All published articles of this journal are available on ScienceDirect.
Contrast-enhanced Magnetic Resonance Imaging Revealing the Joint Capsule Pathology of a Refractory Frozen Shoulder
Abstract
Background:
Frozen shoulder (FS) is clinically diagnosed on the basis of patients’ medical history and physical examination. Its confirmation is based on joint capsule and coracohumeral ligament thickening, subcoracoid fat obliteration, and joint capsule contrast enhancement on magnetic resonance imaging (MRI). We performed bilateral contrast-enhanced MRI (CE-MRI) in FS patients to compare the outcomes with those of their unaffected contralateral counterparts.
Methods:
Ten patients (3 men, 7 women, median age: 54.5 years) with unilateral FS, requiring arthroscopic capsular release after failed conservative treatment, were included. The median forward elevation, abduction, external rotation, and internal rotation of the 10 patients were 100°, 60°, 7.5°, and the buttock, respectively. The median visual analog scale score was 5.3, and American Shoulder and Elbow Surgeons (ASES) score was 42. Bilateral CE-MRI was simultaneously performed on the day before surgery, and MRI findings were compared between FS and contralateral healthy shoulders (controls).
Results:
Significant axillary pouch enhancement and rotator interval were observed in all FS, but not in the unaffected comparable sides (p=0.002, respectively). The thickness of the axillary pouch (FS: 4.8 mm, C: 4.4 mm, p=0.58), coracohumeral ligament (FS: 3.9 mm, C: 4.1 mm, p=0.33), and subcoracoid fat obliteration (p=1.00) were not significantly different between FS and controls.
Conclusion:
CE-MRI aids in the clinical diagnosis of FS. However, axillary pouch joint capsule and coracohumeral ligament thickening or subcoracoid fat obliteration differences were not characteristic findings when contralateral shoulders were compared.
1. INTRODUCTION
Frozen shoulder (FS) or adhesive capsulitis is a common disease, and most cases can be managed successfully with con-
servative treatments [1-3]. Inflammation and subsequent fibrosis of the joint capsule are thought to be one of the main pathologies of FS [4-7], although its etiology is not fully understood. The condition has long been considered self-limiting, and more recent literature has reported that less optimistic outcomes were achieved through conservative treatments such as a combination of anti-inflammatory medications, physical therapy, and domestic fitness regimens [1, 8]. Following FS diagnosis, physical therapy and intra-articular steroid injections have been found to decrease pain and assist in regaining range of motion (ROM) [9, 10]. Manipulation under anesthesia or arthroscopic capsular release may be considered upon failure of conservative treatments [7, 11, 12].
Medical history and physical examination findings, such as pain and restricted active and passive ROM in all planes, support the clinical diagnosis of FS [1, 13, 14]. However, imaging is commonly required to assist FS diagnosis as other shoulder problems might mimic FS [15]. Radiography is generally unremarkable and is only beneficial for ruling out other shoulder disorders [16]. Magnetic resonance imaging (MRI) is a noninvasive modality that could clarify changes in the joint capsule or synovial membrane, thereby distinguishing FS from other painful shoulder disorders [5, 13]. Previous studies using MRI as a diagnostic tool for detecting FS revealed features such as thickening of the joint capsule in the axillary pouch (AP), thickening of the coracohumeral ligament (CHL), and fibrosis of the subcoracoid fat in the rotator interval (RI) [16-20]. However, other investigators described that MRI alone showed no specific abnormalities with FS, but these alterations were often observed in other shoulder disorders, such as in rotator cuff tear [4, 21].
The usefulness of contrast-enhanced MRI (CE-MRI) in the diagnosis of FS has been widely recognized [5, 14, 15]. CE-MRI can display hypervascularity of the joint capsule in FS or other inflammatory conditions such as rheumatoid arthritis [4, 14, 16, 17, 22]. Additionally, observations of joint capsule enhancement have been made in most cases of FS, but not in cases of rotator cuff tears [17, 22]. During arthroscopic capsular release for FS, excessive bleeding from the capsule is often observed. Moreover, Okuno et al. recently reported that increased abnormal blood vessels were observed in patients with refractory FS, where angiography and transcatheter embolization of the abnormal vessels were found to be effective [23, 24]. Increased abnormal blood vessels are detected with CE-MRI and might be another pathologic feature of FS. However, this has to be elucidated through a comparatively large-scale study with respective normal opposite shoulder side counterparts.
The purpose of this study was to 1) compare CE-MRI features between the FS and contralateral non-affected shoulder and 2) compare AP joint capsule thickness, CHL thickness, and subcoracoid fat obliteration on both sides. We also performed a reliability analysis of the measurements, as no gold standard methods had been developed.
2. MATERIALS AND METHODS
2.1. Patients’ Demographics, Physical Shoulder Measurements, and Imaging
From April 2017 to March 2018, 10 FS patients who had an arthroscopic capsular release after failed conservative treatments for at least 3 months were included in this study. Patients with diabetes mellitus and stiffness after apparent trauma or previous surgery were excluded from the study.
The study cohort comprised three male and seven female FS patients, with a median age of 54.5 years. FS was present in six right shoulders and four left shoulders; all patients were right-side dominant. The patients were diagnosed with FS based on clinical symptoms, radiography, and conventional MRI. All patients met the criteria for FS outlined by the ISAKOS Upper Extremity Committee [7]. Briefly, FS was identified by forward elevation <100°, external rotation <10°, and internal rotation <L5 level without anatomic, imaging, or systemic abnormalities. Conservative treatment included nonsteroidal anti-inflammatory drugs, a few intra-articular injections of corticosteroid or hyaluronan, and rehabilitation by physiotherapists for at least 3 months. On the day before surgery, physical findings were re-examined, and CE-MRI of the bilateral shoulders was performed. The median symptom duration from onset to the time of CE-MRI and ROM measurements of the affected and unaffected shoulders using a conventional goniometer were recorded. The visual analog scale (VAS) scores ranged from 0 (no pain) to 10 (worst pain imaginable), with 1-cm intervals. Patient questionnaires regarding shoulder joint pain, instability, and activities were administered, and the American shoulder elbow surgeons (ASES) scores before surgery were recorded (Table 1).
| Median | IQR | |
|---|---|---|
| Age (years) | 54.5 | 49.5, 62.0 |
| Disease period (month) | 7.5 | 5.8, 9.0 |
| Number of steroid injections | 3 | 3, 3 |
| VAS | 5.3 | 4.0, 7.4 |
| ROM of the affected side (°) | ||
| FE | 100 | 87.5, 120 |
| ABD | 60 | 40, 80 |
| ER | 7.5 | 0, 10 |
| IR | B | L5, B |
| ROM of the unaffected side (°) | ||
| FE | 170 | 167.5, 175 |
| ABD | 170 | 167.5, 175 |
| ER | 60 | 55, 62.5 |
| IR | T8 | T7, T9 |
| ASES score | 42 | 33, 56.5 |
2.2. Contrast-enhanced MRI Assessment
The subject was asked to lie in a supine position with the upper arm relaxed in neutral position. Then, contrast material at a dose of 0.2 ml/kg (Gadoteridol, Eisai Corp., Tokyo, Japan) was injected intravenously before initiation of MRI examinations, which were undertaken using a 1.5-Tesla scanner (Siemens Aera, Siemens AG, Munich, Germany) with a dedicated shoulder coil. Pulse sequences were as follows: 1) oblique coronal, oblique sagittal, and axial T2-weighted turbo spin-echo (TSE) (TR/TE: 3600/78 ms); 2) oblique coronal, oblique sagittal, and axial T1-weighted TSE (TR/TE: 700/13 ms); and 3) oblique coronal, oblique sagittal, and axial post-gadolinium T1-weighted fat-suppressed TSE (TR/TE: 600/13 ms) of the bilateral shoulders. CE-MR images were obtained within 30 min of the injection.
The thickness of the AP joint capsule and CHL, subcoracoid fat obliteration, and AP joint capsule and RI enhancement were evaluated following the method of Ahn et al., outlined in Table 2 [15, 16]. MR images were reviewed by two orthopedic surgeons who were blinded to the clinical data of the patients. The first orthopedic surgeon measured each MR image twice at 3-week intervals to ensure intraobserver reliability. The second orthopedic surgeon also measured each MR image for interobserver reliability. A comparison was made between the scores recorded by each of the orthopedic surgeons.
2.3. Statistical Analysis
Values were expressed as median (interquartile range; IQR) or as a percentage. Wilcoxon’s signed-rank test was used for AP joint capsule and CHL thicknesses. Friedman test was employed for subcoracoid fat obliteration, AP joint capsule enhancement, and RI enhancement. Two-tailed tests were performed to determine the statistical differences between and within groups of affected shoulder side and unaffected side. A p-value < 0.05 in each test was considered statistically significant. Intraobserver and interobserver agreements of MRI assessments were evaluated, and the intraclass correlation coefficient (ICC) for AP joint capsule and CHL thicknesses was calculated. For subcoracoid fat obliteration, AP joint capsule enhancement, and RI enhancement, Gwet’s agreement coefficient (AC) 1 was calculated. Values were rated as poor (0.00-0.20), fair (0.20-0.40), good (0.40-0.75), and excellent (0.75-1.00). SPSS version 24.0 (SPSS Japan Inc., Tokyo, Japan) or STATA version 14 (Lightstone Corp., Tokyo, Japan) was employed in all statistical analyses.
| Measurement method | |
|---|---|
| AP thickness | Widest portion of the capsule, which was at the mid-portion of the humeral head, measured with a digital caliper on oblique coronal T2-weighted images |
| CHL thickness | Widest portion of CHL at the level of the tip of the coracoid process measured with a digital caliper on oblique sagittal T2-weighted images |
| RI obliteration | Graded on oblique sagittal T2-weighted images None: absence of fat obliteration Partial: partially disrupted fat signal Complete: totally obliterated fat signal |
| AP enhancement | Graded on oblique coronal contrast-enhanced T1-weighted fat-suppressed images None: no enhancement Mild: insufficient enhancement or sufficient enhancement involving less than 1/3rd of the capsule hemicircumference Moderate: sufficient enhancement involving less than 2/3rd of the capsule hemicircumference Severe: sufficient or strong enhancement involving over 2/3 of the capsule hemicircumference |
| RI enhancement | Graded on oblique sagittal contrast-enhanced T1-weighted fat-suppressed images None: no enhancement Partial: insufficient enhancement or partial sufficient enhancement Complete: total sufficient enhancement |
3. RESULTS
3.1. Patient Demographics and Verbal and Physical Shoulder Assessment
The median symptom duration from onset to the time of CE-MRI was 7.5 months, and the median number of steroid injections was three. The median VAS at the time of CE-MRI was 5.3 points. The median forward flexion, abduction, external rotation, and internal rotation of the affected and unaffected sides were 100°, 60°, 7.5°, and the buttock, and 170°, 170°, 60°, and the 8th thoracic vertebra, respectively. The median ASES score was 42 points (Table 1).
3.2. MRI Assessment
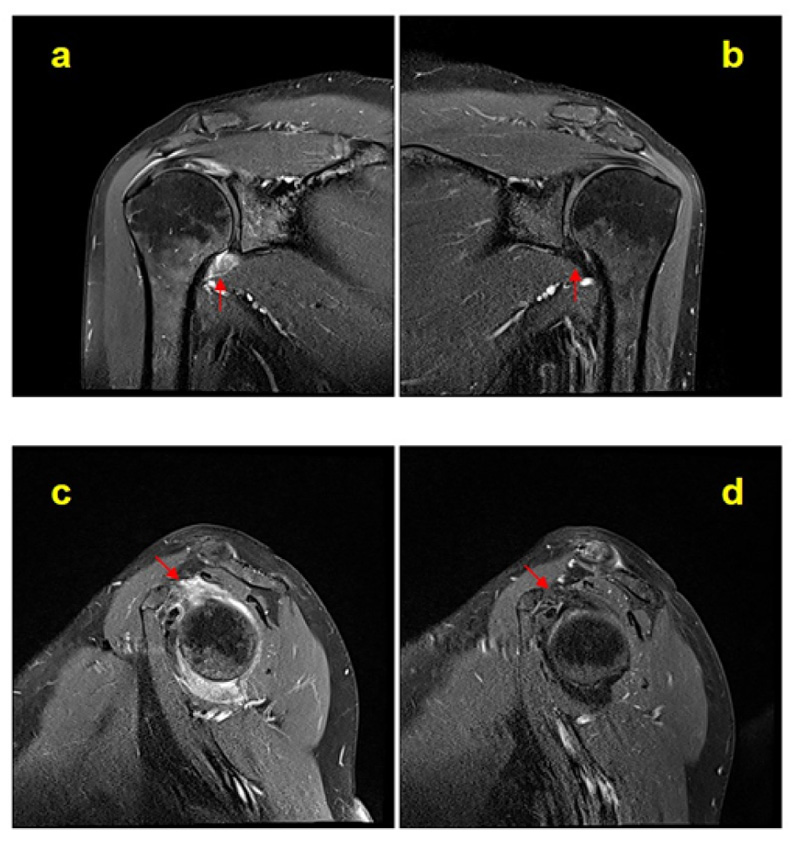
We determined the AP joint capsule and CHL measurement sites on the oblique coronal and oblique sagittal T2-weighted MR images. The widest region of the low-signal area of the AP joint capsule and CHL was measured. Fig. (1) shows an example of bilateral T2-weighted MR coronal and sagittal images of the measurement site of the joint capsule and CHL. Enhancement of the AP joint capsule and RI was evaluated with contrast-enhanced T1-weighted fat-suppressed coronal and sagittal images. Contrast-enhanced T1-weighted fat-suppressed coronal and sagittal images are illustrated in Fig. (2).
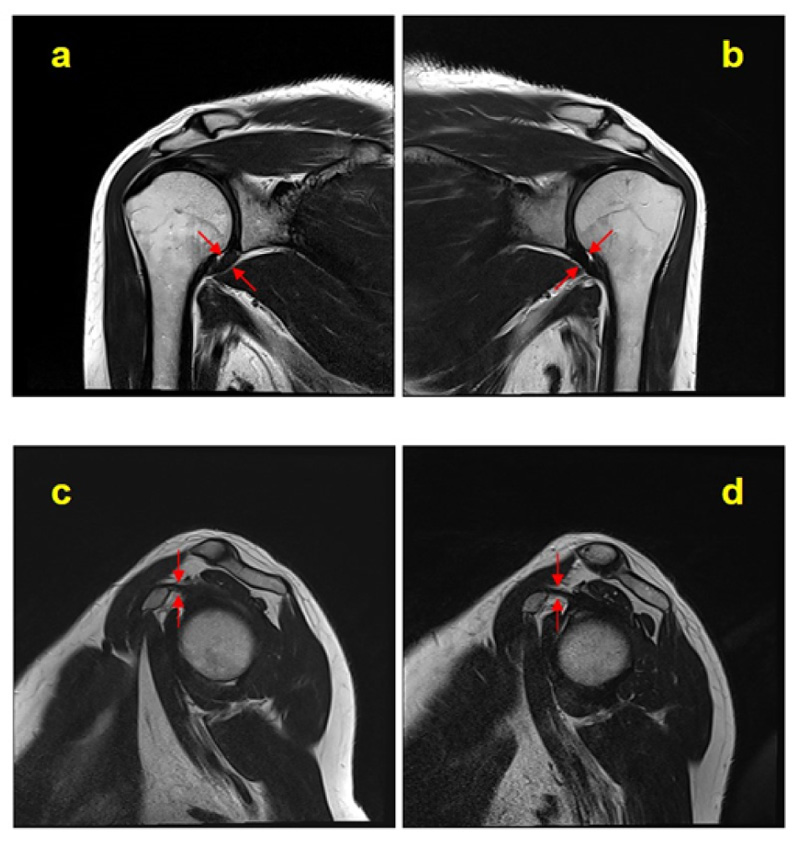
Table 2 presents the methods of MRI evaluation previously described by Ahn et al [15, 16]. The AP thickness, CHL thickness, subcoracoid fat obliteration, and AP joint capsule and RI enhancement in MR images were obtained, and measurements of the FS and unaffected shoulder were compared.
Our MRI assessment parameter measurements of the FS and the contralateral shoulder demonstrated no significant differences in AP joint capsule thickness (p = 0.58), CHL thickness (p = 0.33), and subcoracoid fat obliteration (p = 1.00) (Table 3). Significant differences were observed in AP joint capsule enhancement (p = 0.002), which was severe in the affected side and nonexistent in the unaffected side. Significant differences regarding RI enhancement were observed (p = 0.002), which was 90% complete and 10% partial in the affected side and 90% none and 10% partial in the unaffected side (Table 3).
3.3. Reliability Assessment
Good to excellent correlation was found regarding the intraobserver reliability and interobserver reliability for most anatomic structures, such as AP joint capsule thickness, CHL thickness, subcoracoid fat obliteration, and AP and RI enhancements (Table 4). However, the intraobserver reliability of CHL thickness in FS, interobserver reliability of AP joint capsule thickness in FS and control cases, and RI enhancement in FS cases ranged from fair or poor.
4. DISCUSSION
In this study, contrast-enhanced bilateral MRI was performed in 10 patients with unilateral refractory FS after failed conservative treatments. This study found global enhancement of the joint capsule to be significantly higher in the affected than the unaffected side, indicating abnormal blood flow in the joint capsule. Moreover, no differences were observed concerning the AP joint capsule thickness, CHL thickness, or subcoracoid fat obliteration when comparing affected to unaffected contralateral shoulders.
| Affected side | Contralateral | P-value | |
|---|---|---|---|
| AP thickness, median (IQR) | 4.8 (4.0, 5.6) | 4.4 (3.0, 5.5) | 0.58 |
| CHL thickness, median (IQR) | 3.9 (3.3, 5.7) | 4.1 (2.6, 5.3) | 0.33 |
| RI obliteration, number (%) | |||
| None | 0 | 0 | 1 |
| Partial | 5 (50) | 5 (50) | |
| Complete | 5 (50) | 5 (50) | |
| AP enhancement, number (%) | |||
| None | 0 | 10 (100) | 0.002 |
| Mild | 0 | 0 | |
| Moderate | 0 | 0 | |
| Severe | 10 (100) | 0 | |
| RI enhancement, number (%) | |||
| None | 0 | 9 (90) | 0.002 |
| Partial | 1 (10) | 1 (10) | |
| Complete | 9 (90) | 0 |
| ICC (1, 1) or Gwet’s AC1 | ICC (2, 1) or Gwet’s AC1 | ||
|---|---|---|---|
| AP thickness | FS | 0.84 | 0.31 |
| C | 0.79 | 0.42 | |
| CHL thickness | FS | 0.46 | 0.82 |
| C | 0.86 | 0.69 | |
| RI obliteration | FS | 0.80 | 0.65 |
| C | 0.80 | 0.65 | |
| AP enhancement | FS | 1.00 | 0.76 |
| C | 0.89 | 0.89 | |
| RI enhancement | FS | 1.00 | 0.083 |
| C | 0.87 | 0.87 |
The diagnosis of FS is often based on physical findings rather than imaging studies. However, this is problematic as other shoulder diseases can mimic FS. Imaging is, therefore, likely to play an essential role in the avoidance of misdiagnosis [15]. AP capsular thickening, CHL thickening, subcoracoid fat obliteration in RI, and T2-weighted signal increase in the AP joint capsule have been identified as features of FS [14, 15, 17, 25]. Emig et al. previously measured the thickness of the AP joint capsule and reported a thickness greater than 4 mm to be a useful criterion for the diagnosis of FS [25]. Ahn et al. further suggested that a thickness of 5 mm provided the highest sensitivity and specificity [15]. In contrast, no significant difference in AP joint capsule thickness was observed by Mengiardi et al. and Manton et al. [19, 26]. Further, FS patients have been identified as having a thicker CHL than control subjects [19]. However, this finding has also not been consistent, as further findings discovered no significant CHL thickness differences between FS and control patients [25]. Thus, MRI outcomes of FS pathology studies have been controversial. In the current study, AP joint capsule thickness and CHL thickening differences between FS and contralateral shoulders were not significantly different, according to our CE-MRI results, which supported the current presumption that thickening of the joint capsule or CHL could be a natural aging phenomenon and not necessarily an FS pathology. Diagnosing patients as having FS because the joint capsule or CHL “looks” thickened is wrong.
A previous study using MRI has found subcoracoid fat obliteration in 50% of FS patients and 26% of controls [14]. Furthermore, Mengiardi et al. identified that partial or complete subcoracoid fat obliteration was significantly more frequent in FS patients [19]. In agreement with previous studies, Ahn et al. observed partial or complete obliteration in all FS patients, in addition to a lack of correlation with ROM limitation [16]. Our CE-MRI evaluation demonstrated no subcoracoid obliteration differences between the FS and contralateral shoulders, thereby suggesting that subcoracoid obliteration might be due to an aging process. Thus, clinicians are advised to consider the aging process upon identification of the thickened joint capsule in association with shoulder pain.
Carrillon et al., using CE-MRI to detect increased blood flow in the joint capsule, reported enhancement of the joint capsule in all FS patients (n = 25), but in only one of the 15 rotator cuff tear patients [22]. This finding was supported by Connell et al.’s study in which enhancement of the joint capsule was found in 22 of 24 surgically treated FS cases and two of the patients with rotator cuff tear as controls (n = 22) [17]. Furthermore, AP joint capsule enhancement was identified in 49 of 50 FS patients, but only 19 of 53 control subjects [15]. In our present study, all FS patients demonstrated global high-intensity changes in the joint capsule, but none in the contralateral shoulders. Hence, CE-MRI is able to detect the qualitative alteration of the joint capsule [27]. The procedure could, therefore, be performed in severe cases of failed conventional conservative therapy. Intra-articular steroid injections were considered useful in case of high enhancement, and surgery such as arthroscopic capsular release was required in patients with prolonged motion limitations despite having low enhancement [4]. However, in this study, global enhancement of the joint capsule was identified in all patients (n = 10), even after sufficient periods of conservative therapy, including several intra-articular steroid injections. Therefore, in some cases, inflammation was not resolved even after adequate conservative therapy. In contrast, when the joint capsule was not enhanced, extra-articular joint stiffness factors such as conditions of the scapula, muscle stiffness, or central problems should be considered. Recently, Okuno et al. described the results of angiography in patients with refractory FS [23, 24]. Neovessels were found to have been generated around the RI, mainly from the thoracoacromial artery or coracoid branch, and around the AP from the circumflex scapular artery or anterior or posterior circumflex artery. Moreover, the pain was found to improve rapidly after the embolization of the abnormal neovessels. These results indicate increased abnormal vessels to be the source of pain which is supported by findings of ongoing persistent inflammation in patients with refractory FS. Performing angiography in every FS patient is challenging. However, our study provided evidence in support of the possibility of predicting the existence of abnormal neovessels with the use of CE-MRI.
This study has several limitations. First, most FS patients recover sufficiently through conservative therapy and invasive therapy is no longer in use. Therefore, only a small number of patients could be included in this study. However, to ensure the characteristic findings of FS, most severe cases were chosen, which will be a strength of this study. Despite the limited sample size, the study findings are valuable for developing insight into FS as a guide for clinical practice. Since there is no established quantitative MRI evaluation technique to date, the method presented by Ahn et al. was applied in the current study. The overall reliability of the method was found to be acceptable, although some constituents had low reliability, suggesting evaluation methodology. Finally, characteristics of MRI might differ depending on the stages of frozen shoulder (i.e., freezing, frozen, or thawing), which determine the severity of the disease. As the current study focused exclusively on surgically treated refractory cases, associations between disease stage, pain intensity, and MRI features remain unknown. Future studies should aim to address this gap in current knowledge.
CONCLUSION
CE-MRI can detect the qualitative alteration of the joint capsule and could be used to assist the clinical diagnosis of FS. Significant differences, however, were not observable, regarding the AP joint capsule thickness, CHL thickness, or subcoracoid fat obliteration when compared to the contralateral shoulders, implying these observable changes in FS could be normal wear and tear which comes along with aging rather than the disease state.
LIST OF ABBREVIATIONS
| ABD | = Abduction |
| AC | = Agreement Coefficient |
| AP | = American Shoulder and Elbow Surgeons |
| B | = Buttock |
| C | = Contralateral |
| CE | = Contrast-Enhanced |
| CHL | = Coracohumeral Ligament |
| ER | = External Rotation |
| FE | = Forward Elevation |
| FS | = Frozen Shoulder |
| ICC | = Intraclass Correlation Coefficient |
| IQR | = Interquartile Range |
| IR | = Internal Rotation |
| L | = Lumber Vertebrae |
| MRI | = Magnetic Resonance Imaging |
| RI | = Rotator Interval |
| ROM | = Range of Motion |
| T | = Thoracic Vertebrae |
| TSE | = Turbo Spin-Echo |
| VAS | = Visual Analog Scale |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This research has been approved by the institutional review board of the Matsuda Hospital (approved number is 29-1). Written informed consent was obtained from all participants.
HUMAN AND ANIMAL RIGHTS
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the Helsinki declaration.
CONSENT FOR PUBLICATION
All patients participated on a voluntary basis and gave their informed consent.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


