4.3 (3.4–5.2) | 3.8 (2.8–4.7) | 47.5 (40.8–54.2) | 0.54 (0.44–0.65) | 51 (43–60) |
| p-value | < 0.001 | < 0.001 | 0.093 | 0.299 | 0.939 | |
| 6 months (n = 31) | Mean (95% CI) | 3.4 (2.5–4.4) | 2.9 (1.9–4.0) | 30.6 (23.3–37.8) | 0.72 (0.62–0.83) | 64 (55–73) |
| p-value | 0.139 | 0.199 | < 0.001 | 0.012 | 0.028 | |
| 2 years (n = 14) | Mean (95% CI) | 3.6 (2.5–4.8) | 3.4 (2.2–4.8) | 24.3 (15.7–33.0) | 0.85 (0.71–0.98) | 59 (47–71) |
| p-value | 0.788 | 0.516 | 0.246 | 0.150 | 0.509 |
3.3. Clinical Results
The mean pain, functional, and quality of life outcomes improved postoperatively (Table 3). In terms of disability and health-related quality of life but not for back or leg pain a significant improvement occurred from one week postopera- tively to six months. No significant changes in any of the out- comes were seen between six months and two years postope- ratively.
4. DISCUSSION
The radiographic results of this study demonstrate a favo- rable medium-term follow-up review following the insertion of an expandable TLIF cage in patients with lumbar degenerative disc disease who had undergone one-level to three-level TLIF procedures. Three primary radiographic objectives were achie- ved using this lordotic, expandable cage: the restoration of the intradiscal height without significant subsidence; a reduction in the degree of spondylolisthesis; and an improvement in the segmental lordosis.
Overall, the rate of fusion in the present study, as verified by CT, was in line with the rates reported in previous clinical research on static cages [16]. A recently published meta-analysis of level III and level IV studies reported a pooled fusion rate of 93% for lumbar interbody fusion for degenerative spine diseases [16]. This lordotic change shows potential for significant segmental correction within the sagittal plane, with a mean lordosis improvement of 2.2° at the two-year follow-up. In terms of correction of the sagittal alignment, TLIF procedures using static cages yield variable outcomes. While some studies report similar degrees of segmental correction in the sagittal plane to those reported here [5, 17-19], others report a lesser degree of correction [20, 21].
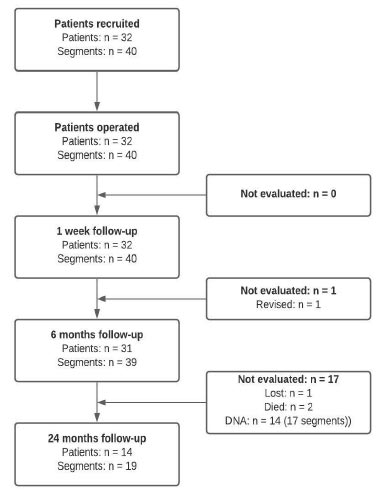
Abbreviations: DNA, did not attend.
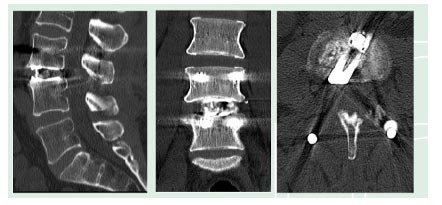
Sagittal view (left) and Coronal view (middle). Axial view shows cage contact on the ventral and dorsal part of the cortical ring of the vertebral body (right).
Although these findings are positive, an important point to note is that the literature has not yet established whether improvement in lumbar lordosis translates into better clinical outcomes for the treatment of spondylolisthesis [22]. Similarly, height restoration, which was also improved in this study, is unproven in terms of its clinical efficacy. Regarding the sagittal alignment parameters, because the vast majority of patients only had one-level pathology, there was unlikely to be a significant degree of sagittal imbalance preoperatively, and thus we did not expect to significantly alter the overall sagittal balance postoperatively.
In general, the results of this study are consistent with those of previous studies that have demonstrated clinical improvements in back pain, leg pain, and disability after TLIF. One systematic review and meta-analysis reported similar reductions in NRS back and leg pain as well as in the ODI [23]. On average, the improvements in back disabilities in this study were greater than the published minimum clinically important difference for the ODI [24] and demonstrated a sustainable reduction throughout the follow-up period.
For this particular implant, a noteworthy detail is that, historically, some doubts have been raised regarding the use of expandable interbody cages in relation to the endplate violation and subsidence due to the edge loading seen with this cage type [25]. In our series, this was shown in four study patients with endplate violation and four cases where radiographic subsidence was observed, but none of these was clinically significant (i.e., resulting in recurrent symptoms or requiring a reoperation). A reported risk factor of cage subsidence and endplate fracture is low bone mineral density, as seen in osteoporosis [26]. Based on these observations, as the study progressed, we became more conservative in our approach with osteoporotic patients, and we now prefer using static cages for them as well as when we have intraoperative doubts regarding the density of their endplates.
A notable finding was the observation of no significant deterioration between the first postoperative week and two years, either in terms of clinical or radiographic outcomes. This observation is consistent with the purported advantages of the cage, i.e., its provision of bi-apophyseal support. However, the absence of a control group in our study precludes any verification of this.
This study has some notable limitations. One was the substantial attrition between six months and two years postoperatively. Another was that this was a monocenter study with a relatively small sample size. A third limitation was that the mean follow-up period of two years requires further long-term follow-up to demonstrate the sustainability of the radiographic findings beyond two years. Future studies should evaluate whether an increase in segmental correction hinders the development of adjacent-segment degeneration and reduces the need for additional interventions.
CONCLUSION
In conclusion, this study has demonstrated that favorable outcomes can be achieved by using an expandable titanium cage in TLIF procedures. Compared with the historically published data, the use of technology like this demonstrates the potential for additional segmental lordotic restoration. However, whether improved clinical outcomes in the longer term will result from these radiographic differences remains to be seen.
AUTHOR'S CONTRIBUTION
Idea,design and planning of the study (K.K., C.F., M.A., M.Ra. M.Ri); data collection (K.K.); analysis and interpretation of data (K.K., C.F., M.A., M.Ri.); writing the article (K.K., B.W., S.H., M.Ri.). All authors read and approved the manuscript.
LIST OF ABBREVIATIONS
| CI | = Confidence interval |
| CT | = Computed tomography |
| DNA | = Did Not attend |
| F | = Females |
| EQ-5D | = Euro Qol-5D |
| M | = Males |
| NRS | = Numeric Rating Scale |
| ODI | = Oswestry Disability Index |
| TLIF | = Transforaminal lumbar interbody fusion |
| VAS | = Visual Analog Scale |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The ethics committee of the Johann Wolfgang Goethe-University Frankfurt am Main, Germany approved this prospective study (reference number: 399/13).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
All patients provided written consent.
AVAILABILITY OF DATA AND MATERIALS
Data and Materials are archived at Orthopaedic University Hospital Friedrichsheim, Frankfurt a.m., Germany.
FUNDING
None
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
None declared.


