All published articles of this journal are available on ScienceDirect.
Disc Height and Sagittal Alignment in Operated and Non-Operated Levels in the Lumbar Spine at Long-Term Follow-Up: A Case-Control Study
Abstract
Purpose:
To compare lumbar disc degeneration at 9-year follow-up in patients with chronic low back pain who had instrumented lumbar fusion or no fusion.
Material and methodology:
The main outcomes were disc height and sagittal alignment measured by Distortion Compensated Roentgen Analysis (DCRA). Secondary outcome included the Oswestry Disability Index and VAS back pain.
Results:
Forty-eight patients with baseline and 9-year radiographs from L2- S1 were included. Twenty-three had lumbar fusion and 25 had no fusion. Disc height was reduced at all levels independent of fusion. No difference in disc height and sagittal alignment was observed between patients fused and not fused. There were weak correlations, ranging from 0.04 to 0.36, between clinical and radiological parameters.
Conclusion:
Lumbar disc degeneration increased at all measured levels independent of fusion and correlated poorly with clinical outcome.
INTRODUCTION
Degeneration of the lumbar spine is a common and age-related finding in the normal population [1]. Both genetic and behavioral factors contribute to disc degeneration [2]. Intervertebral disc degeneration is reported to start in the second decade of life and progress slowly in some patients and more rapidly in others. The reason for the different progression is not clear, but twin studies suggest that up to 70% of the variation is hereditable [3].
The impact of disc degeneration on chronic low back pain (CLBP) is debated [4]. Livshits et al. [5] reported that individuals with severe lumbar disc degeneration had more back pain compared to those with minor degeneration, while Carragee et al. [6] reported weak associations between structural changes on magnetic resonance imaging (MRI) and back pain and disability.
The number of lumbar fusion operations performed for CLBP and disc degeneration has increased rapidly in the past two decades, but the effectiveness of lumbar fusion surgery has been questioned after the publication of three randomised trials [7-9], two of these concluding that lumbar fusion is no more effective than cognitive behavioral intervention.
Patients fused for disc degeneration and CLBP may be more prone to further degeneration than the rest of the population. The level cranial or caudal to the fusion level is considered to be more susceptible to subsequent degeneration because the adjacent level is suggested to be exposed to higher mechanical stress compared to the other levels. It is believed that this may accelerate the degenerative process and result in new symptoms from the unfused adjacent segment. However, conflicting results exist and several studies have demonstrated degenerative changes at multiple levels above a previous fusion, not limited to the first adjacent segment [10, 11]. Thus, we expect that disc degeneration will worsen over time in all lumbar segments independent of fusion, although fusion and genetic factors may accelerate this process [12-14].
The clinical implication of observed degeneration of the adjacent segment to fusion is uncertain. Several authors have reported poor correlation between radiological adjacent segment degeneration and clinical symptoms [15-17]. Valid methods for assessment of symptoms from the adjacent segment are lacking and decisions for eventual elongation of the fusion are made from clinical judgment. Thus, empirical results about the association between adjacent segment degeneration and clinical symptoms in the long-term are of interest.
Animal studies have demonstrated increased degeneration of articular cartilage and connective tissue after immobilization [18, 19]. Lack of mobilisation of the intervertebral discs may alter intracellular properties and predispose the disc to degeneration [19]. The main technical goal of lumbar fusion is to achieve rigidity and intersegmental fusion. It may be hypothesized that disc degeneration will continue within the fused segments, but to our knowledge this hypothesis has not been examined.
The main objective of the current study was to compare disc height and intervertebral sagittal alignment in operated and non-operated levels in the lumbar spine at long-term follow-up after instrumented lumbar fusion and compare these results with those of non-operated patients. The secondary aim was to examine the association between clinical outcome (pain and disability) and disc degeneration at long-term follow-up.
MATERIAL AND METHODOLOGY
Patients
A flow-chart of the included patients is demonstrated in Fig. (1). Forty-eight patients previously included in a randomized study to compare lumbar fusion with cognitive behavioural treatment [7, 8, 20] were available for participation in the present case-control study. Of these, 23 had undergone lumbar fusion and 25 had no fusion. There were no significant differences in baseline characteristics between the patients included in the present study and all patients included in the original clinical trial (Table 1).
Baseline Characteristics of All Patients Randomised in the Clinical and Patients as Treated in the Present Study. Means ± SD or Number of Patients (%) are Given
| Characteristics | Lumbar Fusion | Cognitive Intervention and Exercises | ||
|---|---|---|---|---|
|
|
||||
| All Randomised (n = 66) | Had Fusion in the Present Study (n = 23) | All Randomised (n = 58) | Had No Fusion in the Present Study (n = 25) | |
|
|
||||
| Age (years) | 42.7 ± 8.0 | 44.0 ± 9.0 | 42.4 ± 8.0 | 41.7 ± 8.0 |
|
|
||||
| Men | 27 (41) | 11 (48) | 29 (50) | 14 (56) |
|
|
||||
| Years from first pain episode | 8.9 ± 7.9 | 9.6 ± 8.1 | 9.6 ± 7.4 | 8.3 ± 5.3 |
|
|
||||
| Married/living together | 57 (86) | 20 (87) | 49 (81) | 21 (84) |
|
|
||||
| Occupational education < 3 years | 45 (68) | 16 (70) | 38 (66) | 14 (56) |
|
|
||||
| Work status (%) | ||||
| - working | 9 (14) | 2 (9) | 9 (15) | 5 (20) |
| - on sick leave | 14 (21) | 6 (26) | 6 (26) | 5 (20) |
| - on rehabilitation | 29 (44) | 8 (35) | 22 (38) | 10 (40) |
| - disability pension | 10 (15) | 5 (22) | 10 (17) | 5 (20) |
| - others 1 | 4 (6) | 2 (8) | 1 (2) | 0 |
|
|
||||
| Back pain (0-100 = worst possible) | 63 ± 15 | 65 ± 15 | 65 ±13 | 60 ± 13 |
|
|
||||
| Comorbidity | 24 (36) | 8 (35) | 18 (31) | 7 (28) |
|
|
||||
| Taking analgesics daily or weekly | 40 (61) | 18 (78) | 40 (69) | 13 (52) |
|
|
||||
| Smoking | 36 (55) | 11 (48) | 30 (52) | 12 (48) |
|
|
||||
| Previous surgery for disc herniation | 29 (44) | 10 (43) | 31 (53) | 13 (52) |
1 Others are students, homemakers, unemployed and patients on retirement pensions.

Flow chart of patients included in the present study.
Eight of 23 fused patients had re-operation because of persistent pain. Implants were removed in five of the eight patients and three had an extended fusion. Ten of 23 patients had a single level fusion at L5/S1, and 13 patients had a two-level fusion from L4 to S1. At baseline, 12 of 25 non-operated patients were considered candidates for having a L5/S1 fusion, and 13 patients considered candidates for having a L4 to S1 fusion.
The Ethics Committee for Medical Research in Health Region South-East of Norway approved the study.
Treatments
The lumbar fusion consisted of posterolateral autologous bone transplantation and transpedicular screw fixation of the L4/L5 and/or L5/S1 segments and postoperative physiotherapy prescribed by the surgeon. The cognitive intervention and exercises programme was conducted over a 3-week period [7, 8]. The goal was to make the patients confident in participating in daily activities previously labelled as not recommended.
Radiographic Outcome Measures
Lateral radiographs were obtained both at inclusion (i.e., before treatment) and at long-term follow-up. Radiographs were obtained from levels L1 to S1, but L1/L2 level images were excluded because of poor quality. Consequently, we included the two fused levels (L4/L5 and L5/S1) and two non-fused levels (L2/L3 and L3/L4) in the analyses.
All evaluations of disc height and sagittal alignment were performed by an independent reviewer who measured disc height and sagittal alignment (e.g., the position of the cranial vertebra in relation to the caudal vertebra) by Distortion Compensated Roentgen Analysis (DCRA) [21]. This computer-assisted method, based on measurements from plain lateral radiographic views, has been validated previously by comparison with Roentgen Stereophotogrammetric Analysis (RSA) [22]. Fig. (2) illustrates the DCRA method and summarizes the definitions of the parameters used. The DCRA protocol [21] compensates for distortion caused by axial rotation, lateral tilt, and off-centre positioning of the spine. This permits processing of radiographs taken in normal clinical settings; knowledge of the exposure geometry is not required. All motion segments imaged on a lateral radiograph can be evaluated.
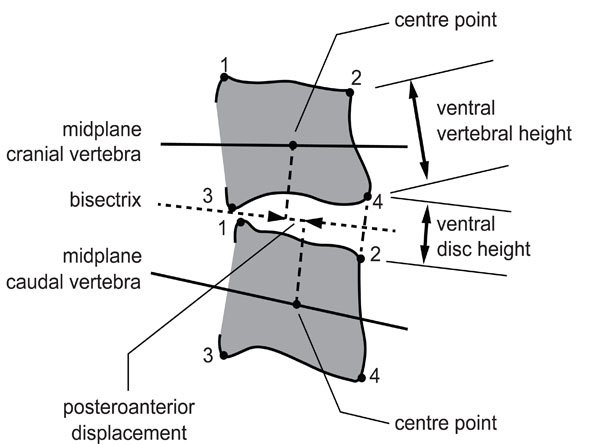
Definition of parameters defined by DCRA.
| DCRA | Definition |
|---|---|
| Mean vertebral depth | Mean of distances of corners 1 and 2 and corners 3 and 4 |
| Sagittal plane angel | Angle between vertebral midplanes, which is defined as the line running through midpoints between corners 1 and 3 and 2 and 4, respectively. |
| Disc height | Sum of the distances from the corners 2 and 4 from the bisectrix between midplanes, divided by the mean depth of the cranial vertebra. As disc height depends linearly on sagittal plane angle, a correction is applied in order to permit comparisons among radiographs taken in different postures of the lumbar spine. |
| Sagittal translation or postero-anterior (dorso-ventral) displacement | Distance between the projections of the centre points (geometric center of corners 1-4) of the vertebrae onto the bisectrix, divided by the mean depth of the cranial vertebra. Displacement is counted positive if the cranial vertebra is displaced in anterior direction with respect to the caudal vertebra. Similar to disc height a correction is applied in order to permit comparisons among radiographs taken in different postures of the lumbar spine. |
Printed with permission from G. Leivseth
Sagittal alignment and disc height are given as the standard deviation from the gender-, age-, and level-appropriate normal values [21]. By example, a value of minus 1.0 denotes that the respective parameter is one standard deviation below normal.
In the present study heights of the vertebrae, measured from the radiographs, taken pre- treatment and at follow-up, agreed on average within an error of 2% of the vertebral depth. Assuming that mean vertebral depth is 35 mm, this corresponds to a measurement error in disc height in the order of 0.7 mm. A previous methodological study reported that the error in sagittal plane displacement amounts to 0.015 (measured in units of mean vertebral depth); e.g., for a vertebral depth of 35 mm, this corresponds to an error of 0.5 mm [21].
Clinical Outcome Measures
The Norwegian version 1.0 of the Oswestry disability index (ODI) [23] was used to evaluate condition-specific disability and pain. This score has 10 questions about pain and disability and ranges from 0% (no pain and disability) to 100% (worst possible pain and disability).
Back pain was rated on vertical visual analogue scales from 0 to 100 (worst pain imaginable). Maximum pain and minimum pain during the last week and current pain were scored on three different scales. The mean of these scores provides the pain index for back pain [24].
Statistical Methods
Data were checked for normal distribution. Results in cases (fused patients) and controls (non-operated patients) were compared using differences in standard deviation from the gender-, age- and level-appropriate normal values. In addition, paired samples t-tests were used to compare changes in disc height and alignment from baseline to long-term follow-up within groups. Analysis of covariance (ANCOVA) was used to compare differences in change between cases and controls at long-term follow-up with adjustments for previous surgery, disc herniation, and baseline measures in disc height and alignment. We did not adjust for re-operation.
The Spearman R correlation coefficient was used to examine the association between disc height, sagittal alignment, ODI, and back pain at the 9-year follow-up. Analyses were performed with the Statistical Package for the studies of Social Sciences, version 18 (SPSS, Inc., Chicago, IL).
RESULTS
At the 9-year follow-up, disc height was significantly reduced from baseline at levels L3/L4 and L4/L5, but not at L2/L3 and L5/S1 in both cases and controls (p<0.05) (Table 2).
Values are Disc Heights Given as the Difference in Standard Deviations from an Age and Gender Adjusted Normal Population. Mean ±SD at Baseline and 9-Years Follow-Up in Fused and Non-Fused Patients and the Mean Difference (95% CI) Between Groups are Given
| Level | Lumbar Fusion (n = 23)1 | No Lumbar Fusion (n=25)1 | Adjusted2 Mean Difference Between Groups | ||
|---|---|---|---|---|---|
| Baseline | 9-Years | Baseline | 9-Years | ||
| L2-L3 (non-fused level) | -0.65±1.23 | -1.13±1.39 | -0.29±0.88 | -0.65±1.29 | 0.14 (-0.51 to 0.79) |
| L3-L4 (non-fused level) | -0.54±1.33 | -1.32±1.25* | 0.18±1.02 | -0.45±1.17** | 0.36 (-0.27 to 0.98) |
| L4-L5 (fused level) | -0.88±1.33 | -1.61±1.70** | -0.81±1.30 | -1.35±1.42** | 0.03 (-0.57 to 0.63) |
| L5-S1 (fused level) | -1.19±1.62 | -1.55±1.47 | -1.08±1.02 | -1.38±1.08 | 0.20 (-0.38 to 0.78) |
1 At level L5-S1: lumbar fusion n = 19 and no lumbar fusion n= 21.
2 Adjusted for previous discectomy and baseline values.
* p<0.05
** p<0.01.
Sagittal alignment was significantly increased at level L2/L3 in cases and controls (p<0.001), and in addition at level L5/S1 in controls (p<0.05). We observed no difference in the other investigated levels (Table 3). There was no difference between cases and controls at any level (Table 3).
Values are Sagittal Alignment Given as the Difference in Standard Deviation from an Age and Gender Adjusted Normal Population. Mean ±SD at Baseline and 9-Years Follow-Up in Fused and Non-Fused Patients and the Mean Difference (95% CI) Between Groups are Given
| Level | Lumbar Fusion (n= 23)1 | No Lumbar Fusion (n=25)1 | Adjusted2 Mean Difference Between Groups | ||
|---|---|---|---|---|---|
| Baseline | 9-Years | Baseline | 9-Years | ||
| L2-L3 (non fused level) | -0.07±1.01 | -0.69±1.07** | -0.21±0.87 | -0.70±0.94** | 0.24 (-0.30 to 0.78) |
| L3-L4 (non fused level) | -0.34±1.19 | -0.34±1.71 | -0.41±1.37 | -0.55±1.44 | 0.04 (-0.80 to 0.73) |
| L4-L5 (fused level) | -0.32±1.21 | -0.15±1.06 | -1.06±1.31 | -0.27±1.22** | 0.13 (-0.49 to 0.75) |
| L5-S1 (fused level) | 0.26±1.57 | 0.24±1.21 | 0.09±2.66 | 0.51±2.77* | 0.32 (-0.33 to 0.99) |
1 At level L5-S1: lumbar fusion n = 19 and no lumbar fusion n= 21.
2 Adjusted for previous discectomy and baseline values.
* p< 0.05
** p<0.01.
There were weak correlations between clinical and radiological parameters ranging from r = 0.04 (p = 0.79) to r = 0.36 (p = 0.01). This means that from zero to 13 % of the variation in clinical parameters were explained by variation in radiological parameters.
There were weak correlations between lumbar levels for disc height and sagittal alignment (Table 4).
Correlations Between Lumbar Levels for Disc Height and Sagittal Alignment
| Correlation Between Levels for Disc Height | Spearman’s Rho (r) | |
|---|---|---|
|
|
||
| L2/L3 vs L3/L4 | 0.61 | p<0.001 |
| L3/L4 vs L4/L5 | 0.41 | p= 0.004 |
| L4/L5 vs L5/S1 | 0.34 | p=0.02 |
|
|
||
| Correlation Between Levels for Sagittal Alignment | ||
|
|
||
| L2/L3 vs L3/L4 | 0.16 | p=0.28 |
| L3/L4 vs L4/L5 | 0.19 | p=0.20 |
| L4/L5 vs L5/S1 | 0.15 | p=0.33 |
DISCUSSION
We found increased degeneration of the lumbar discs in both cases and controls at long-term follow-up. There were no difference between cases and controls and no association with changes in pain and disability.
Disc degeneration is a normal feature of ageing [25], and it is unclear how fusion influence on this. In the present study, reduction of lumbar disc height was independent of whether the level was fused or not. Reductions in disc height at the fused levels L4/L5 and L5/S1 were similar to reductions in disc height at non-fused levels, and to the identical levels in non-operated patients. Results are in agreement with previous studies, but these earlier studies did not include a control group of non-operated patients [10, 11].
In agreement with previous studies, we found weak associations between changes in pain and disability and changes in disc height [26]. The observed correlations in the present study ranged from 0.04 to 0.36, which means that only 0- 13% of the observed variation in disability and pain could be explained by radiological changes. Berg et al. [26] reported that despite fulfillment of surgical goals for either the lumbar fusion or disc prosthesis group, there was poor correlation between disc degeneration and clinical outcome at 2-years follow-up.
Spine surgeons often use radiological signs of disc degeneration in decision-making, with respect to eventual re-operation at adjacent levels. However, the observed weak associations reported between radiological and clinical outcomes do not support this strategy.
Sagittal alignment describes the position of a vertebra in relation to an adjacent vertebra. We found small changes within groups and no differences between groups at long-term follow-up. Unfavourable sagittal alignment has been proposed as a risk factor for development of degenerative changes at the adjacent level, but this remains controversial [10]. In agreement with Pellisé et al. [11] we found increased sagittal alignment in the second adjacent level cephalad to the fusion. This tendency was observed both in fusion cases and in non-fusion patients, and we believe that this observation raises questions regarding the influence of fusion on sagittal alignment.
There were weak correlations between disc degeneration in neighboring segments in both patient groups [11, 14] and weak correlations between sagittal alignment in neighboring levels. This finding suggests that factors other than fusion (for example, genetic factors and ageing) are important factors contributing to disc degeneration [2, 25].
The extent and severity of reported disc degeneration vary widely from study to study and are influenced by the technology used to examine the disc [2]. Both magnetic resonance imaging (MRI) and radiographs correlate with the morphological degree of degeneration [27]. MRI is considered to be the gold standard modality for imaging spinal structures in vivo, and signs of degeneration (i.e., altered T2 signal intensity, annular tears, and Modic changes) are seen at an earlier stage on MRI compared to plain radiography [28]. However, interpretation of MRI images is difficult because of artifacts in patients who have steel implants. The fused patients in the present study had steel implants; therefore we used plain radiography and applied the validated DCRA method to obtain indicators of disc degeneration such as disc height and sagittal alignment [22].
The strengths of the present study are the application of the DCRA method, the inclusion of both fused and non-fused patients, the long observation time and the use of age- and gender-adjusted values from a large database of approximately 3,000 healthy individuals to calculate results in SD from population values.
An important limitation of our study is the lack of randomization and the small number of patients. The DCRA method is resource intensive (and precise) and corresponding patient samples are reported in previous studies using this method [11, 29]. The estimates of mean differences and their corresponding confidence intervals reflect both the inter-individual differences and the sample size. Differences between cases and controls were much smaller than the observed variation between patients. By example, with the standard deviation of 1.3 and the observed group difference of 0.34 at the L3-4 segment, 205 patients are required in each group to detect 0.05 % significance with 80 % power. The lack of correlation between clinical and radiological parameters suggests that the observed difference has no clinical significance.
We conclude that disc degeneration at long-term is not associated with lumbar fusion or other clinical parameters. This finding opposes the commonly held belief that lumbar spine fusion leads to increased adjacent level degeneration that may require re-operation. Our findings need to be validated in future, larger studies.
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
AKNOWLEDGEMENTS
We thank the Research Council of Norway and Vestre Viken Hospital Trust for providing the financial support for this study.


