All published articles of this journal are available on ScienceDirect.
The Use of Megaprostheses for Reconstruction of Large Skeletal Defects in the Extremities: A Critical Review
Abstract
In the case of primary malignant tumors, extensive metastatic disease, major trauma or end-stage revision arthroplasty, the orthopaedic surgeon often has to deal with the need to reconstruct large skeletal defects, or replace bone of low quality. In the past years this was frequently impossible, and the only solution was amputation of the extremity. Later, the introduction of custom-made endoprostheses capable of reconstructing large skeletal defects, also known as megaprostheses, allowed for sparing of the extremity. This was especially valuable in the case of oncologic orthopaedic surgery, as advances in the medical treatment of sarcoma patients improved prognosis and limb-preserving surgery proved to have comparable patient survival rates to amputation. However, custom-made designs were implicated in frequent mechanical failures. Furthermore, they were extremely difficult to revise.
The introduction of modular endoprostheses in the 1980s marked a new era in orthopaedic oncologic surgery. Modular megaprostheses consist of a number of different components in readily available sets, which can be assembled in various combinations to best address the specific bone defect. Moreover, they proved to have considerably lower rate of mechanical failures, which were also much easier to address during revision surgery by replacing only the parts that failed. The functional outcome after reconstruction with megasprostheses is often very satisfactory and the patient can enjoy a good quality of life. Nowadays, the major challenge is to eliminate the rate of non-mechanical complications associated with surgery of that magnitude, namely the risk for wound dehiscence and necrosis, deep infection, as well as local recurrence of the tumor.
In our present mini-review, we attempt to make a critical approach of the available literature, focusing on the multiple aspects of reconstructive surgery using megaprostheses. We present the evolution of megasprosthetic implants, the indications for their use, and describe the outcome of surgery, so that the non-specialized orthopedic surgeon also becomes familiar with that kind of surgery which is usually performed in tertiary centers. A special interest lays in the recent developments that promise for even better results and fewer complications.
INTRODUCTION
Oncologic orthopedic surgery had long been confined to amputation in order to remove malignant tissue and avoid recurrence and metastases. However, in the case of primary bone tumors, the advent of adjuvant therapies (radiation and chemotherapy) helped to dramatically improve patient survival and local tumor control so that limb salvage surgery could compare to amputation and become the gold standard of current treatment [1-3]. In the frame of radical excision surgery, special segmental bone and joint replacement systems were developed, that are usually referred to as tumor endoprostheses or megaprostheses. The rationale behind their use subsequently led to application even in revision arthroplasty with great loss of bone stock, as well as in trauma surgery, for extremely comminuted fractures with poorly vascularized fragments, where traditional osteosynthesis cannot be applied.
Total femur reconstruction was first described by Buchman in the middle of the 20th century. However, it was around the 70s when use of megaprostheses started to appear more often in literature and by the 90s they were becoming increasingly popular [4-7]. The term itself “megaprosthesis” seems to have been first used in The International Workshop on Design and Application of Tumor Prosthesis, held in Mayo Clinic in 1981.
THE EVOLUTION OF MEGAPROSTHESES. CUSTOM MADE AND MODULAR DESIGNS
Tumor endoprostheses in their beginning came usually in custom-made monoblock form made of cast steel alloys (eg vitallium). Material evolution continued to titanium and cobalt-chrome-molybdenum alloys while in the meantime experimenting with various acrylic polymers, which though failed prematurely and were abandoned. In general, the requisites for a successful implant design are resistance to corrosion, high biocompatibility and resistance to fatigue fractures as well as potential for osteointegration and soft tissue ingrowth. Additional qualities like infection repelling are also more than desired. Metallurgy and synthetic material industries are providing a variety of coatings (hydroxyapatite, porous tantalum, elemental silver) and soft tissue attachment materials (polyester, polypropylene, carbon fiber, expanded polytetrafluorothylene) to address these requisites in an improving manner.
Prosthesis design has also evolved from the monoblock and fixed hinge models to modular endoprostheses and rotating platforms, with improved geometry to enhance fixation and stability [7-9].
Modular endoprostheses are currently and since the 1980s almost dominating surgical practice. They consist of a number of different components in readily available sets. These components can be assembled in various combinations to best address the specific bone defect of the patient in the operating theatre (Fig. 1). In this manner, surgery can proceed without the fabrication delay (4-6 weeks) of the custom made models. Importantly, more freedom of options is given to the surgeon in order to reconstruct defects that might prove to be different from what preoperative planning indicated. Literature supports superiority of modular implants even in terms of limb survival, complication rate and functional outcome.
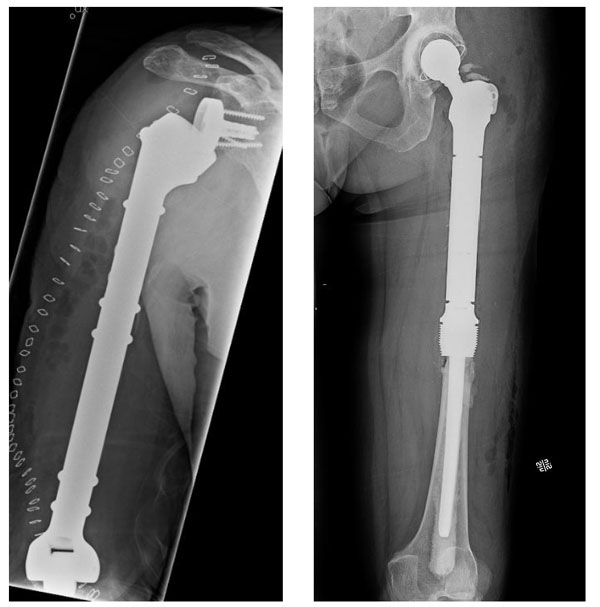
A case of reconstruction with a modular total humerus prosthesis (MUTARS) for an extensive destruction of the humerus with pathological fracture (left panel), and a case of proximal femur reconstruction using a modular METS prosthesis by Stanmore (right panel).
Fixation, as in regular prostheses, can be achieved with or without the use of polymethymethacrylate cement. In the case of cemented models, the limitation in weight-bearing depends most on the soft-tissue parts, and restrictions are sometimes implemented until there is reliable healing and attachment of the soft tissues to the prosthesis. Typically, orthoses are used for reconstructions around the knee joint and the shoulder. In the case of the hip, the patient can often be instructed immediate full-weight bearing. A main disadvantage of cemented implants is aseptic loosening, which has been reported to occur in up to one third of reconstructions around the knee joint. Recent advances in implant manufacturing are the use of hydroxyapatite coated collars in order to achieve osteointegration and minimize the risk of loosening, silver-coated stems in order to further reduce the infection rate and highly cross-linked polyethylene cups in order to minimize wear.
INDICATIONS FOR THE USE OF MEGAPROSTHESES
As the bulk of the literature testifies, reconstruction after resection of primary malignant bone tumors is the most established indication for use of megaprosthesis. Occasionally, even locally aggressive benign tumors of the bone (for example giant cell tumors) will necessitate the use of a megaprosthesis. Malignant soft-tissue tumors that are in close proximity or engage the bone sometimes require skeletal reconstruction, if a wide surgical margin is to be achieved. Obviously, the aggressive dissection associated with the proper removal of tumor tissue in these cases leaves large skeletal and soft tissue defects that regular prostheses cannot cover. The primary goals with this kind of limb salvage surgery are: a. to achieve disease control, with recurrence or metastasis risk no greater than with amputation, b. to not delay administration of adjuvant therapy and c. to provide a stable, functional and durable extremity without too many local complications.
In the same field of oncological diagnoses, metastatic tumors are also discussed as an indication for megaprosthesis [10]. Indeed, the mainstay of metastatic pathological fracture treatment has traditionally been osteosynthesis (with or without cement augmentation) and radiation therapy. However, failure due to non union, inadequate fixation due to poor bone quality, compromised vascular supply of the irradiated soft tissue or local progression of the disease reflect the inherent weakness of traditional fracture treatment, when applied to locally and systemically disturbed bone healing conditions. Megaprostheses on the other hand replace the affected bone tissue instead of trying to achieve bone healing. Immediate weight bearing is allowed, which even in patients with unfavorable prognosis might be a strong argument for the use of the method, since it provides for a good quality of life, as it bypasses the need for fracture healing.
Revision arthroplasty is the other expanding field of application for megaprosthetic systems [11]. In that case, the surgeon faces the problem of extensive bone loss or poor bone quality after sequential revision surgery, and the use of megaprosthesis appears to be a good rescue option. The available data in this context are nonetheless limited, as the cases where the orthopaedic surgeon decides to use a tumor endoprosthesis as a last resort when conventional revision arthroplasty fails are rare.
Finally, trauma surgery is for the time being another quite unexplored field when it comes to megaprostheses. Literature is quite sparse on reports regarding treatment of traumatic bone defects or recurrent non union [12]. Bone loss and concomitant severe soft tissue damage is the main problem undermining possibility of fixation in these conditions. Megaprostheses have also been used in the case of revision trauma surgery, such as cases of non-union of severely comminuted or periprosthetic fractures. Generally, when extreme bone loss occurs, skeletal reconstruction with a megaprosthesis is undoubtedly a good option, and according to our opinion should be considered early in the treatment arsenal of elderly patients, who have poor chances of healing a fracture.
FUNCTIONAL OUTCOME AFTER SURGERY
As mentioned above, the main goal of megaprostheses is to achieve disease control as effectively as amputation would and salvage the limb in a durable and functional way. It seems though that literature is more concentrated on reporting patient and limb survival and complications rather than functional outcome postoperatively. In a way, when malignancy treatments are evaluated the instinctive and initial focus lays on survival length. It is probably after the achievement of substantial increase in survival that questions arise regarding the quality of life. Complication rates are also probably thought to reflect one aspect of that parameter. In most studies the MusculoSkeletal Tumor Society (MSTS) score is used as measure of functional outcome. It is a widely validated tool that comprises of six sections, descriptively grading from 1 to 5 pain, general function, emotional acceptance and specific function for upper (hand positioning, dexterity, lifting ability) and lower extremity (walking ability, gait, supports). In most studies the result is expressed as a percentage and the literature presents results ranging quite consistently between 60-88% [10, 13-17].
There are very few studies concentrating in objective data regarding function after endoprosthetic surgery, such as oxygen consumption, muscle strength and activity levels [15, 18-21]. One such study reviewed by Bernthal et al., including patients that underwent reconstructions in the lower extremity, reports no significant difference in oxygen consumption and walking speed [17]. These patients have a satisfactory gait and activity level at home and at the community. However, they also report that patients who underwent proximal tibial reconstructions had reduced flexion and extension strength, a fact which probably reflects the effect of the attachment of the patellar tendon to the endoprosthesis. Generally, in the lower extremity, proximal and distal femur reconstructions have better functional results as compared to proximal tibia reconstructions [17, 22, 23].
In the upper extremity, reconstructions involving the distal humerus give satisfactory functional results [24-26]. In the case of proximal and total humeral resection, the patients generally have considerable limitations in shoulder movement as a result of rotator cuff resection and scar tissue formation in the area. As a rule, abduction is limited to below 90 degrees, and sometimes proximal migration of the humeral lead is observed [27, 28]. However, the function of the hand remains excellent [27].
Results can also be categorized according to the indication for surgery (tumor surgery or not), as soft-tissue resection is not the case for non-oncological operations. When megaprostheses are used in the context of revision arthroplasty for aseptic loosening, reported functional results according to Harris Hip Score were good, showing a considerable improvement after revision surgery [29]. As expected, these studies include mainly patients operated in the hip and proximal femur. Likewise, significant improvement of the Knee Society Score was reported in revision knee arthroplasty [11]. Similarly, the few studies that concentrate on the use of megaprostheses after trauma focus on the proximal femur and knee, and include severely comminuted periprosthetic fractures. The reported results also show good functional outcome after surgery [12, 30, 31].
When interpreting the results of such studies, it should be kept in mind that data collection is subject to a variety of factors affecting the end result, such as equipment and method evolution through the years as well as the inherent distortion of what might resemble everyday activity by testing in a laboratory. One conclusion though that could be drawn is that limb salvage oncological surgery is accompanied by relatively normal function. This dimension is important in the patients’ perception of treatment.
LIMB AND IMPLANT SURVIVAL AFTER RECONSTRUCTION WITH MEGAPROSTHESES
Limb survival and implant survival are terms used alternatively in various reports and defined according to varying outcomes, like amputation, aseptic loosening, any reoperation or operation with resection of the prosthesis, last clinical follow-up. The temporal aspect of survival is not always included and often specification according to anatomical location isn’t available. Of the reviewed articles Shehadeh, Gosheger and Mittermayer report on the largest series and include specifically limb and/or implant survival [9, 14, 15]. Shehadeh reports a 92% limb survival at 5 years and 90% at 10 years with amputation or date of censuring as endpoint [9]. Gosheger reports 5-year limb survival, with any outcome as endpoint, of 89% for the upper extremity and 88% for the lower extremity [14]. Jeys and Grimer report a 91% survival rate at 20 years from surgery [32].
Implant survival is reported at 84% at 5 years,72% at 10 years and 37% at 20 years by Shehadeh [9]. Ercolano differentiates as to the definition: without any re-operation a 60% implant survival at 5 years and 42% at 10 years is reported while if minor operations are excluded the 5 year survival reported raises to 71% [33]. Mittermayer reports an overall implant survival rate of 79% at 5 years and 71% at 10 years [15]. It has to be noted that the above data arise from literature referring to use of endoprostheses for mostly primary tumors and that follow-up periods range from around 1 year to over 11 years.
On the whole though, it seems that at least in the oncological setting, megaprostheses offer a functioning limb in a satisfactory number of patients and for a satisfactory period of time. Notably, implants used in the upper extremity, and more precisely in the shoulder region, have a considerably better survival than lower-extremity implants, particularly the ones used for reconstruction around the knee joint. Indeed, Shehadel et al. reported a 100% 10-year survival of scapular implants and a 78% 10-year survival for proximal humerus implants [9]. This probably reflects the lower mechanical loads and superior soft tissue coverage in the shoulder region. The challenge for the future lies in establishing the factors that will allow the endoprostheses to endure the mechanical and biological strain of long surviving patient groups. Thus, systematic assessment of the factors leading to complications and failure is necessary.
MAJOR COMPLICATIONS AFTER RECONSTRUCTION WITH MEGAPROSTHESES AND RECENT DEVELOPMENTS THAT DIMINISH FAILURE RATES
Wirganowicz et al. introduced a seemingly rational classification of endoprosthesis failures into mechanical and non-mechanical [34]. Palumbo et al. published a review in 2010 where this classification was used to discuss actual literature findings [23]. It appears reasonable to continue in the same track, so that the data that accumulates in a prospective manner can be readily comparable between the reporting institutions.
Mechanical complications are inherent to the implant design. Typical mechanical complications that are reported in the literature are aseptic loosening, fatigue fractures of the stems, bushing failure as well as failure of the attachment of the soft tissues to the prosthesis. Mechanical complication rate between 5 and 48% are reported in various studies, and aseptic loosening was the leading cause in early reports [9,11,34-36]. According to the data, the rate of mechanical complications has diminished considerably over time, reflecting the introduction of modern, robust modular designs. Furthermore, biocompatible materials have been introduced, which have improved attachment of the soft-tissue envelope to the prosthesis, in the hip, knee and shoulder joint. Soft-tissue failure in the area of the shoulder and the knee often results in a poor range of motion, whereas in the area of the hip it is accompanied by an increased risk for dislocation. In that localisation, the routine use of constrained cups can further reduce the risk for dislocation, especially in the case of periacetabular tumors [37]. Other techniques that can be useful include the reconstruction of the capsule using local muscle transfer or composite materials (Dacron tape, Trevira tube) [28]. Another improvement was the introduction of parts that can be osteointegrated, mainly hydroxyapatite coated stems, thus dispersing load in the skeleton and reducing repetitive mechanical stress in the bone-prosthesis or cement-bone interface. Modern cementation techniques have been used quite for a long time, and reduced the incidence of loosening. In the case of cementless implants, modern press-fit designs rely on the improved geometry (i.e. hexagonal shape) and texture (porous coating) in order to attain immediate stable fixation and less risk of loosening in the long term. Moreover, rotating-hinge knee prostheses have shown remarkably lower risk for mechanical complications in comparison to previous, fixed-hinge implants, as a result of the reduction of the torsional loads that are transferred to the stem [7].
Major non-mechanical complications are infection, wound necrosis and tumor relapse. Failure due to infection is reported in the 2-12% rate in the literature. Infection and wound healing problems are common in oncologic orthopaedic surgery, because of the presence of large implants, often with inadequate soft tissue coverage after the extensive surgical excision, in an environment of immunosuppression and malnutrition induced by chemotherapy and local irradiation. This renders even modern antibiotic therapies quite ineffective, and poses a significant risk for amputation. As far as tumour recurrence is concerned, it is almost inadvertently associated with amputation of the limb. The ordinary use of muscle flaps, especially in the case of megaprostheses around the knee joint, where there is poor soft-tissue coverage, has diminished the risk for wound complications and infections. These can be further reduced with appropriate use of antibiotics as well as meticulous surgical technique in order to avoid hematoma formation and high-tension adaptation of the wound [22, 27]. Recent developments include the introduction of silver-coated or iodine-impregnated implants [32, 38, 39]. Although the data thus far are sparse, there is good reason to believe that this technology can contribute to the significant reduction of post-operative infections. However, it is obvious that non-mechanical complications continue to constitute the major threat for the satisfactory outcome of surgery, and are likely to do so also in the near future. Advances in the field of infection prevention and oncologic therapy of the patients are expected to offer solutions to the surgeon who faces the challenge of dealing with a non-mechanical complication.
GENERAL CONCLUSION
Modern modular megaprostheses have allowed for limb preserving surgery being the gold standard in oncologic orthopaedic surgery, as they facilitate efficient reconstruction of large skeletal defects, combined with high limb salvage rate. They also offer valuable solutions in the case of major trauma or end-stage revision of standard endoprostheses. They do have a relatively high complication rate, approximately 10-times the one observed in regular arthroplasty. This is mainly due to non-mechanical events, as improvements in the design of the implants have apparently reduced the rate of mechanical complications. In addition, the functional outcome after surgery appears to be very satisfactory, offering a good quality of life to the patient. Research should focus mainly on the elimination of non-mechanical events, such as infection, wound dehiscence and tumor relapse, whereas there are also some mechanical drawbacks present even in modern designs, such as the attachment of soft tissues to the prosthesis. However, additional improvements and advances in the field are awaited in order to further improve the results of surgery.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
Declared none.


