All published articles of this journal are available on ScienceDirect.
Persistent Wound Drainage After Tumor Resection and Endoprosthetic Reconstruction of the Proximal Femur
Abstract
Purpose:
To examine the prevalence of prolonged wound drainage (PWD) after tumor resection and endoprosthetic reconstruction of the hip.
Methods:
Retrospective review of 86 consecutive patients with metastatic bone disease, malignant hematologic bone disease or bone sarcoma, treated with tumor resection and subsequent endoprosthetic reconstruction of the proximal femur, between 2010 and 2012, in a single center.
Results:
PWD for 7 days or more was observed in 41 cases (48%). The wounds only ceased oozing after a mean of 8.4 days, leading to prolonged administration of prophylactic antibiotics (mean 8.7 days) and length of hospital stay (mean 10.2 days). Total femur replacement, bone sarcoma and additional pelvic reconstruction were identified as significant independent risk factors for an even longer duration of PWD.
Conclusion:
Compared to conventional hip arthroplasty, PWD appears to be significantly more prevalent in patients undergoing tumor arthroplasty procedures of the hip. Given the potentially increased risk for periprosthetic joint infection (PJI), increased awareness, identification and implementation of adequate strategies for prevention and treatment of this avoidable complication are warranted.
INTRODUCTION
Patients, who undergo endoprosthetic reconstruction subsequent to malignant bone tumor resection, are at high risk of prosthetic joint infection (PJI) (10-11%), as established in two large reviews [1, 2]. Deep infection is a devastating complication, which may require further revision surgery, prolonged hospitalization, antibiotic treatment and rehabilitation. Moreover it exposes the tumor patient in particular, to significant further risks and complications, such as amputation [1-3] and may interfere with adjuvant radio- or chemotherapy, possibly even compromising overall survival. The substantial cost associated with treatment of these complications has also been well documented [4]. Prolonged wound drainage (PWD) is a well known predisposing risk factor for surgical site infection [5-9] and may result in prolonged hospital stay and delay of adjuvant therapy. While reported to occur in about 4% of conventional hip arthroplasties [10], PWD appears to be considerably more frequent in our tumor patients and as a consequence, it is our practice, to neither discharge the patient, nor to discontinue prophylactic intravenous antibiotics, before the surgical wound is dry. This study was conducted to investigate and quantify the prevalence of PWD, the duration of administration of postoperative intravenous antibiotic prophylaxis and the length of the hospital stay after endoprosthetic reconstruction of the proximal femur performed in patients with a primary or secondary malignancy of bone.
MATERIALS AND METHODS
Study Design and Patient Population
We retrospectively reviewed the medical records of all patients with metastatic bone disease, malignant hematologic bone disease and bone sarcoma, who underwent endoprosthetic reconstruction of the proximal femur in our specialized orthopedic oncology unit between 2010 and 2012. All relevant data (age, gender, nature and location of pathology, details of the surgical procedure, implants used, duration of antibiotic treatment, preoperative radiation therapy, hospital stay and time to a dry surgical wound) were collected from the patient files. The primary indication for surgical treatment in the 86 patients (mean age=64 years, M/F = 40/46), included in our study, was de-facto pathological fracture (n=48), impending pathological fracture (n=29) and bone sarcoma (n=9) of the proximal femur (Table 1). In 12 patients (14%), substantial concurrent lesions of the acetabulum or the distal femur were present, requiring a more extensive surgical procedure, either acetabular reconstruction with a cemented partial pelvic replacement (n=7), (Fig. 1B), or total femur replacement (n=5). Complete data for patient survival and duration of hospital stay was available in all cases and in 83, for wound status and duration of antibiotic treatment. In four patients, who required transfer to local hospital (for social reasons, n=3) or another department within our hospital (for treatment of a cerebral abscess, n =1), in spite of modest continued wound drainage, we considered the wound dry and antibiotics discontinued on the day of discharge from our unit, to avoid inadequate overestimation of these parameters. Despite multiple operative wound revisions for continuous drainage, one patient progressed to chronic infection and was ultimately discharged to hospice on post-op day 43 on long-term antibiotics with a draining sinus. In eight cases antibiotics had to be continued after the surgical wound was dry, due to other causes: infection in the chest (n=3), gastrointestinal tract (n=2), urinary tract (n=1), brain (n=1) and one unknown primary focus. In these, antibiotics were considered discontinued the same day as the surgical wound was dry. The study was approved by the Danish Data Protection Agency (no. 2013-412591), but approval from an Ethics Committee is neither possible, nor required in our country for studies based on review of medical records only.
Demographics and pathology of all patients treated with tumor resection and endoprosthetic reconstruction of the hip between 2010 and 2012.
| Number of Patients | 86 |
| Female/Male | 46/40 |
| Mean Age at Surgery (years) (range) | 64 (35-92) |
| Primary Tumor Breast Lung Kidney Sarcoma Prostate Myeloma Lymphoma Colon Unknown Esophagus Bladder Stomach Anal Oncocytoma |
26 15 9 9 6 6 3 3 3 2 1 1 1 1 |
| Indication for Surgery Pathological Fracture Impending Pathological Fracture Bone sarcoma |
48 29 9 |
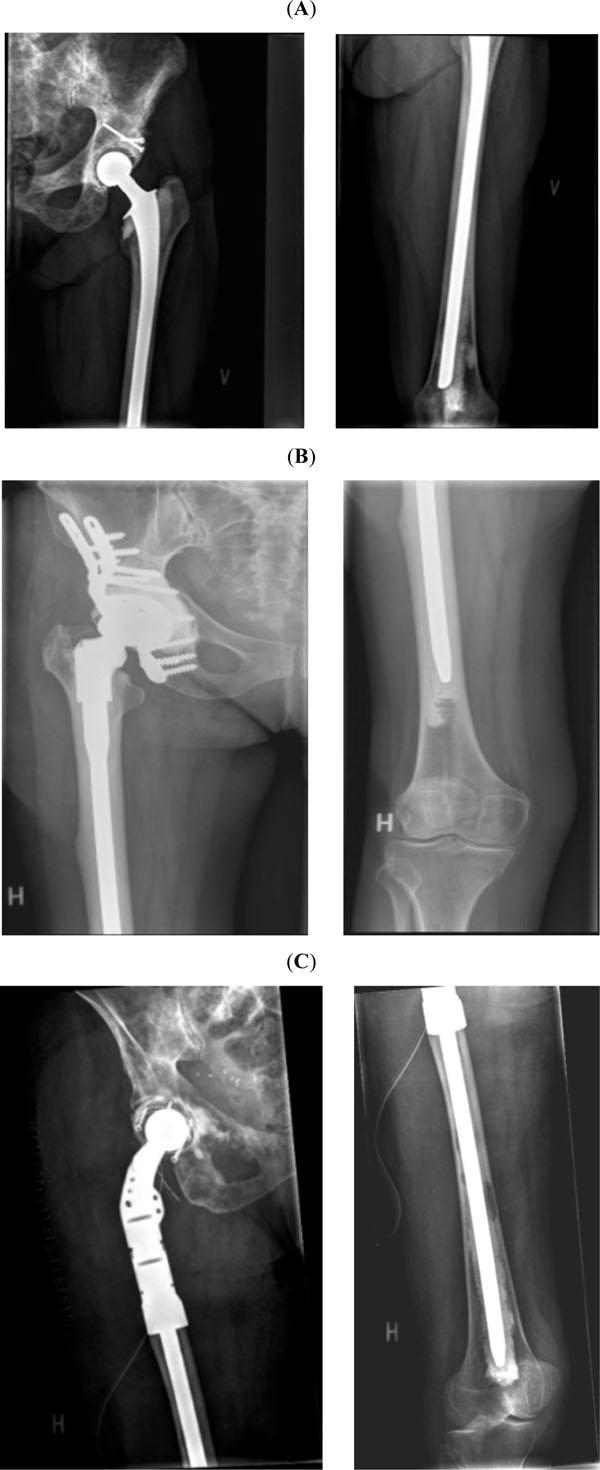
A: Conventional neck resection (for a pathological femoral head fracture), reconstructed with a full length (350mm), cemented standard stem (Link SPII), to bridge metastatic involvement of the distal femur. Note screw fixation and cement augmentation for a partially healed pathological fracture in the superior acetabular rim. B: Conventional calcar resection (for a pathological femoral neck fracture) and reconstruction of the proximal femur with a cemented modular revision prosthesis (Link MP). Note reconstruction and cement augmentation of a large concurrent acetabular lesion with a pelvic reconstruction cage (Link Partial Pelvic Replacement). C: Proximal femur resection (15cm), reconstructed with a long, cemented mega-prosthesis (Zimmer Segmental System) and a cemented (Link Lubinus Eccentric) acetabular component. Note screw fixation of a polyethylene anti luxation device.
Surgical Procedure and Post-Operative Routine
A routine posterior approach to the hip was employed in all patients. In 38 patients confinement of the tumor to the femoral head or neck allowed a conventional neck resection, preservation of the abductor mechanism and endoprosthetic reconstruction with a cemented standard stem (Biomet Bimetric (n=13)) or a long (200 mm or longer) cemented standard stem (Link SP2 (n=17), or Zimmer CPT (n=1)) (Fig. 1A). The remaining 45 patients required an extended posterior approach, to accommodate the necessary proximal femoral resection (mean resection length 124mm (54-220mm)) and endoprosthetic reconstruction with either a modular revision stem (Link MP (n=33) and Zimmer ZMR (n=1)) (Fig. 1B) or a tumor megaprosthesis (Zimmer Segmental (n=8), Stryker GRMS (n=6), Link Mega C (n=3)) (Fig. 1C). All stems were cemented with the exception of the GMRS stems (short fluted with proximal HA coating), which were used in the youngest patient group after resection of primary bone sarcomas. On the acetabular side, with exception of seven multipolar hemiarthroplasties, the majority of patients received a cemented acetabular component (Lubinus Excentric, (n=75)), or an uncemented cup (Stryker MDM (n=2), Zimmer Trilogy (n=2)). Seven cases required additional peri-acetabular tumor removal and pelvic reconstruction with a cemented pelvic reconstruction ring (Link partial pelvis replacement (Fig. 1B) and in 5 cases of metastatic disease involving the entire femur, total femur replacement, Stryker GRMS total femur (n=1) and Link Mega C total femur (n=4) was required. All incisions were closed in a layered fashion over deep drains, after detached musculature had been reattached to the prosthesis, in an attempt to restore the abductor mechanism and cover the entire prosthesis with vital tissue. Postoperatively all patients were mobilized, weight bearing as tolerated, from post-operative day 1. The sterile compressive dressing applied at conclusion of the procedure was left unchanged until day 2 or 3, to coincide with removal of any surgical drains still present. Prophylactic intravenous antibiotics (Cefuroxime 1.5g x 3) were started 15 to 30 minutes prior to incision and not discontinued before a senior member of the surgical team considered the wound dry. Thromboprophylaxis with Tinzaparin 3500-4500 IE x 1 sc. was maintained until the patients were well mobilized, at least until discharge from hospital.
Statistical Analysis
All data are presented as mean with total range, and p values of <0.05 considered significant. We used standard IBM SPSS software (version 19) for the following statistical calculations: linear regression analysis with calculation of the coefficient of determination (R2) for evaluation of the relation between the main variables (time to dry surgical wound, duration of antibiotic treatment and hospitalization), non-parametric tests for un-paired data (Mann-Whitney) for comparison of subgroups, and Kaplan-Meier survival analysis for estimation of overall patient survival. Calculation of 95% confidence intervals for the survival data was done in Microsoft Excel 2010 using Greenwood’s formula for calculation of standard error.
RESULTS
Overall Survival
As an indirect measure for severity of the underlying disease, we performed an analysis of overall patient survival. Ten patients died within the first 30 post- operative days and probability of overall survival was 74% at 3 months, 63% at 6 months and 45% at 1 year (Fig. 2), indicating that most patients were in advanced stages of their underlying disease.
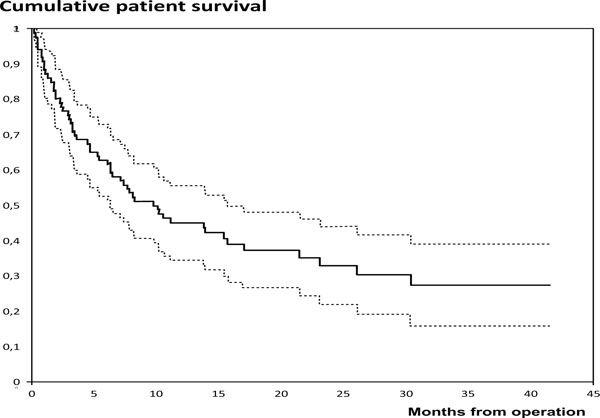
Kaplan Meier survival analysis showing cumulative survival rate (solid line) and 95% confidence interval (dotted lines) for all patients (n=86), who had an endoprosthetic reconstruction of the hip and proximal femur for malignant bone disease between 2010 and 2012.
Prevalence of Prolonged Wound Drainage
Prolonged wound drainage of 7 days or more was observed in 41 cases (48%), and on average, the surgical wound was dry after a mean of 8.4 days (3-24 days), (Fig. 3). When focusing on selected subgroups, we found an even longer duration of PWD in patients with total femur replacement (p=0.005), a primary diagnosis of bone sarcoma (p=0.047) and in patients who required additional pelvic reconstruction (p=0.058). We found no association of wound drainage with overall survival, the extent of the proximal femoral resection or whether the patients had received preoperative radiotherapy or not (Table 2).
Specific analysis of duration of wound drainage of relevant patient subgroups.
| Days [Mean (Range)] | P-Value | |
|---|---|---|
| All patients with data for day of dry surgical wound (n=83) | 8.4 (3-24) | |
| Survival < 6 months (n=31) Survival > 6 months (n=52) | 9.1 (3-24) 8.0 (3-20) | P = 0.81 |
| Proximal femoral resection (n=53) Conventional neck cut (n=30) | 8.5 (3-24) 8.2 (3-20) | P = 0.46 |
| Pelvic reconstruction (n=7) No pelvic reconstruction (n=76) | 12.9 (4-24) 8.0 (3-20) | P = 0.058 |
| Sarcoma (n=9) Metastasis or hematologic (n=74) | 11.4 (5-20) 8.0 (3-24) | P = 0.047 |
| Total femur (n=5) No total femur (n=78) | 13.6 (10-16) 8.1 (3-24) | P = 0.005 |
| Preoperative radiotherapy (n=23) No preoperative radiotherapy (n=60) | 8.9 (3-24) 8.2 (3-20) | P = 0.65 |
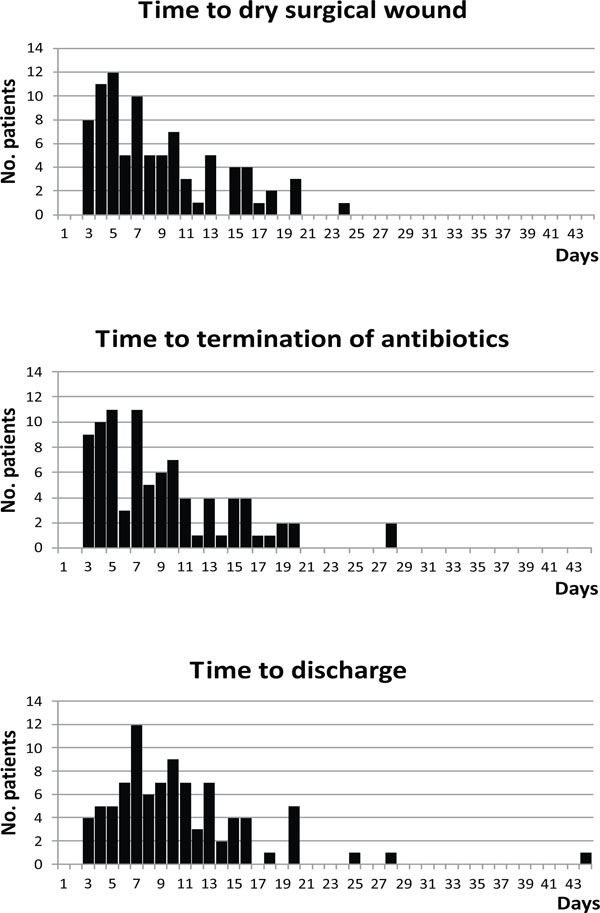
Patient distribution with regard to the day of dry surgical wound, the length of antibiotic treatment, and the length of admission.
Duration of Antibiotic Prophylaxis and Hospital Stay
Prophylactic antibiotics were administered for an average of 8.7 days (3-28 days) and mean length of hospital stay was 10.2 days (3-44 days) (Fig. 3). Linear regression analysis showed a highly significant relation of wound drainage with the duration of antibiotic treatment (p<0.0005, R2=97%) and the length of hospital stay (p<0.0005, R2=74%).
DISCUSSION
The prevalence of PWD after malignant bone tumor resection and endoprosthetic reconstruction of the proximal femur has not been specifically determined previously. We found a surprisingly high rate and duration of PWD, involving almost every other patient in our cohort. Given the substantial increase in infection risk of 42% with each day of persistent wound drainage, reported by Patel et al. [5] for conventional THA, a PWD over 7 days, observed in 48% of our patients, could reasonably be expected to further increase the already elevated risk for PJI in this patient population. Not surprisingly, we were able to demonstrate a significant correlation of PWD with duration of antibiotic prophylaxis and length of hospital stay, which indicates that each occurrence of this, obviously not so infrequent complication, directly resulted in increased cost of treatment, even if later infection was prevented.
Compared to conventional arthroplasty, the considerably higher rate of PWD observed in our study, can likely be attributed to a combination of multiple factors. Increasing extent and complexity of the surgical procedure, with larger areas of soft tissue dissection, bone resection, implant dimension, increased blood loss and prolonged operating time, may well explain why total femur replacement, additional pelvic reconstruction and resection of a primary bone sarcoma showed a higher prevalence and duration of PWD. However, these factors were apparently not relevant enough, to lead to a detectable difference between the groups receiving a proximal femur resection compared to a standard resection of the femoral neck in our relatively small patient cohort.
Undoubtedly, patients undergoing tumor arthroplasty exhibit a substantially higher profile of recognized risk factors, compared to routine arthroplasty patients, due to the burden of the primary disease, its associated co-morbidities, such as malnutrition [11,12], immunosuppression [13,14], impaired renal or liver function and prior and on-going chemotherapy and/or radiotherapy. However, the impact of these factors on our relatively small sample was not significant enough to demonstrate an association between postoperative survival and PWD, even though a higher degree of co-morbidities and severity of disease might have been expected in patients with shorter postoperative survival.
Furthermore, the ultimate manifestation of an actual PJI is of course directly dependent on the quantity and virulence of bacteria that reach and colonize the operative site and the efficacy of preventive measures (or lack thereof). There is wide disparity of practice with regard to type of prophylactic antibiotic as well as length of its administration and there is insufficient evidence for firm recommendations. Despite this lack of sufficient evidence, reportedly high infection rates of up to 35%, even higher reinfection rates as high as 43% [15] and the often dire consequences of PJI resulting in amputation in up to 35% in some series [3], a recent multinational consensus meeting on PJI (led by the Musculoskeletal Infection Society and the European Bone and Joint Infection Society), recommended the use of routine antibiotic prophylaxis not exceeding 24h, for patients undergoing major reconstruction with mega-prosthesis [15]. A position statement published on the Musculoskeletal Tumor Society (MSTS) website appears to express the opposite opinion, acknowledging the unique antibiotic requirements of tumor patients, potential for unacceptable adverse outcomes and costs associated with treatment of an infected limb salvage joint replacement. They explicitly discourage routine application of standard prophylactic measures to patients with bone and soft-tissue tumors undergoing limb salvage surgery. Instead, they recommend that musculoskeletal oncologists should be supported in their clinical judgment with regards to choice of adequate type and length of post-operative antibiotic prophylaxis for their tumor patients on an individual basis [16]. Definitive high level evidence to resolve this controversy will probably not become available for many years, as a randomized, controlled, international multicenter trial, to determine the role of long term antibiotics in patients undergoing surgical excision and endoprosthetic reconstruction of a primary bone tumor, has only recently started enrolment of an anticipated total of 920 patients [17]. Meanwhile, a recent systematic review of 48 studies including a total of 4838 adult patients having undergone long bone tumor resection and endoprosthetic reconstruction, reported significant reduction in pooled infection rates with long term postoperative antibiotic prophylaxis [1], which supports our current practice. Due to consistent use of extended antibiotic prophylaxis, the infection rate in this high risk population has remained very low at our institution, for over a decade and while it may appear tempting to push for ever shorter durations of hospital stay and antibiotic prophylaxis, short sighted attempts to capitalize on immediate cost savings may well not be of long term benefit to either patient or health economy. Undoubtedly, a reduction of PWD is desirable and should diminish the risk for PJI, as well as the need for prolonged use of prophylactic antibiotics and hospitalization in a highly specialized ward. However, although strategies to reduce PWD should be sought and investigated, their implementation should be carefully monitored, to avoid interference with the potentially beneficial effect of long term prophylactic antibiotics.
We recognize the limitations of our study, inherent to retrospective data collection in general. In particular, we acknowledge some degree of uncertainty about the exact day the surgical wound was considered dry, as the surgical wounds were not inspected daily and neither precise quantification of significant drainage, nor an exact definition of what constituted a dry surgical wound were used. From the wide range of definitions of persistent or prolonged wound drainage used in the literature, we have decided to use and report the most conservative PWD of 7 days or longer, which would be considered abnormal by most orthopedic surgeons. As such, we consider our results sufficient to demonstrate the existence and the scope of an obvious problem and to indicate a need for raised awareness and efforts to identify, develop and implement effective preventive and therapeutic measures to prevent this complication.
CONCLUSION
Prolonged wound drainage (PWD) may be significantly more prevalent in patients undergoing endoprosthetic reconstruction of the hip for malignant bone disease involving the proximal femur, than reports suggest for conventional hip arthroplasty. This may increase the already substantially elevated risk profile of this patient population even further. The occurrence of this complication may prompt prolonged administration of antibiotic prophylaxis, prolonged hospital stay or even revision surgery. Total femur resection, bone sarcoma and pelvic reconstruction were found to be significant independent risk factors for even further persistence of PWD. Given the potentially increased risk for PJI in particular, increased awareness of this complication is recommended and further research directed at identification and implementation of adequate strategies for its prevention and treatment is warranted.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest related to the content of this article.
ACKNOWLEDGEMENTS
Declared none.


