All published articles of this journal are available on ScienceDirect.
Effect of a Direct Thrombin Inhibitor Compared with Dalteparin and Unfractionated Heparin on Human Osteoblasts
Abstract
Purpose:
Osteoporosis is a relevant problem after long term administration of unfractionated heparin (UFH) and low molecular weight heparin. Melagatran is a representative of a new group of direct thrombin inhibitors with comparable data in the prevention of thromboembolic events after orthopaedic surgery. The aim of our in vitro study was to investigate the effect of a direct thrombin inhibitor compared with dalteparin and UFH on human osteoblasts.
Materials and Methods:
Melagatran, dalteparin and UFH were added to primary osteoblast cultures in their therapeutic range and two decimal powers below and above. Cell number, protein synthesis, mitochondrial and alkaline phosphatase activity and collagen type I synthesis were evaluated.
Results:
Melagatran showed the least influence on protein synthesis and cell proliferation with a reduction of cell number to 83.5 ± 9% (p = 0.027) of the control group only in the highest investigated concentration after 15 days of incubation.
Mitochondrial and alkaline phosphatase activity and collagen type I synthesis in osteoblasts incubated with melagatran and dalteparin showed similar patterns. UFH showed the most pronounced influence on cellular metabolism.
Conclusions:
Melagatran showed less inhibitory in vitro effects on human osteoblasts than dalteparin or UFH. The presented study gives first hints that direct thrombin inhibitors may help prevent heparin-induced negative effects on bone metabolism.
INTRODUCTION
Anticoagulant therapy is capable of drastically reducing the incidence of thromboembolic events after orthopaedic and traumatologic interventions [1-3]. A relevant side effect of unfractionated heparin (UFH) and low molecular weight heparins is the induction of osteopenia during long-term therapy [4-6], which can progress to osteoporosis with the risk of pathological fractures [7, 8]. Shaugnessy et al. showed a 30% decrease of cancellous bone volume in a rat model after only 28 days of UFH administration [9]. Tinzaparin, a representative of the low molecular weight heparins (LMWH), reduced the bone mineral density by 18.5% after an application of 32 days [10].
The direct thrombin inhibitor ximelagatran is a member of a new group of anticoagulants, which can be administered orally in contrast to the heparins. After intake ximelagatran is converted to the active agent melagatran through dealkylation and dehydroxylation.
Clinical studies proved the effectiveness of melagatran during long-term prophylaxis of thromboembolic events [11, 12].
Side effects of melagatran on bone metabolism have not been investigated yet. One of the most sensible markers for heparin-induced osteopenia is a loss in osteoblast number [13-15]. Therefore the aim of this study was to describe the effect of melagatran on human osteoblasts in vitro in comparison to conventional heparins, namely UFH and the LMWH dalteparin. Apart from the influence of the drugs on cell proliferation we also investigated parameters of osteoblast metabolism as mitochondrial activity, collagen production, alkaline phosphatase activity and protein synthesis.
MATERIALS AND METHODS
Osteoblast cultures were derived from cancellous bone from six individuals (aged 63 – 78 years, males 3, females 3) harvested during total knee replacement according to a previously published procedure [15]. Bone metabolism disorders were excluded preoperatively by laboratory tests and bone density measurement (Lunar-DPXL, General Electric, Germany).
Cell Culture
The cancellous bone was separated into fragments of approximately 1 mm3 size, which were rinsed with phosphate buffered saline (PBS) until no fat droplets and erythrocytes were macroscopically visible in the supernatant anymore. Eight cancellous cubes were seeded on a Petri dish (BD Bioscience, Germany) and incubated with 8 ml of medium (DMEM Ham’s F12 with 10% of fetal calf serum + penicillin + streptomycin, Gibco, Germany) which was renewed at 3 day intervals. Upon confluence of the cells, they were transferred to cell culture flasks (BD Bioscience, Germany) using 0.25% trypsin (Sigma, Germany). After two weeks the cells could be used for the investigations and were seeded onto 48-well plates (BD Bioscience, Germany) with a density of 2500 cells/well in 0.5 ml medium.
After adherence of the cells, the culture medium was renewed and melagatran, dalteparin, UFH, or PBS was added. Dosages of the drugs were referenced to the therapeutic plasma concentrations with additional dosages at two decimal powers above and beneath the latter (Fig. 1).

Media and substances were changed in two day intervals. Measurements were performed on the 15th day after addition of the substances (n = 12 wells per concentration and timepoint).
Cell Proliferation
Cell number was determined every second day (n = 6 wells per concentration and timepoint).
Cells were rinsed twice with PBS and incubated with 0.25% trypsin for 5 min. Total detachment was controlled microscopically before measuring cell concentration using a cell counter (CASY II, Schärfe Systems, Germany).
Cell Vitality Test
Activity of mitochondrial dehydrogenase was determined to evaluate cell vitality. For this purpose a test kit (Cell Proliferation Kit II, Roche Molecular Biochemicals, Switzerland) based on the metabolization of XTT (sodium 3´-[1-[(phenylamino)carbonyl]-3,4-tetrazolium]-bis(4-methoxy-6-nitro)benzolsulfonic acid hydrate) was used [16, 17]. Two hours after the addition of 120 µl reagent and 240 µl medium per well, 300 µl from each well were transferred to a 96-well plate and read off spectrophotometrically at 450 nm against a reference of 630 nm using an ELISA reader (Dynex Revelation 4.22, Microtek, Taiwan).
Measurement of Protein Concentration
Due to the high amount of protein in FCS, cells were rinsed for three times and incubated with FCS-free medium plus substance 24 hours before measurement. After this period 150 µl of supernatant of each well were transferred to a 96-well plate and mixed 1: 1 with reagent (BCA Protein Assay, Pierce, USA). After 30 minutes the protein value was determined against a standard curve of bovine standard albumin (0-512 µg/ml BSA) at 630 nm.
Measurement of Alkaline Phosphatase (ALP) Activity
After evaluation of the protein concentration the remaining medium was aspirated and the cells were rinsed with PBS. 500 µl of reagent (37.5 parts by volume 0.5 M NaHCO3, 12.5 parts by volume 0.01 M MgCl2, 25 parts by volume distilled water and 25 parts by volume 0.1 M PNPP, p-nitrophenyl phosphate, Sigma, USA) were added. After an incubation of 8 h at 37 °C, the activity of ALP was determined photometrically at 405 nm against a standard curve.
Measurement of Matrix Collagen Type I
The plates were rinsed three times with PBS and incubated overnight at 4 °C with anti-collagen type I rabbit polyclonal antibodies diluted 1: 150 (Chemicon, USA), and treated by the EnVisionTM method (DAKO, Denmark). After repeated washes with PBS, the wells were reacted with diaminobenzidine solution containing 0.01% H2O2.
The plates were scanned using the resolutions 600 x 600 dots/inch and 8 bits/colour (red, green, blue). After separation of the magenta-coloured channel, representing the immunostaining [18], the mean greyscale value of each well was determined by defining the region of interest and calculating the histogram. All image operations were performed on an Intel® Pentium® M 715 processor using Corel Draw 10®.
Preliminary Tests
In order to determine the influence of the substances on the measuring procedure, all of the above-mentioned concentration and activity determinations were first conducted without cells.
Statistical Analysis
All measurements were repeated three times under the same conditions, whereby two parallel sets were conducted at each time. Mean and standard deviations were determined for each group. Significance analysis was performed with the non-parametric Mann-Whitney U test for independent samples.
RESULTS
The preliminary tests showed that the substances added to the medium did not influence the results in the investigated concentration ranges.
Proliferation Assay
UFH and dalteparin caused an increase of cellular proliferation up to the 2.3-fold, respectively 1.5-fold of the control group without substance at concentrations between 0.05 and 5 U/ml (p < 0.001) (Fig. 2). 50 U/ml UFH inhibited cellular proliferation to 10 ± 3%, 50 U/ml dalteparin to 39 ± 15% of the control group (p < 0.001 both)
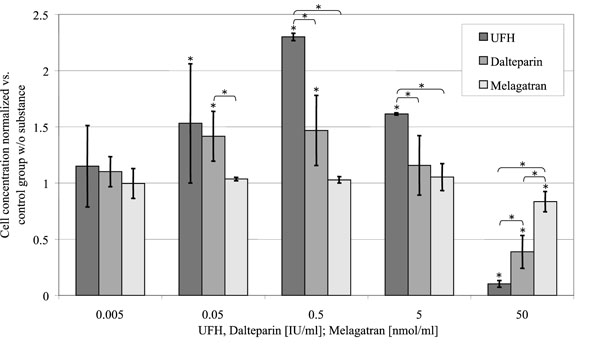
Effect of melagatran, dalteparin and UFH on cell proliferation. Relative cell content per well on day 15 after incubation, standardized to the control group without substance in relation to the concentration of the substances investigated. (control = 1).
In contrast to that melagatran only had an influence on osteoblast proliferation in the highest investigated concentration with a reduction of cell number to 84 ± 9% of the control group (p = 0.027).
UFH and dalteparin inhibited osteoblast proliferation from the fourth day on, melagatran from the sixth day on after application (Fig. 3).
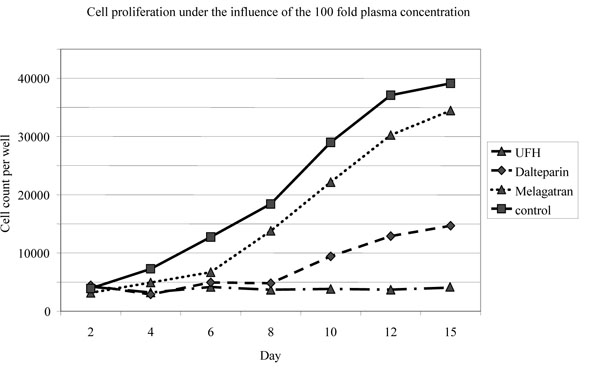
Cell proliferation (absolute cell numbers per well) under the influence of the highest investigated concentration (UFH and dalteparin; 50 IU/ml, melagatran: 50 nmol/ml).
Mitochondrial Activity (Fig. 4)
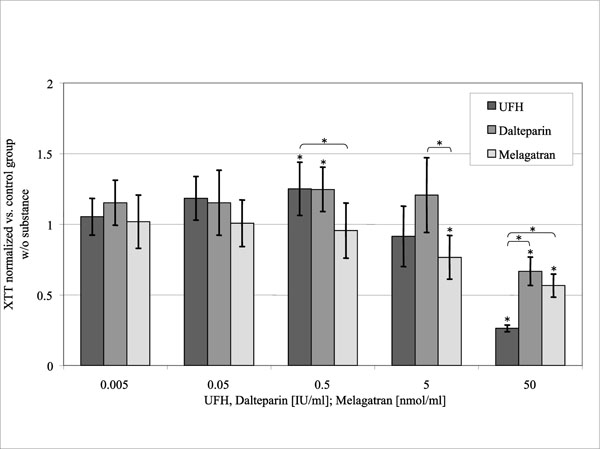
Optical density according to the XTT-Test, standardized to the control group without substance in relation to the concentration of the substances investigated. (control = 1).
UFH and dalteparin caused an increase of mitochondrial activity at 0.5 U/ml to 125% respectively 124% of the control group (p = 0.022 and p = 0.027). In contrast to that higher concentrations of UFH and dalteparin (50 U/ml) reduced cellular activity to 26 ± 2% (p < 0.001) respectively 67 ± 10% of the control group (p = 0.003). Melagatran inhibited cellular activity at 5 and 50 nmol/ml to 77 ±15% (p = 0.04) respectively 57 ± 8% of the control group (p < 0.001).
Protein Synthesis (Fig. 5)
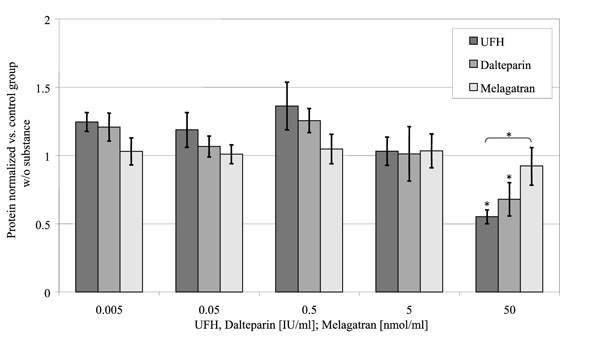
Relative protein concentrations of the medium in relation to the concentration of the substances investigated. (control = 1).
At 50 U/ml UFH and dalteparin reduced osteoblast protein synthesis to 55 ± 5% (p < 0.001) respectively 68 ±12% (p = 0.013) of the control group without substances. In contrast to that melagatran had no influence on the protein synthesis of the cells in the investigated concentrations.
Alkaline Phosphatase Activity (Fig. 6)
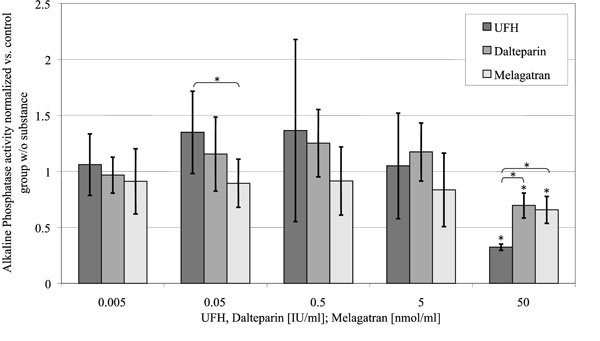
Alkaline phosphatase activity of the osteoblasts, standardized to the control group without substance in relation to the concentration of the substances investigated. (control = 1).
Activity of alkaline phosphatase was reduced to 32 ± 3% of the control group by 50 U/ml UFH (p < 0.001). In comparison to that 50 U/ml dalteparin only caused a reduction to 70 ± 11% of the control group (p = 0.013). 50 nmol/ml melagatran inhibited ALP activity to 66 ± 12% of the control group (p = 0.012).
Collagen Type I Synthesis (Fig. 7)
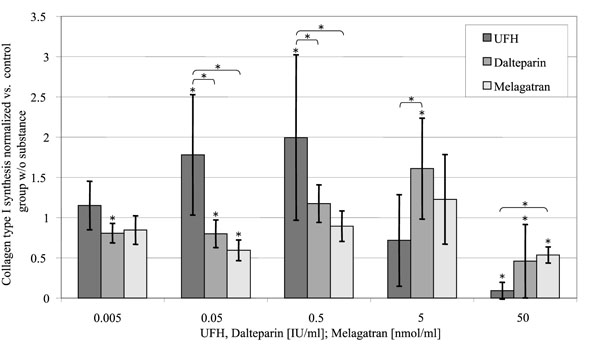
Relative collagen type I deposition per well, standardized to the control group without substance in relation to the concentration of the substances investigated. (control = 1).
UFH increased the collagen type I synthesis in a concentration dependent manner between 0.005 and 0.5 U/ml up to the two-fold value of the control group at 0.5 U/ml. In contrast to that higher concentrations of UFH (5 U/ml and 50 U/ml) lead to a decrease of collagen type I deposition (72 ± 57% (p = 0.056) respectively 9 ± 10% (p < 0.001) of the control group). Melagatran and dalteparin effected a reduction of collagen type I deposition at their highest in vitro concentrations also, but only to 46 ± 45% (p = 0.013) respectively 54 ± 10% (p < 0.001).
DISCUSSION
Osteoblast number and function is crucial for the process of fracture healing, the integration of endoprosthetic implants but also for the permanent remodelling processes of bone.
The decreased bone mineral density after or during heparin therapy results in a higher risk of fractures [7], in an inhibition of fracture healing [19] (Kock, Werther 2002) and may compromise the integration of endoprosthetic implants [20-22] In the present study we investigated for the first time the effects of a member of a new family of oral anticoagulants on human osteoblasts. Changes in osteoblast number and function were compared with changes under the influence of dalteparin und UFH.
Limitations of the study are the investigation of only one member of the group of direct thrombin inhibitors, melagatran, on the one hand and on the other hand the fact that an in vitro study can only mimic in vivo conditions but not entirely describe them. Furthermore the study focussed on osteoblasts and did not investigate osteoclast function or evaluate transcription factors. Therefore the results of our study can only be seen as promising first hints towards a possible advantage of direct thrombin inhibitors concerning osteometabolism.
In the investigated parameters cell number, activity of mitochondrial dehydrogenase, protein synthesis, collagen type I synthesis and activity of alkaline phosphatase melagatran showed similar or less impact on the osteoblasts than dalteparin and UFH in clinically relevant concentrations.
Heparins accumulate in bone and high substance concentrations around osteoblasts can be expected [8, 9]. Hence, the results obtained under the influence of the 10-fold and 100-fold therapeutical concentration of the substances are crucial for their clinical use.
In the highest investigated concentration (50 nmol/ml) melagatran only reduced the cell count to 84% of the control group after 15 days of incubation, whereas reduction of cell count was far more pronounced under the influence of dalteparin (39%, 50 U/ml) and UFH (10%, 50 U/ml). This loss in osteoblasts is to be held responsible for the development of heparin-induced osteopenia [10, 13, 14].
In the present in vitro model negative effects of UFH on cellular proliferation and function could be seen in accordance with the literature. In contrast to that melagatran affected human osteoblasts to a lesser extent, comparable or even less than dalteparin. Based on the presented results advantages concerning the prevention of heparin-induced osteopenia, particularly during long term therapy, could be possible with the use of direct thrombin inhibitors. If this group can be used for long term anticoagulation in future still has to be elucidated. In a study with a 35-day period of intake of ximelagatran, being the first member of oral direct thrombin inhibitors, one patient showed a severe although reversible liver damage and ximelagatran was taken from the market - also since elevated liver enzymes had been reported as an adverse effect beforehand. (EXTEND Study, ClinicalTrials.gov Identifier NCT00206089) Since the effectiveness of melagatran in long term prophylaxis of thromboembolic events had already been shown [11, 12], the future replacement of subcutaneously injected substances by oral anticoagulants can be expected. The oral application is superior to subcutaneous therapy, on the one hand because of better patient acceptance [10] and on the other hand because of fewer side effects of the application itself [23, 24].
The anticoagulatory potential of melagatran could be proven to be equal respectively better than that of LMWHs. The METHRO II study showed a reduction of incidence of deep venous thrombosis to 15.1% in the melagatran group compared to 28.2% in the dalteparin group (p < 0.001) [25]. The comparison with enoxaparin in a study with 2835 patients, who underwent total knee or hip arthroplasty, also revealed a more pronounced reduction of the incidence of deep venous thrombosis in the melagatran group (20.3% versus 26.6%, p < 0.001). Bleeding rates did not differ within groups [26].
Particularly in patients with a reduced bone mineral density, e.g. after steroid therapy or because of renal insufficiency, hyperparathyroidism or idiopathic osteoporosis, prophylaxis of thromboembolism with substances with a smaller osteocatabolic potential than heparin is decisive. The presented study gives firsts hints that direct thrombin inhibitors could represent an alternative in this place.


