All published articles of this journal are available on ScienceDirect.
Long Term Results of Innovative Procedure in Surgical Management of Chronic Lymphedema
Abstract
Background:
Lymphedema is the result of impaired lymphatic drainage by the affected organ. This abnormality can be primary or secondary. Different operative approaches have been introduced to treat chronic lymphedema.
Materials and Methods:
This retrospective study included 816 patients who were diagnosed with chronic lower extremity lymphedema and did not respond to non-operative management for at least six months. Data was collected over 25 years, between March 1987 and March 2013. Doppler ultrasonography of the deep venous system was routinely undertaken in all patients to confirm patency. The patients underwent surgery and their progress was followed for at least one year postoperatively.
Results:
All patients were operated by the suggested technique and long term fallow-up which is a modified form of the Homan’s technique. The outcome was excellent, and 89.2% of patients were free of complication and 2% had poor results. The most common complication was wound seroma and wound infection.
Conclusion:
The long term results and considering the difficulties associated with the treatment of chronic lymphedema and the variety of surgical options, our method achieved excellent results, and may be proposed for the standard operative procedure for treating intractable forms of this disease.
INTRODUCTION
Lymphedema is a disease that in the congenital (primary) and acquired (secondary) forms causes numerous problems in the individual life [1-3]. Despite of many attempts that have been accomplished in order to cure this disease, but no certain cure has been still mentioned for it. Lymphedema due to the abnormalities of Lymphatic system leads to generate the inter-tissue liquid excessively which causes the symptoms by the side of patients in the form of inflammation below the knee or the whole of the limb [4-8].
The known lymphatic forms are as the inter-tissue liquid, stagnant lymph liquid containing protein and cellular remains which cause the inflammation in the affected limb. Lymphatic glands are responsible to transport the immunity chemical materials which are necessary for the body and cell. In the event of lack of cure, Lymphedema leads to create chronic inflammation, infection and harden the skin that in turn leads to increase the appearance changes and also heaviness of affected limb. Inter-tissue liquid can be created in any point of body which has improper lymphatic drainage [9-11]. Lymphedema is a disease which has been created slowly and gradually with increase of age and weight, the symptoms are also increased. If the patient is diagnosed with abnormalities in the lymphatic system since the birth, this kind of Lymphedema will be diagnosed as the primary Lymphedema.
The purpose of surgery operation in Lymphedema is to return the function of affected limb to the almost natural life. Numerous methods have been suggested for the Lymphedema surgery but none of them has been already able to be a standard method for this disease. From Excisional methods to the regenerative surgery methods, all have solved a part of problems of these patients.
We hope to witness the full recovery of these patients with progress of lymphatic system knowledge in the near future.
MATERIALS AND METHODS
This retrospective study was done on 816 patients since March 1987 till March 2013. All patients were affected by primary Lymphedema, some of them in the whole of limb from groin and some others who had later start of clinical symptoms from below the knee mentioned the inflammation. The age distribution of patients was 20-45 years and the start of disease symptoms was 1-35 years. The diagnosis progressed according to the patient, history and physical examination such as non-pitting edema of limb and inflammation from the distal toward proximal part. The inflammation without pain and heaviness of foot is a part of patients, specifications. Doppler ultrasound from deep venous system is necessary for the health of deep venous system. All patients have been under the conservative cures such as wearing special socks.
Surgical Technique
We performed staged subcutaneous excision beneath flaps in two or sometimes four stages.
Preoperative and Operative Management
At least two days before the operation patients were admitted to the hospital and antibiotic prophylaxis was begun, elevation of limb was done and bed rest was advised.
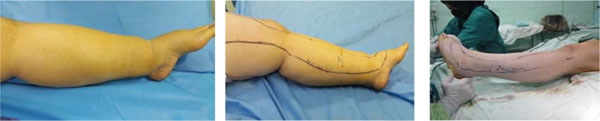
The patients were put in supine position under general or spinal anesthesia. A tourniquet was fastened around the highest part of the thigh (if possible). The affected limb was prepared and draped in such a way that the whole extremity was exposed. After limb exsanguination by elevation and rubber bandage, a tourniquet was inflated. Incision line was marked beforehand (Fig. 1a-c). The incision area included forefoot, ankle, and the whole calf from malleolus to a point at the same level of the tibial tuberosity at the first stage beginning from the medial part. Medial and lateral sides of the forefoot were excised with the same part of the calf.
The forefoot incision line was 4 cm apart from the metatarsophalangeal joints, 1 cm posterior to the malleolus and extended through the midsagittal plane (medial or lateral) of the calf (Fig. 2).
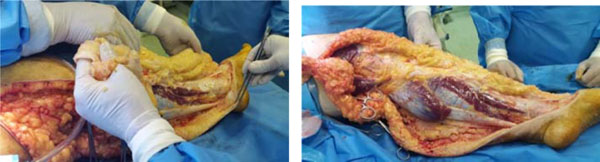
Flap thickness and length were variable depending on the part of the limb undergoing the procedure. Flap thickness was increased gradually from distal to proximal and it was 5 mm in forefoot, 6 mm in ankle, 8 mm in mid-calf, 10 mm near the knee and 15 mm in the thigh.
Flap length in both anterior and posterior directions is calculated as one-sixth of the limb circumference measured before making the incision -; thus, one-third of the limb perimeter was flapped during each period of surgery to minimize flap ischemia .
All subcutaneous tissues beneath the flaps are removed including deep fascia of the calf covering the muscles (Fig. 3).
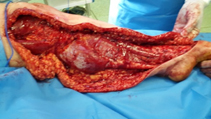
After tourniquet deflation and hemostasis, redundant skin from the flap edges was resected, closed suction drains have inserted beneath each flap, subcutaneous tissues were approximated by absorbable 2\0 Vicryl sutures and the skin was closed using sub-cuticular technique with absorbable strings. We did not immobilize the limb and we used non elastic bandage. After eight-nine days, drains were removed and the patient mobilized with natural rubber anti-varicose stockings; three months later, the second stage for the lateral part was performed. Such stages can be done for the thigh. Because the great saphenous vein is sacrificed during the medial side operation, deep venous patency must be confirmed in all patients preoperatively.
The patients underwent follow up examinations at one and two weeks and then - limb circumference was recorded at each session after one and a half year. During this period, they were questioned for any probable complaint or complication over the phone.
RESULTS
Among 816 patients, bilateral involvement was significant in 204 (25%) and surgery was indicated for both of the limbs. In 266 (32.7%) patients, unilateral involvement was found and the remaining 344 (42.2%)had bilateral involvement but operation was necessitated for one limb; 496 (60.8%) male and 320 (39.2%) female patients were treated; the male to female ratio was 3:2.
At one-year follow up, 716 (87.8%) patients were symptom- and complaint-free with minimal limb swelling and no disability(Table 1); 63 (7.7%) patients had acceptable results and became socially active but a disfiguring edema remained; 98 (4%) patients suffered slight skin changes and pigmentation.
The most common complication of our procedure was seroma that developed in 44 (5.4%) patients and was alleviated by repeated aspirations. Wound infection during the early postoperative period was rare and noted in 24 (3%) patients. Flap ischemia was rare and occurred in 19 (2.3%) patients, which was managed conservatively.
Final limb circumference reduction was ascertained in three levels at two follow up visits. Table 1 presents the average limb circumference reduction which was calculated after assessment of the patients postoperatively.
| Follow-up time | Six months after surgery (%) | One year after surgery (%) |
|---|---|---|
| Limb Level | ||
| Supra-malleolar | 29 | 36 |
| Mid-calf | 38 | 46 |
| Mid-thigh | 34 | 40 |
DISCUSSION
Diagnosis of lymphedema is at first clinical and must be differentiated from deep venous insufficiency and lip edema. On inspection, foot edema on the dorsum (buffalo hump), sausage-like shape of fingers, edema at the posterior internal, and external malleolus are evident. Edema is pitting in stages I and II. It may or may not be pitting in stage III and it is non-pitting in stage IV (Table 2).Water displacement volume try, wasn’t commonly used; however, it measures leg volume [12] and is more accurate than calculating the leg volume by circumferential measurements by a tape measure [13]. We did not use lymphangiography, because it is technically demanding, painful, time-consuming, associated with an increased risk of hypersensitivity reactions, and emboli [14]. The ultrasound in lymphedema may show minimal thickness of the dermis to an increase in the subcutaneous layer [15, 16]. The duplex ultrasound may be a useful tool in patients with lower limb swelling [18]. In our study, Doppler ultrasound was used for two purposes namelya diagnosis guide and evaluation of the venous system. Computed tomography (CT) scanning can be used not only to confirm the diagnosis, but also to monitor the efficacy of treatment [17].Magnetic resonance imaging (MRI) can differentiate among lymphedema, lip edema, and phlebedema [18]. We did not use CT scan or MRI in our study. Treatment for lymphedema is conservative and surgical. The conservative treatment may contain diet (low salt and lipoprotein) and elastic stockings. Compression with custom-made elastic stockings (minimum pressure of 40 mm Hg) is an effective method, particularly for secondary lymphedema [19]. Another form of compression and massage comes from pneumatic pumps [20, 21]. All of these methods are employed to achieve optimal benefit [22, 23]. Pharmacological therapy (for example benzopyrones) is reported by some studies [24-30]. All patients should be admitted for a few days to allow leg elevation and compression to optimize the leg for surgery [31]. Some surgical procedures have been documented for management of lymphedema such as subcutaneous drainage of lymphedema [32], radical excision of subcutaneous tissue and skin graft in one [33] and two stages [34, 35]. Liposuction improves symptoms but leads to foot edema [36]. The Charge is done only for lymphedema stage 5, because this procedure does not have a cosmetically pleasing outcome.
| Grade I | Mild edema + pitting + disappearsafter 2-3 hours |
| Grade II | Moderate edema + pitting + disappearsafter 24 hours |
| Grade III | Sever edema + non pitting edemawithout skin reaction |
| Grade IV | Skin reaction: elephantiasis |
Liposuction has been used with up to 23% reduction in volume only [37]; severe cellulitis has also been reported as its main complication. Bypass procedures are only done in selective patients; all bypass procedures need microsurgery, and patency rate is not good. Surgical treatment is palliative, not curative, and after surgical therapy, continued medical therapy is required. Therefore, it is rarely indicated as the primary treatment. Surgical treatment is reserved for patients who do not improve with conservative measures or for cases in which the extremity is so large that it impairs daily activities; recurrent lymphangitic also prevents successful conservative management. As mentioned earlier, the main treatment strategy for lymphedema is non-operative. After at least six months of conservative management based on compression stockings with class III compression, if predominant swelling and disability remain, surgical therapy will be considered.
The role of surgery is more prominent when a primary chylous disorder is encountered [18]. Potential indications include impaired limb function, recurrent episodes of cellulitis and lymphangitis, intractable pain, angiosarcoma, and patient preference for cosmoses [19]. Physiologic operations have been aimed at restoring lymphatic transport capacity more frequently with lymphovenous anastomoses. Long-term patency rates have been reported but poor functional alleviation, limited experience, and expertise have made their -controversial use [19]. The best limb volume reduction is achieved by staged resection of the subcutaneous tissues but poor wound healing, long surgical scars, sensory nerve loss residual edema of the foot, and ankle are the main complications that limit its application [19]. Our study demonstrated that resection technique can be modified to a more cosmetic and volume reducing one with less disfiguring scars, better functional results, and minimal wound complications. More surveys with larger sample sizes and long-term follow-ups are required to confirm the priorities of our procedure and to compare it with other resection techniques.
CONCLUSION
This excisional method such as reducing the volume of proper and acceptable both in terms of cosmetic can be used as a new method for the surgical treatment of lymphedema.
LIST OF ABBREVIATIONS
| (CT) | = Computed tomography |
| (MRI) | = Magnetic resonance imaging |
DISCLOSURE
I must mention about a“Part of this article has been reproduced from the previous publication in “Surgical Practice, Volume 13, Issue 2, May 2009, Pages 48–52; DOI: 10.1111/j.1744-1633.2009.00435.x.” I had to act because the technique is the same.
CONFLICT OF INTEREST
The author confirms that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
I would like to thank the chief and professor of department of Vascular surgery of Shohada Medical Center, Shahid Beheshti University Medical Sciences, Mohammad Reza Kalanter Moatamedi for help me during long time study.


