All published articles of this journal are available on ScienceDirect.
One Year After Navigated Total Knee Replacement, No Clinically Relevant Difference Found Between Fixed Bearing and Mobile Bearing Knee Replacement in a Double-Blind Randomized Controlled Trial
Abstract
Background:
The mobile bearing designs have not yet been shown to improve clinical outcome of total knee arthroplasty (TKA). In this prospective randomized study, we compared the short-term clinical results of a mobile bearing implant with those of the fixed bearing version of the same implant.
Methods:
We randomized 100 knees into two double-blind groups who received either the fixed (FB, 52 knees) or the mobile bearing (MB, 48 knees) version of the same implant. We used navigation to standardize the surgical technique. For up to one year, we recorded the Knee Society (KSS) and Oxford (OXF) scores. We performed an exploratory analysis of variance (ANOVA) to determine the influence of baseline scores as covariate and the extent of improvement in clinical outcome over time.
Results:
After one year, we did not detect any statistically significant difference between the two groups. The KSS scores differed by 2 points, the OXF scores by 1.1 points.
Conclusion:
Even with identical geometry of implant surfaces and a navigated surgical technique, first-year results do not support a preference for either a fixed or a mobile design.
INTRODUCTION
Initially, knee prostheses with fixed bearings showed good long-term results, but patients repeatedly complained of pain after surgery [1-3]. To address these common complaints, several new designs were introduced, with gliding or rotating tibial inlays [4-7].
The mobile bearing design claims to facilitate “self alignment” and greater overall range of motion by allowing the femur to roll back during flexion and to rotate during extension. In addition, it supposedly reduces PE wear [8]. Yet, these claims have not been confirmed by clinical studies to date [9-11]. However, these studies, mostly non-randomized, compare implants that differ in more aspects than just the tibial tray’s proximal surface and the PE component itself.
In most studies on the subject, computer-assisted implantation was found superior to the conventional regarding component alignment [12-15]. The clinical benefits of precise component alignment are controversial [16, 17], but some evidence suggests that navigation improves the alignment significantly [18]. We used this technology for all patients to standardize the surgical technique.
We consider these short-term results worthwhile publishing for two reasons: (1) If the claimed functional benefit of the mobile platform was real, it would have emerged in the short-term results; (2) until now, no other study succeeded in eliminating the variability in surgical technique to such an extent.
The aim of our randomized double-blind study was to compare mobile (MB) versus fixed bearings (FB), while keeping constant all other parameters, such as implant design, surgical technique, or postoperative rehabilitation. Our hypothesis was that there would be no clinically relevant difference between the groups.
PATIENTS, MATERIALS AND METHODS
Patients scheduled for bicondylar, posterior cruciate-retaining TKA were offered enrolment in the study if they met the inclusion criteria (Table 1). They were operated by one of the two senior surgeons (E.H. and F.L.), using computer-assisted instrumentation (Orthopilot TKA 4.2, BBraun Aesculap, Tuttlingen, DE) in a medial parapatellar approach under general or epidural anesthesia.
Inclusion Criteria for the Study Population as Assessed During Enrollment
|
In the FB version of the implant (Columbus CR [FB group] and RP [MB group], BBraun Aesculap, Tuttlingen, DE), the PE inlay is rigidly fixed to the tibial tray. In the MB version, it rotates around a cylindrical post within a range of ±10° limited by a second post placed anteriorly on the surface of the tray. The femoral components are identical in both versions. Both components were cemented, and no patellar components were implanted.
The patients followed a standard TKA rehabilitation protocol, including self-controlled epidural analgesia with ropivacaine, non-steroidal oral analgesia, antithrombotics, physical therapy and continuous passive motion starting the day after surgery. They spent approximately one week in the hospital and three weeks of inpatient rehabilitation. They were examined by a resident physician in a double-blinded manner before and after operation, using the Oxford and Knee Society scores.
As primary endpoint, we compared the difference in Knee Society scores (KSS) [19] after one year, and as secondary endpoints, the differences in Oxford scores (OXF) [20], ROM, and functional sub-items of the scores, as well as the differences in leg and prosthesis alignment as measured radiologically.
Patients were stratified by age (below or beyond 70) and gender, and double-blinded to the treatment group.
STATISTICAL METHODS
Based on KSS values reported in the literature, the required sample size was calculated to be 50 patients per group, accounting for 20% lost to follow-up [21, 22]. To assess the KSS differences between the treatment groups after one year as primary outcome, we used the Kolmogorov-Smirnov test for normal distribution, the Levene test for homogeneity of variances, and subsequently the Mann-Whitney-U test for significant differences as defined in the study protocol. In addition, we carried out an explorative ANOVA analysis on the KSS subscales (Functional and Knee, KSS-F and KSS-K), OXF, and flexion ROM FLX) and repeated measurements to identify significant effects between follow-up intervals in the FB and MB group.
Registered under ClinicalTrials.gov, no. NCT00822640, the study was approved by the review board of the Hamburg Ärztekammer upon its review of Application #2226. Written informed consent was obtained from each patient prior to enrolment.
RESULTS
Baseline
From April 2004 until June 2007, 100 knees (97 patients) were followed up, 52 knees being randomly assigned to the FB group, and 48 knees to the MB group. The patient flow is shown in Fig. (1). Table 2 provides the demographic details. The distributions of age (p = 01.000) and gender (p = 0.659) between the groups were not statistically different. Preoperative clinical data are presented in Table 3 and Figs. (2, 3) and preoperative radiographic data in Table 4. The average preoperative radiographic deformity was 8°±4° in both groups with reference to the mechanical axis. The postoperative values are given in Table 4.
Demographic Data of the Study Population
| Demographics | FB | MB | ALL |
|---|---|---|---|
| Patients [knees] | 52 | 48 | 100 |
| Gender [female; male] | 39; 13 | 34; 14 | 73; 27 |
| Age groups [<70a; >=70a] | 24; 28 | 22; 26 | 46; 54 |
| Age [a] | 69±8 (53–84) | 70±7 (52–84) | 70±8 (52–84) |
| BMI [kg/m2] | 29±5 (20–45) | 30±6 (21–45) | 30±6 (20–45) |
| Diagnosis [OA; PT; RA; AVN] | 46; 3; 3; 0 | 43; 1; 2; 2 | 89; 4; 5; 2 |
Descriptive summary data is presented in the format “mean ± standard deviation (minimum – maximum)” for the two treatment groups FB and MB and the whole collective (ALL). Diagnosis is encoded as primary (OA), post-traumatic (PT), rheumatoid (RA), and osteoarthritis secondary to avascular necrosis (AVN).
For Each of the Pre- and Postoperative Examinations, the Knee Society Score Functional (KSS-F) and Knee (KSS-K) Scales, the Oxford Knee Score (OXF) and the Range of Motion in Passive Flexion (ROM FLX) are Given
| Descriptives – Clinical | FB | MB | ALL |
|---|---|---|---|
| Preoperative [no. of knees] | 52 | 48 | 100 |
| KSS-F | 52±18 (0–70) | 42±21 (0–70) | 47±20 (0–70) |
| KSS-K | 30±12 (0–56) | 29±10 (15–55) | 30±11 (0–56) |
| OXF | 41±7 (22–55) | 43±5 (31–53) | 42±6 (22–55) |
| ROM FLX [deg] | 111±15 (80–140) | 109±12 (90–130) | 110±14 (80–140) |
| FLX contracture [0–4; 5–15; >15] | 25; 24; 3 | 30; 16; 2 | 55; 40; 5 |
| 3 months follow-up [no. of knees] | 43 | 45 | 88 |
| KSS-F | 70±17 (25–100) | 72±16 (30–100) | 71±17 (25–100) |
| KSS-K | 71±20 (31–99) | 77±15 (46–99) | 74±17 (31–99) |
| OXF | 27±9 (12–49) | 27±7 (13–50) | 27±8 (12–50) |
| ROM FLX [deg] | 105±14 (70–140) | 111±13 (90–150) | 108±14 (70–150) |
| FLX contracture [0–4; 5–15; >15] | 27; 15; 1 | 32; 13; 0 | 59; 28; 1 |
| 6 months follow-up [no. of knees] | 52 | 48 | 100 |
| KSS-F | 83±14 (40–100) | 83±14 (50–100) | 83±14 (40–100) |
| KSS-K | 80±15 (45–99) | 85±13 (44–100) | 82±14 (44–100) |
| OXF | 23±8 (12–46) | 22±8 (12–46) | 22±8 (12–46) |
| ROM FLX [deg] | 111±13 (80–140) | 114±13 (90–150) | 112±13 (80–150) |
| FLX contracture [0–4; 5–15; >15] | 37; 15; 0 | 38; 10; 0 | 75; 25; 0 |
| 12 months follow-up [no. of knees] | 52 | 44 | 96 |
| KSS-F | 88±13 (45–100) | 87±13 (50–100) | 87±13 (45–100) |
| KSS-K | 85±14 (41–100) | 88±12 (40–100) | 86±13 (40–100) |
| OXF | 21±9 (12–52) | 20±8 (12–43) | 21±8 (12–52) |
| ROM FLX [deg] | 113±13 (90–145) | 115±11 (95–145) | 114±12 (90–145) |
| FLX contracture [0–4; 5–15; >15] | 43; 9; 0 | 40; 4; 0 | 83; 13; 0 |
For data format, see the description of Table 2. Additional information presented includes the number of patients examined at each follow-up (no. of knees) and the flexion contracture (FLX contracture) in terms of three categories generated from the respective KSS-K subscale (the first category containing absence of any flexion contracture).
Radiographic Data was Obtained by Measuring the Angle Between the Femur and the Tibia (Mechanical Axis) and Between the Components and the Respective Bone on Anteroposterior (AP) and Mediolateral (ML) Long-Leg, Standing-Position X-ray Images
| Descriptives – Radiographic | FB | MB |
|---|---|---|
| Preoperative axes deviations [no. of knees] | 52 | 48 |
| mechanical axis [deg] | 7.9±3.9 (0–17) | 8.0±3.6 (1–16) |
| femoral a-p. [deg] | 2.4±2.5 (0–11) | 2.6±2.0 (0–7) |
| tibial a-p. [deg] | 3.8±2.3 (0–9) | 3.7±2.3 (0–12) |
| Follow-up axes deviations [no. of knees] | 52 | 48 |
| mechanical axis [deg] | 2.3±1.6 (0–6) | 1.9±1.5 (0–5) |
| femoral a-p. [deg] | 1.2±1.2 (0–4) | 1.5±1.1 (0–5) |
| tibial a-p. [deg] | 1.2±1.1 (0–6) | 1.3±1.0 (0–5) |
| femoral m-l. [deg] | 2.3±1.7 (0–6) | 2.6±2.1 (0–8) |
| tibial m-l. [deg] | 2.4±1.6 (0–7) | 2.3±1.7 (0–6) |
Values indicate the absolute differences from the target alignment (straight or perpendicular, respectively).
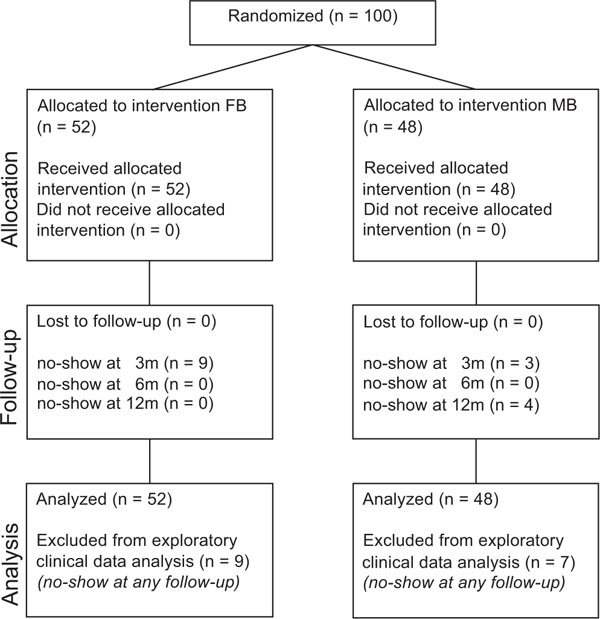
Patient flow according to the CONSORT statement. There is no data available regarding patients who were not randomized.
Complications
Because of infection, the implant in one patient from the MB group needed to be exchanged in two stages nine months after the original operation. One patient from the FB group received a retro-patellar implant because of anterior knee pain unresponsive to conservative treatment. One pulmonary embolism occurred in the FB group. In each group, one patient underwent soft tissue revision for hematoma, and one mobilization treatment under anesthesia.
Follow-Up
Some patients did not attend the three-month and twelve-month follow-up examinations, for reasons unrelated to the TKA procedure, except for the re-operated patient. The number of patients returning at each interval is shown in Table 3.
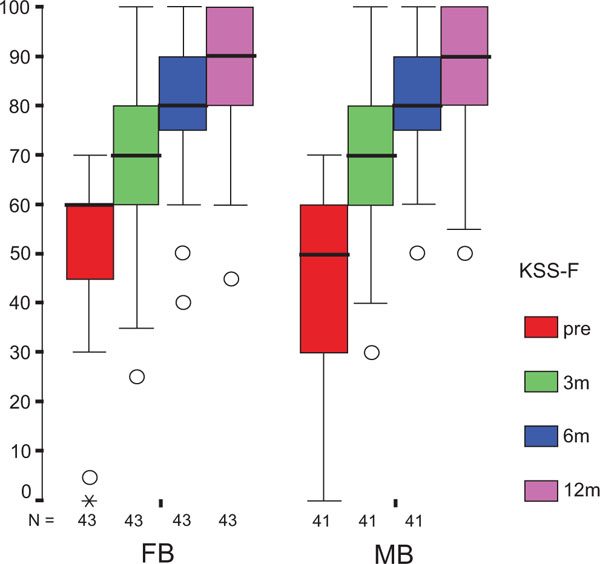
After 12 months, statistical analysis revealed no significant differences between the groups in both components of the KSS score as primary endpoint. Both KSS-F (Fig. 2a) and KSS-K (Fig. 2b) values were not normally distributed, but of homogeneous variance. Furthermore, the difference in results was significantly influenced by the baseline KSS-F (p = 0.003), but not by the treatment group (p = 0.694) with values of 87±2 (83–90) for FB and 88±2 (84–92) for MB. For KSS-K, neither the baseline score (p = 0.090) nor the treatment group (p=0.328) contributed significantly to the difference in results with values of 85±2 (81–89) for FB, and 88±2 (84–91) for MB. Repeated measurements from preoperative evaluation to follow-up reviews revealed significant differences at each interval for both KSS-F and KSS-K (p<0.001). The preoperative and follow-up KSS scores did not yield any significant difference in the two treatment groups, except for the KSS-F difference between the preoperative value and that of the three-month follow-up.
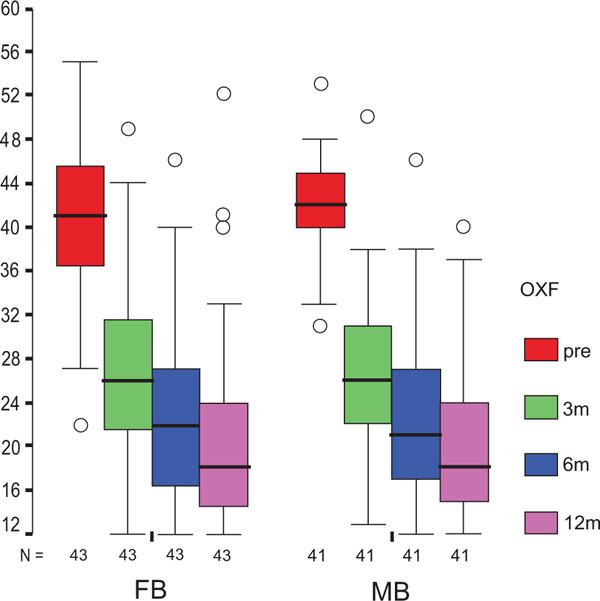
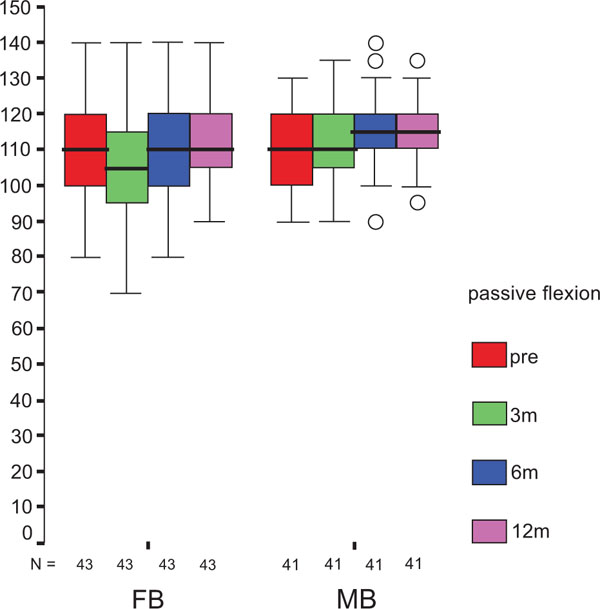
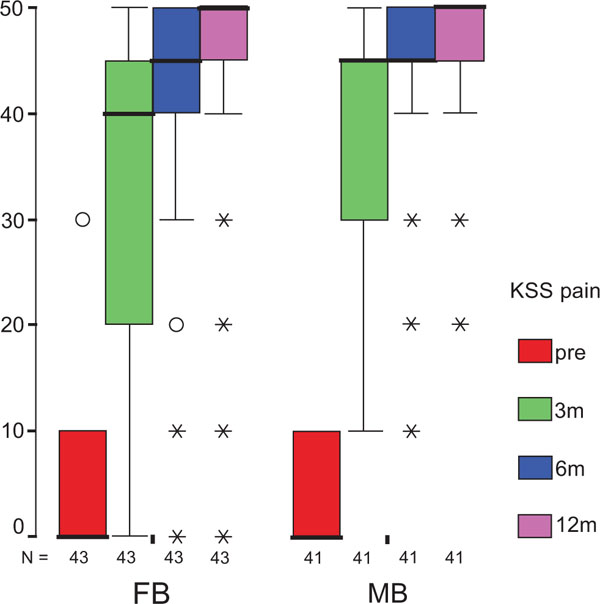
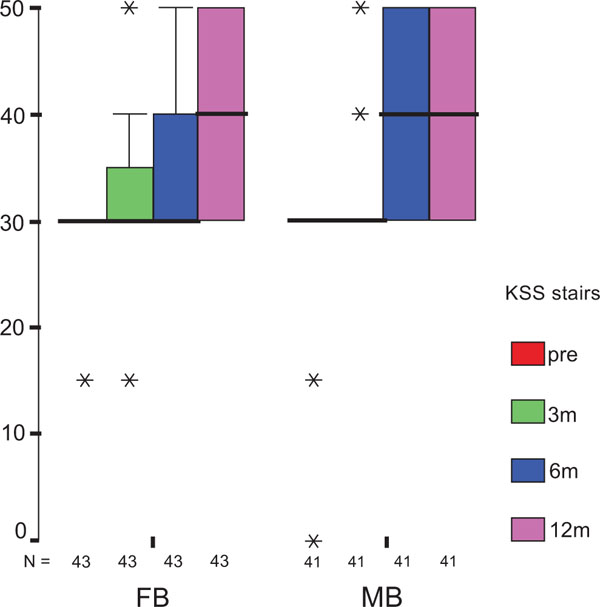
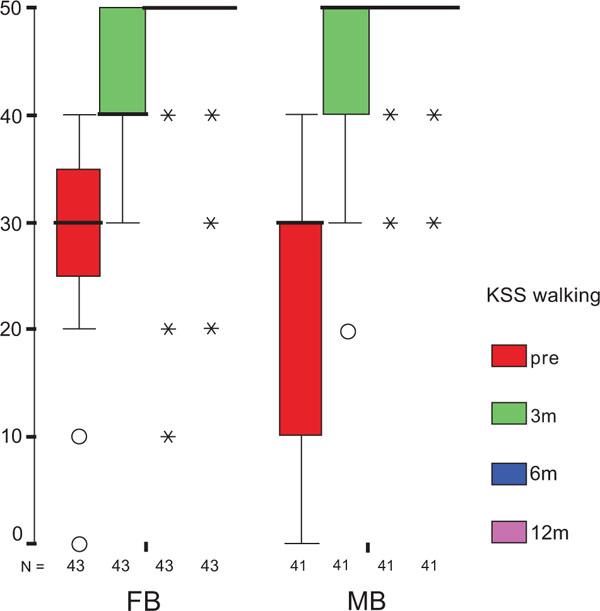
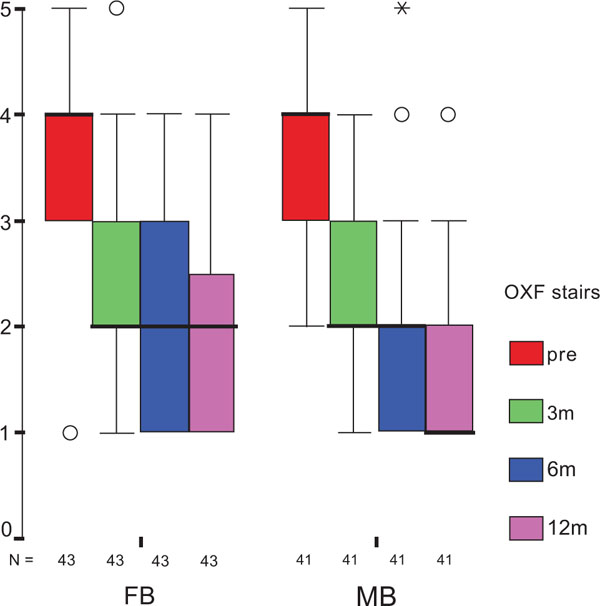
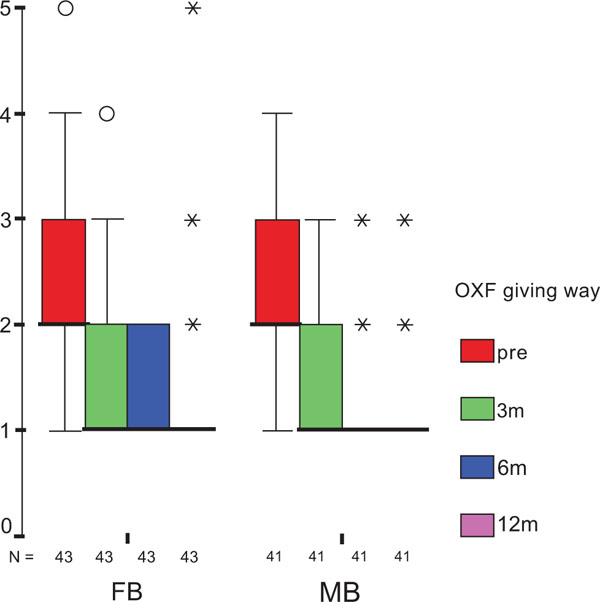
The treatment group did not influence the one-year OXF difference (Fig. 2c) significantly (p = 0.434) while the baseline score did (p = 0.001), with adjusted means of 21 ±1 (19–23) for FB, and 20 ±1 (18–22) for MB. On the range of motion (ROM FLX, Fig. 2d), the type of the platform, fixed or mobile, did not have a significant influence (p = 0.363), but, it did have a significant influence on preoperative flexion (p<0.001), with means of 113°±2° (110°–116°) for FB, and 115°±2° (112°–118°) for MB. Functional sub-items of the two scoring schemes are shown in Fig. (3). Low baseline KSS walking scores were identified as the source of the random KSS-F baseline difference between the groups. No clinically relevant differences could be identified.
Radiographically (Table 4), there were no significant differences between the FB and MB groups with regard to mechanical axis (p = 0.291), femoral frontal (p = 0.104), tibial frontal (p = 0.813), femoral sagittal (p = 0.560), or tibial sagittal (p = 0.929) alignment.
DISCUSSION
In the first post-operative year, the design of primary TKA for osteoarthritis (FB or MB) does not account for more than an eight-point difference in the KSS score. The study design allowed to vary only one factor, i.e. a fixed or mobile platform, while keeping all other relevant factors constant.
All other components of the implant are identical in both mobile or fixed versions. We used computer-assisted instrumentation to ensure consistent implant alignment [23, 24]. We noted an anomaly in the clinical data, the baseline KSS-F scores being 10 points lower for the patients properly randomized to the MB group. Nevertheless, the results still indicate that the influence of the prosthesis variant was not significant, the difference remaining below the postulated clinical relevance of an eight- point score difference. Yet, it would have been better to control baseline score values already in the randomization phase.
Several randomized studies compared FB and MB TKA, with some comparing different grades of mobility within one implant family [25-29], and others different implants [30, 31]. Furthermore, bilateral comparisons were performed in both randomized [22, 32-36] or non-randomized studies [37-40]. Seven out of the ten bilateral studies employed one-stage bilateral surgery. Identifying the implant’s contribution to the difference in functional result seems difficult when each patient is his own control. Our study is the only one with patients stratified by age and gender. In addition, no other randomized study used computer-assisted instrumentation. In a recent review comparing fixed versus mobile inlays, Van der Bracht et al. found only six papers meeting their quality criteria [41], none of which using a similar implant design that differed only in the tibial bearing interface. Only two of them included a power analysis. In four studies, they found weak statistical reporting and were unable to perform a meta-analysis due to the heterogeneity of the statistical methods applied.
The results of this study are within the ranges reported in previous research. However, in our study population, the baseline KSS-F tends to be higher, the baseline KSS-K lower. There is only 1 [30] out of 17 studies with a higher baseline KSS-F and only 3 [36, 39, 40] out of 17 studies with a lower baseline KSS-K. But the other features of the respective study populations do not provide a conclusive explanation for this finding.
Most cited studies confirm the lack of evidence for a difference between FB and MB treatment. Only Price et al. [22] report statistically significant differences in KSS-K scores (6 points) and OXF scores (2 points) and less pain at the one year follow-up, favoring the mobile design. The difference disappeared after 3.7 years [35]. Ranawat et al. [38] covered 2×26 two-stage bilateral TKAs examined after 1.3 and 3.8 years and found no statistically significant differences between the FB and MB knees in the final follow-up examination. Wohlrab et al. [29] showed a significantly better Hospital for Special Surgery Score (HSS) and less pain for the mobile design and also improved ROM in the MB patient group. However, the differences disappeared after three years.
One study produced lower KSS-K values at final follow-up (83 for both FB and MB) [25] and only one study similar KSS-K values for FB [22]. These were also the only ones to cover a maximum one-year follow-up, whereas the remaining 12 studies yielded better results (89–98 for FB and 90–97 for MB) over follow-up periods between 3 and 13 years [28, 30-40]. Among the studies reviewed, 10 studies used KSS F values as endpoint with 2 showing better results after 1 to 7 years (93–96 for FB, 93–95 for MB) [32, 38] and the others worse results after 1 to 13 years (65–86 for FB, 61–86 for MB) [25, 28, 30, 31, 34, 36, 39, 40].
Concerning OXF scores, the literature reports a slightly worse [22] or comparable outcome compared to this study (21–24 for FB, 21–26 for MB after 1–4 years) [25] and concerning ROM, smaller flexion angles in 6 cases [22, 25, 29, 30, 33, 37] and higher ones in 9 cases [27, 28, 31, 32, 34, 36, 38, 40].
Regarding implant-specific complications leading to revisions, 7 studies report inlay dislocations [22, 31, 33, 35, 36], occurring both with posterior stabilized and cruciate retaining implant designs, and all within the first six months. We did not encounter this implant-specific complication. This is due, we suggest, to the optimal gap balancing and joint line restoration of the standardized computer-assisted workflow.
The most important limitation of our study is its short follow-up which is not sufficient to demonstrate a potential reduction of wear on the mobile platform. However, it does allow to realistically evaluate the function of the artificial joint as it can be assumed that a potential difference in function will not grow with time between the two groups [29, 38]. In the follow-up over the next five years, we plan to assess anterior knee pain as a symptom of malfunction of the knee joint, since it has already been demonstrated that a posterior stabilized TKA with a mobile platform implant can lead to significantly less anterior knee pain than the fixed version [25].
Even with a computer-assisted operative technique for better control of operation parameters, and identical implant components, except for the mobility-relevant bearing, the results to date of this study do not show any difference in clinical outcome. Nevertheless, future follow-up of the present cohort will allow to compare the two designs at mid- and long-term.
ACKNOWLEDGEMENTS
The authors would like to thank Klaus-Ulrich Lemke, Schön Klinik Hamburg-Eilbek, Dept. of Orthopedic Surgery, for his help during admission of the patients, and S. David Stulberg, M.D., Northwestern University, Feinberg School of Medicine, Chicago, IL, US, for his helpful discussion with the authors regarding the concept of the study.
This study was funded by BBraun Aesculap, Tuttlingen.