All published articles of this journal are available on ScienceDirect.
Lumbar Solitary Osteochondroma with Lower Extremity Weakness: A Case Report
Abstract
Background:
Osteochondroma is the most common benign bone tumor. However, osteochondroma of the spine is relatively rare, accounting for only 4% to 7% of primary benign spinal tumors and less than 3% of all osteochondromas. Furthermore, symptomatic osteochondroma in the lumbar spine is much rarer. We herein describe a patient who was diagnosed with a lumbar solitary osteochondroma causing neurological symptoms of the lower extremities. The patient successfully underwent surgical removal of the lesion.
Case Presentation:
A 79-year-old man presented with a 3-year history of difficulty walking and gradual development of numbness in his left upper extremity, which further exacerbated his difficulty walking. Magnetic resonance imaging and computed tomography revealed spinal stenosis at C3/4, L4/5, and L5/S1 and an osseous solitary lesion. We performed a single-stage operation of both the cervical and lumbar spine. The osseous solitary lesion at L5/S1 was removed, and the pathological diagnosis was osteochondroma. After the operation, the patient’s upper extremity numbness and walking ability were improved.
Conclusions:
We experienced a rare case of a neurologically symptomatic solitary osteochondroma in the lumbar spinal canal. Moreover, this lesion was found at the same time as cervical spondylosis causing numbness. Therefore, we must perform careful examinations and thoroughly evaluate imaging and postoperative pathological findings.
1. BACKGROUND
Osteochondroma is the most common benign bone tumor, constituting 20% to 50% of all benign bone tumors and 10% to 15% of all bone tumors [1, 2]. Osteochondroma is most often found in long bones; reports suggest that osteochondroma of the spine is relatively rare, accounting for only 4% to 7% of primary benign spinal tumors and less than 3% of all osteochondromas [1, 3-5].
Several reports have shown that spinal osteochondroma is generally associated with multiple hereditary exostoses and that solitary osteochondroma (SOC) is rare [3, 5, 6]. Furthermore, symptomatic osteochondroma in the lumbar spine is much rarer [7].
In this article, we present a case involving the surgical treatment of SOC of the lumbar spine with neurological symptoms affecting the lower extremities.
2. CASE PRESENTATION
A 79-year-old man presented with a 3-year history of difficulty walking. He had developed gradually worsening numbness in his left upper extremity, which further exacerbated his difficulty walking. He visited the nearest general hospital and was referred to our department for further treatment.
Hoffmann’s reflex in both upper extremities was accelerated, and he was unable to perform delicate movements such as buttoning and writing. He had no muscle weakness or sensory disturbance in his upper extremities. However, sensory disturbance was present in both lower extremities with muscle weakness of the iliopsoas (manual muscle testing [MMT]: fair/normal), tibialis anterior (MMT: poor/poor), extensor hallucis longus (MMT: fair/fair), and gastrocnemius (MMT: fair/fair). His bilateral patella tendon reflex was accelerated because of a cervical lesion. His bilateral Achilles tendon reflex was reduced. No bladder or bowel disturbance was found.
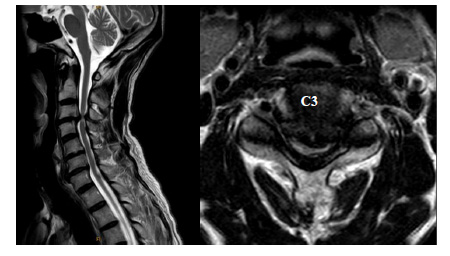
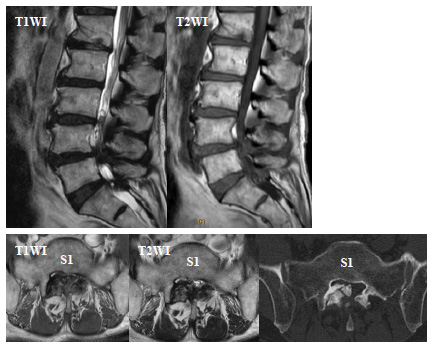
Magnetic resonance imaging (MRI) revealed spinal stenosis at C3/4, L4/5, and L5/S1 (Figs. 1 and 2). The right lesion at the level of L5/S1 appeared as a hyperintense area on both T1- and T2-weighted imaging in the thickened ligament flava. Computed tomography (CT) showed a high-density solitary lesion at this site, and we suspected an osseous tumorous lesion (Fig. 2).
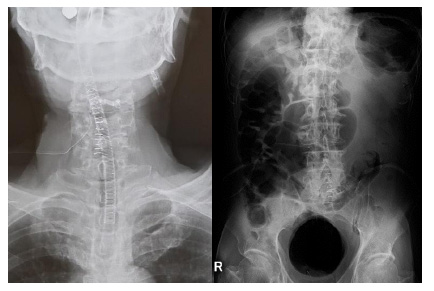
We performed laminectomy of L5 and S1 with removal of the solitary lesion. In the cervical spine, we conducted laminectomy of C3 and laminoplasty of C4 (Fig. 3).
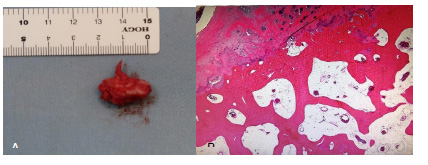
Histological analysis revealed that the lesion was mature cancellous bone and covered with a thin cartilage cap. The final pathological diagnosis was osteochondroma (Fig. 4).
The postoperative course was uneventful. The patient’s upper extremity numbness was improved, and he was able to walk again.
3. DISCUSSION
SOC most commonly affects the cervical spine and usually arises from the posterior arch of the vertebra. The lumbar spine is the third most common site of SOC after the cervical and thoracic spine [3]. In a review including 132 cases by Yakkanti et al. [8], the most commonly involved spinal levels were in the cervical region (65 cases, 52%), followed by the lumbar (35 cases, 27%), thoracic (24 cases, 18%), sacral (9 cases, 7%), and coccygeal (1 case, 1%) regions. The most frequently involved spinal anatomic column was the posterior column (85 cases, 64%), followed by the unknown (28 cases, 21%) and the anterior column (19 cases, 14%); [8]. In addition, neurological symptoms caused by spinal osteochondroma are atypical because most of these lesions grow out of the spinal canal [7]. Kahveci et al. [9] reported a unique case of lumbar SOC presenting with footdrop. In another report, they summarized 21 cases of lumbar SOC associated with symptoms including low back pain, numbness, and weakness, and only 4 of these 21 patients were aged >60 years [10].
Yagi et al. [11] and Sakai et al. [12] reported several cases of growth of SOC even though skeletal maturation had long since been completed, degenerative changes in the spine led to spinal cord compression in elderly patients. In our case, the patient was 79 years old, and MRI showed degenerative changes. As a result, both the L5 and S1 nerve roots were compressed within the spinal canal and at the lateral recess, which was one of the factors that caused the neurological symptoms in this elderly patient with SOC.
CT and MRI are effective for the diagnosis of SOC. CT can show the diagnostic appearance of the marrow and cortical continuity with the underlying vertebra. MRI shows a hypointense peripheral lesion corresponding to the cortical bone and a hyperintense central lesion corresponding to the cancellous bone, making the lesion clear [13]. Therefore, the use of preoperative imaging is important because it may reveal special diseases associated with bone lesions.
Many surgeons agree that surgical treatment of symptomatic osteochondroma is the best method for recovery of neurological function and avoidance of future complications. In one study, surgical treatment reportedly improved the neurological deficit in nearly 90% of patients whose spinal cord was compromised by SOC [9].
Dahlin and Unni [1] reported that the risk of local recurrence of surgically treated SOC of the limbs was 2%, with a highly variable disease-free interval ranging from 1 to 26 years. The risk of recurrence is greater in children [1]. Gille et al. [14] reported that the risk of recurrence of SOC in the spine was 4%, and the mean period until recurrence was 5 years (range, 6 months to 14 years). They also reported that incomplete removal of the cartilaginous cap may lead to tumor recurrence [14]. For this reason, we should not hesitate to completely resect the lesion, even if this compromises the stability of the vertebral column, necessitating osteosynthesis [14]. SOC is associated with a risk of malignant transformation. The estimated frequency of degeneration is about 1%, and its histological type is chondrosarcoma [1]. However, osteochondroma of the spine may have a higher risk of malignant transformation than peripheral osteochondroma. According to Sciubba et al. [15]. Malignant transformation occurred in 3 of 27 patients who were diagnosed with osteochondroma of the spine and received surgical treatment in a multicenter study. The incidence was 11%, higher than that of peripheral lesions [15]. Other risk factors for malignant transformation are the presence of a >3-cm-thick cartilaginous cap, new onset of symptoms, and a rapid increase in the size of the tumor [15, 16].
In our patient, the cartilaginous cap was thin (<3 cm) and the growth was not rapid. However, SOC has a risk of malignant transformation, as mentioned above. Therefore, clinicians must pay attention not only to neurological symptoms but also to recurrence and malignant transformation in patients with SOC.
CONCLUSION
In conclusion, we experienced a rare case of a neurologically symptomatic SOC in the lumbar spinal canal. Moreover, the SOC was found at the same time as cervical spondylosis causing numbness. Therefore, we must perform careful examinations and thoroughly evaluate imaging and postoperative pathological findings.
LIST OF ABBREVIATIONS
| SOC | = Solitary Osteochondroma |
| MMT | = Manual Muscle Testing |
| MRI | = Magnetic Resonance Imaging |
| CT | = Computed Tomography |
AUTHORS’ CONTRIBUTIONS
TK performed the surgery. TS and HK prepared the manuscript. NM assisted in drafting and reviewing the manuscript. All authors read and approved the final manuscript.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
STANDARDS OF REPORTING
Care guidelines were followed.
AVAILABILITY OF DATA AND MATERIALS
The data supporting the findings of this study are available within the article.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGMENTS
We thank Angela Morben, DVM ELS, from Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript.


