All published articles of this journal are available on ScienceDirect.
Bacterial and Hematological Findings in Infected Total Hip Arthroplasties in Norway Assessment of 278 Revisions Due to Infection in the Norwegian Arthroplasty Register
Abstract
Our aim was to assess the bacterial findings in infected total hip arthroplasties (THAs) in Norway. We also wanted to investigate the relationship between causal bacteria and hematological findings. Revisions reported to the Norwegian Arthroplasty Register (NAR) due to infection after total hip arthroplasty during the period 1993 through September 2007 were identified. One single observer visited ten representative hospitals where clinical history, preoperative blood samples and the bacterial findings of intraoperative samples were collected. Bacterial growth in two or more samples was found in 278 revisions, and thus included. The following bacteria were identified: Coagulase-negative staphylococci (CoNS) (41%), Staphylococcus aureus (S. aureus) (19%), streptococci (11%), polymicrobial infections (10%), enterococci (9%), Gram-negative bacteria (6%) and others (4%). CoNS were the most common bacteria throughout the period but in the acute postoperative infections (< 3 weeks) S. aureus was the most frequent bacterial finding. We found no change in the distribution of the bacterial groups over time. S. aureus appears correlated with a higher C-reactive protein value (CRP) (mean 140 (95% Confidence interval (CI): 101-180)) than CoNS (mean 42 (CI: 31-53)). S. aureus also correlated with a higher erythrocyte sedimentation rate value (ESR) (mean 67 (CI: 55-79)) than CoNS (mean 47 (CI: 39-54)).
INTRODUCTION
Approximately 7,000 primary total hip arthroplasties (THA) are performed annually in Norwegian hospitals [1]. Infection after primary THA is a relatively rare event, and large numbers of patients are therefore needed to assess bacteriology and trends. In recent years there have been indications of an increasing risk for revision due to infection after THA [2-4], and the question has been raised as to whether this might be due to changes in bacteriology [5]. The bacteria most frequently causing prosthetic joint infections (PJI) are staphylococci (i.e. Coagulase negative staphylococci (CoNS) and Staphylococcus aureus (S. aureus)), and biofilm formation and emerging resistance towards antibiotics represent a challenge in PJI treatment [6-8]. However, many different bacterial species may cause PJIs and there has previously been no nationwide assessment of bacteria causing revisions of THAs in Norway. Identification and diagnostics of PJI may be challenging, and in addition to bacterial samples, C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) are important hematological markers in diagnostics [9].
Our aim was to investigate the bacterial findings in infected THAs in Norway by using the nationwide Norwegian Arthroplasty Register (NAR) to identify cases of revision due to infection, and then collect additional information on bacterial and hematological findings. We also wanted to assess whether CRP and ESR correlated with bacterial findings, and whether there were any time trends regarding the bacterial findings.
MATERIAL AND METHODS
The NAR has since its inception in 1987 registered data on both primary THAs and THA revisions in Norway [10], and the NAR data has been validated as being of good quality [11, 12]. The register form is filled in by the surgeon immediately after surgery, containing information on reason for surgery and different patient and surgery related variables. A unique identification number of the patient is used to link the primary THA surgery to later revisions. Since the form is filled in immediately after surgery in the case of a revision, the diagnosis of e.g. deep infection or aseptic loosening is based on clinical, biochemical and radiological findings pre- and intraoperatively. The cause of the revision is reported to the NAR before the results from the intraoperative bacterial samples are ready, and may not be corrected later.
From January 1993 through September 2007, 62 hospitals reported 1,089 revisions due to infection after THA to the NAR. These included revisions where parts of or the whole prosthesis was removed or replaced due to infection. Also cases of re-revision that showed different bacterial findings to the prior revision were included. We performed a pilot study at three large university hospitals, before extending the study to include the ten hospitals with the highest number of reported THA revisions due to infection during the period. The ten hospitals were geographically spread throughout the whole country and had similar rates of revision due to infection as the national average (0.6%) [5]. So of all THAs performed in Norway, approximately 0.6% are revised due to infection and reported to the NAR. The hospitals were visited by the first author and information on bacterial findings of intraoperative samples was collected from the patients’ medical records. Results from preoperative blood samples were not collected in the three hospitals in the pilot study, but were added from the seven hospitals in the main study. Preoperative CRP and ESR within three days before the revision due to infection were assessed and compared to the bacteriological findings.
For the revision to be included there had to be growth of the same bacteria in two or more periprosthetic tissue samples collected intraoperatively, according to the definition of PJI [13]. Preoperative joint aspirations were not included. On average five samples were taken. None of the removed parts were sonicated, as this has not been procedure in Norway. Polymicrobial infection was defined as infection with more than one species (at least two of each) in intraoperative samples.
The study period was divided into three 5-year time periods to assess for time trends. Time from index THA to subsequent revision due to infection was divided into acute postoperative (less than 3 weeks), early (3 weeks to 3 months), delayed (3 months to 2 years) and late infections (more than 2 years).
The ten hospitals reported 454 identifiable revisions due to infection to the NAR. 138 of these had no growth inintraoperative tissue samples and 38 of the revisions had growth in only one periprosthetic tissue sample and were therefore excluded (Fig. 1). Hence, 278 THAs reported to the NAR for revision due to infection and verified by bacterial cultures were included. Time from index THA to revision was addressed in only 227 of the cases, mainly due to missing registration of the index THA in the NAR.CRP and SR were addressed in 166 of the cases.
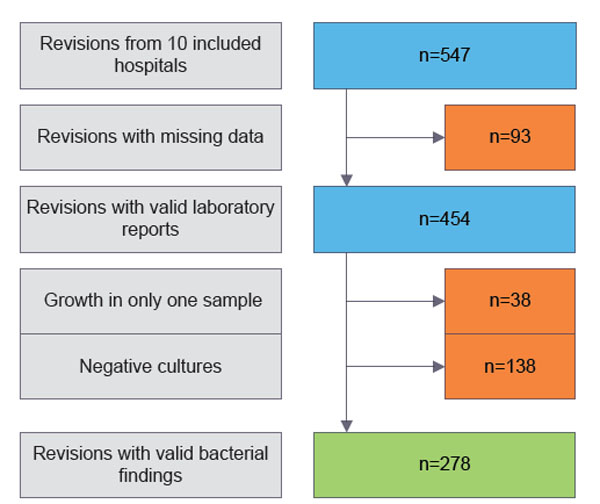
Flow chart showing the patient selection.
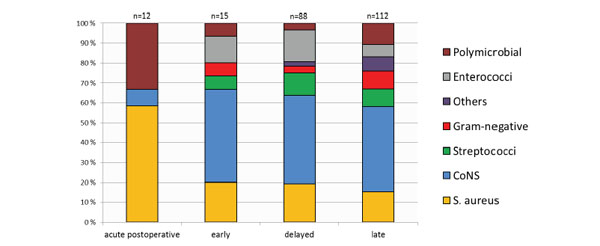
Bacterial findings relative to time after index THA surgery (n=227).
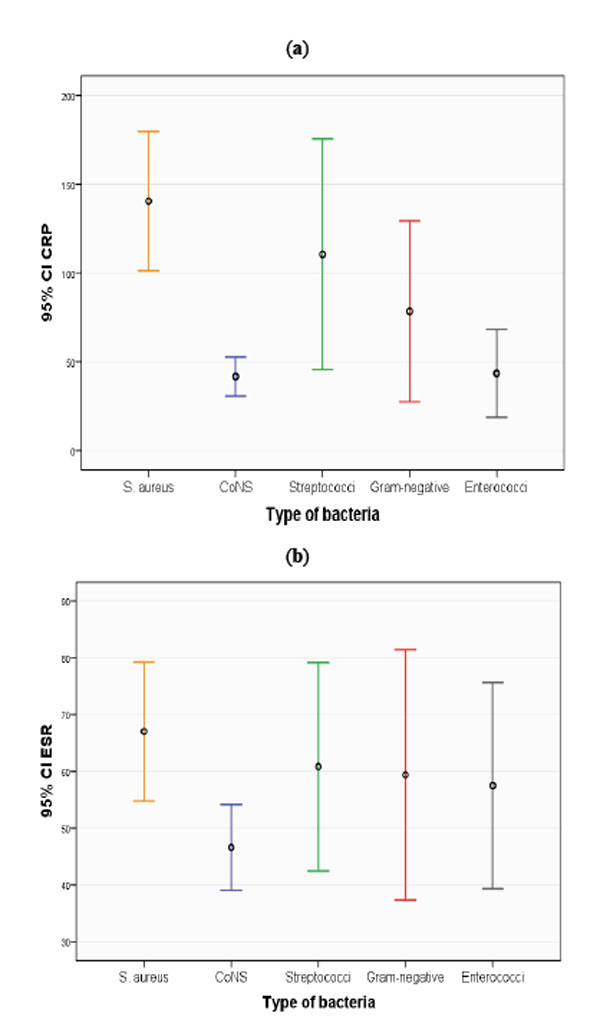
(a, b) Mean values of ESR and CRP level with 95% confidence intervals (CI) for different species of bacteria cultured after revision surgery.
Patient characteristics.
| Sex | |
| Male | 32.4% |
| Female | 67.6% |
| Age | |
| Mean | 69.4 |
| SD | 10.9 |
| Diagnosis | |
| Osteoarthritis | 67.3% |
| Inflammatory | 4.0% |
| Other | 28.7% |
| Antibiotic Prophylaxis Systemically | |
| Yes | 98.4% |
| No | 1.6% |
| Method of Fixation | |
| Uncemented | 11.1% |
| Cement | |
| With antibiotics | 72.2% |
| Without antibiotics | 16.7% |
Bacterial findings throughout the 15-year period (n=278).
| Frequency | Percent | |
|---|---|---|
| S. aureus | 53 | 19% |
| CoNS | 113 | 41% |
| Streptococci | 30 | 11% |
| Gram-negative | 17 | 6% |
| Others | 12 | 4% |
| Enterococci | 26 | 9% |
| Polymicrobial | 27 | 10% |
CoNS subspecies in the bacterial findings (n=113).
| Frequency | |
|---|---|
| S. epidermidis | 43 |
| S. capitis | 3 |
| S. lugdunensis | 3 |
| S. warneri | 1 |
| S. simulans | 1 |
| Unspecified CoNS | 46 |
| S. epidermidis and unspecified CoNS | 12 |
| S. epidermidis, S. xylos and S. lentus | 1 |
| S. epidermidis and S. hominis | 1 |
| S. haemolyticus and S. capitis | 1 |
Bacterial findings in cases with mixed bacteriology (n=27).
| Frequency | |
|---|---|
| CoNS and corynebacteria | 6 |
| CoNS and S. aureus | 4 |
| CoNS and Gram-negative | 4 |
| Enterococci and Gram-negative | 4 |
| S. aureus and streptococci | 2 |
| S. aureus and enterococci | 2 |
| CoNS and enterococci | 1 |
| CoNS, S. aureus and enterococci | 1 |
| CoNS, S. aureus and streptococci | 1 |
| Enterococci and peptostreptococci | 1 |
| CoNS, Gram-negative, enterococci and streptococci | 1 |
The patient characteristics are presented in Table 1 and shows that 98.4% of the patients are reported to have received systemic, antibiotic prophylaxis. From 2003 the prophylaxis has consisted mainly of Cephalotin according to national guidelines. This is supported by a study from the NAR also showing a reduction in the use of β-lactamase resistant penicillin as prophylaxis after 1996 [14].
Pearson’s chi-squared test and linear-by-linear association were used to compare time periods and groups of bacteria. P-values less than 0.05 were considered significant. Statistical analyses were performed using SPSS version 22 (SPSS Inc., 2004). The study was approved by the Regional Committee for Medical Research Ethics (number 2009/856b).
RESULTS
Bacteriology of THA Infection
The distribution of microbes is presented in Table 2. Staphylococci (60%) were the most common, followed by streptococci (11%) and enterococci (9%). 10% of the infections were polymicrobial. Table 3 presents the different CoNS species with S. epidermidis being the most common. Among the polymicrobial infections, staphylococci were involved in all except four. The combination of CoNS and corynebacteria was most common. Table 4 shows the combinations of bacteria for polymicrobial THA infections.
Bacterial Findings and Time After Index THA
Bacterial findings relative to time after index THA are presented in Fig. (2). The first 3 weeks all infections were either with Staphylococci or polymicrobial, and S. aureus was the most frequent bacteria. Later on the bacterial findings were more diverse.
Bacterial Findings, CRP and ESR
S. aureus infections were associated with higher CRP (mean 140 (95% Confidence interval (CI): 101-180)) than infections caused by CoNS (mean 42 (CI: 31-53)) (Fig. 3a). This was also found for ESR when S. aureus infections (mean 67 (CI: 55-79)) were compared to CoNS infections (mean 47 (CI: 39-54)) (Fig. 3b). S. aureus infections were also associated with higher CRP than infections caused by enterococci (mean 43 (CI: 19-68)).
Time Trend of Bacterial Findings
The incidence of the different bacteria was mostly unchanged throughout the study period, but there was a trend towards more polymicrobial infection (p=0.1).
DISCUSSION
CoNS were the most common bacteria causing revision due to deep infection after THA. Since CoNS are regarded as commensal bacteria, this may support the idea that direct contamination is the most common mechanism of THA infection [15-17]. Our bacterial findings were similar to those in other publications [18-28], although the share of S. aureus was somewhat lower than in some of the publications.
S. aureus was the most frequent bacterium in acute postoperative infections, whereas CoNS were the most common cause of revision due to infection in early, delayed and late infections. As late infection is most prevalent in our study, it may be that suppressed and biofilm embedded CoNS infection can emerge even after 2 years postoperatively, or that CoNS are spread through hematogenous seeding.
S. aureus infections had higher CRP and ESR values than CoNS infections. Given high resistance to methicillin and aminoglycosides amongst CoNS [29], in empirical treatment of infected THA with low CRP (<50) in Norway, Vancomycin should be considered until results of intraoperative cultures are known.
The incidence of the different bacteria was mostly unchanged throughout the study period. There was a trend towards more polymicrobial infection and the combination of CoNS and corynebacteria was most common. This may be explained by better sample handling, better culturing methods and increased attention to pathogens formerly considered as contamination by commensal bacteria, not able to cause infection (e.g. corynebacteria).
The retrospective nature of this study represents one of the major limitations of this paper. Another weakness of our study is that only revisions with removal or change of the whole or parts of the prosthesis are reported to the NAR. Accordingly, soft tissue debridement of infected THAs without exchange of prosthesis parts is not reported to the register. Consequently not all surgical site infections after THA are reported. This may have affected the distribution of the bacterial findings since surgical site infection is an early postoperative event [30], and it may have led to an underestimation of e.g.S. aureus in our study. Further, low grade infections with low or normal CRP or ESR may be evaluated by the reporting surgeon as an aseptic loosening and hence erroneously reported to the NAR as such. These low grade infections, not identified in preoperative diagnostics, will not be included in our study, and the prevalence of low-virulent bacteria such as CoNS may be underestimated.
CONCLUSION
In this nationwide study, based on 278 revisions of infected THA, staphylococci were the most common bacteria in THA revision for infection in Norway. S. aureus was more common in acute postoperative infections and CoNS were more common in early, delayed and late infections. CRP and ESR may be of help in differentiating between infections caused by CoNS and S. aureus.
CONFLICT OF INTEREST
The first author received a scholarship from OrtoMedic for his work on this study. We report no conflict of interest.
ACKNOWLEDGEMENTS
HL collected the data. HL, OL and BE performed the analyses. HL wrote the manuscript. All authors contributed in interpretation of the analyses and critical revision of the manuscript. We would like to thank the NAR contacts at all ten hospitals for contributing to data collection. We also wish to thank Norwegian surgeons for persistently and thoroughly reporting primary THAs and revisions to the NAR.


