All published articles of this journal are available on ScienceDirect.
Concepts and Potential Future Developments for Treatment of Periprosthetic Proximal Femoral Fractures
Abstract
Periprosthetic proximal femoral fractures are a major challenge for the orthopaedic surgeon, with a continuously increasing incidence due to aging populations and concordantly increasing numbers of total hip replacements. Surgical decision-making mainly depends on the stability of the arthroplasty, and the quality of bone stock. As patients final outcomes mainly depend on early mobilization, a high primary stability of the construct is of particular relevance. Osteosynthetic procedures are usually applied for fractures with a stable arthroplasty, while fractures with a loosened endoprosthesis commonly require revision arthroplasty. Osteoporotic bone with insufficient anchoring substance for screws poses one major concern for cases with well-fixed arthroplasties. Complication rates and perioperative mortality have remained unacceptably high, emphasizing the need for new innovations in the treatment of periprosthetic fractures. Transprosthetic drilling of screws through the hip stem as the most solid and reliable part in the patient might represent a promising future approach, with auspicious results in recent biomechanical studies.
EPIDEMIOLOGY AND BACKGROUND
With the ongoing aging in today’s modern population, the number of implanted primary hip arthroplasties continues to increase. Following this increase, complications such as periprosthetic fractures are also occuring more frequently and remain a challenge for orthopedic surgeons.
Today, up to 4% of all patients with implanted primary hip arthroplasties will suffer from a periprosthetic fracture at least once in their life [1-3], most commonly caused by a fall on the implanted hip. Mean time from primary total hip arthroplasty to a periprosthetic fracture has been reported as 7.4 years [4] and 8.1 years [5], respectively. Katz et al. reported in their cohort study that periprosthetic fractures occur annually in 26 of 10,000 persons a decade after implantation [2].
Main risk factors associated with periprosthetic fractures are implant loosening, osteoporotic bone, and increasing age [4, 6]. More rare risk factors include rheumatoid arthritis, Paget’s disease, tumors, polyneuropathies, extruded cement, and varus stem position [7]. Average age of patients is commonly reported around their late 70ies [8-10]
The surgical management of fracture patterns and the stability vs instability of the primarily implanted endopros-thesis in periprosthetic fractures still are challenging for the orthopedic surgeon. Until today, levels of peri- and postoperative complications have remained high and long-term functional outcomes are still unsatisfying [5, 11, 12-15]. Likely due to reduced biological capacities of the elderly patients, rates of infections and nonunions are significantly higher than in cohorts with primary arthroplasties [10]. A recent study is given on 58 patients sustaining periprosthetic femoral fractures after hip arthroplasty with a follow-up of 34 months reported good or excellent radiological results in 71% of the patients, but also a complication rate of 56%, and a revision surgery rate of 19%. As probably the most important outcome parameter, 52% of the patients did not regain their per-injury walking status [9]. Overall mortality rates related to periprosthetic fractures have been reported to be similar to those of proximal femur fractures, Bhattacharyya et al. reported mortality rates of up to 11% of patients suffering from a periprosthetic fracture [16-19].
CLASSIFICATION OF PERIPROSTHETIC FRACTURES
A variety of classification systems have been suggested to stratify periprosthetic fractures [20]. The Vancouver classification system (VCS) [21] has been proven most useful and is widely in used (Table 1).
The Vancouver classification system provides information about the fracture pattern in relation to the prosthesis, the stability of the prosthesis, and quality of bone stock. Therefore, this system also provides help for surgical therapeutic decision making [20, 22].
As an expansion of the VCS, the so called Unified Classification System (UCS) has recently been suggested for peri-prosthetic fractures of the pelvis and femur around a total hip replacement [23]. The purpose of the UCS is to expand the VCS to include three more fracture types, to apply it more generally to all bones and joints in a principles-based manner, and to introduce a common language assisting with evaluation, treatment, and outcomes measurement of periprosthetic fractures (Table 2).
Even though excellent intra- and interobserver reliabilities have been reported [23], the future role of this new classification systems still has to be evaluated. The advantage of offering a comprehensive system of periprosthetic fractures goes along with the disadvantage of numerous subgroups, which may impede its daily clinical use.
THERAPEUTIC STRATEGIES
Treatment of periprosthetic fractures mainly depends on the stability of the arthroplasty in situ. Osteosynthetic procedures are usually applied for fractures with a stable arthroplasty, while fractures with a loosened endoprosthesis commonly require revision arthroplasty. The therapeutic strategy always has to be correlated to the fracture location and bone quality. The aim is to re-establish the correct length, axis, bone substance, and therewith the patient´s mobility as fast as possible via a stable osteosynthesis or revision arthroplasty [24].
A variety of surgical treatment options are available. The main concern is the thorough evaluation of the stability of the arthroplasty, with no reproducible and reliable tests available. As a general principle, revision arthroplasty should be performed for cementless arthroplasties if more than one third of the bony anchoring zone is affected by the fracture [22, 25]. For cemented arthroplasties, the indication for revision arthroplasty is even wider due to the potential complete disruption of the bone-cement-inferface by the fracture [22].
Vancouver A Fractures
Vancouver A fractures can be treated conservatively in case of minor dislocations (<2,5cm) and in absence of gluteal insufficiency (Fig. 1) [26]. For more severe dislocations (>2,5cm) or in case of osteolytic destructions, surgical reconstruction by autologous cancellous bone grafting and osteosynthesis with tension-band-wiring, cerclage, or hook plate techniques, is usually performed (Fig. 2) [22, 27].
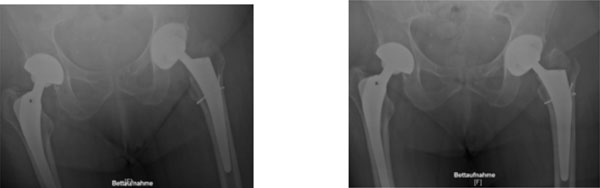
Vancouver AG (greater trochanter) fracture of the left hip with conservative management.
Vancouver AG (greater trochanter) fracture of the left hip, managed with cerclage and k-wires.
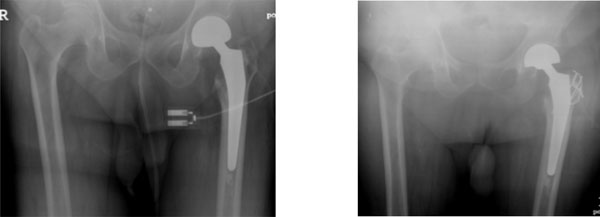
Vancouver B1 fracture of the left hip, managed with locking plate osteosynthesis.
Vancouver B2 fracture of the right hip, managed with a modular diaphyseally anchoring revision arthroplasty.
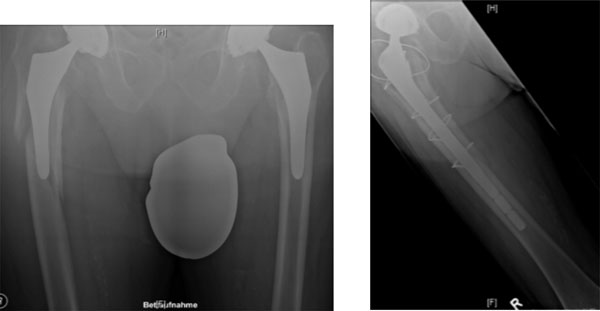
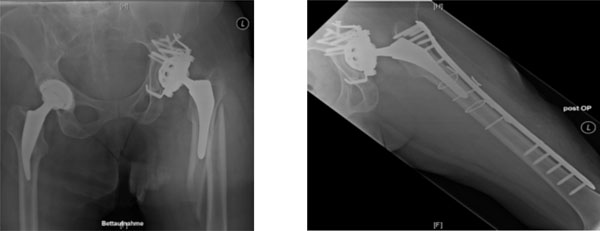
Vancouver B3 fracture of the right hip with osseous defect, managed with a modular revision arthroplasty and cerclages.
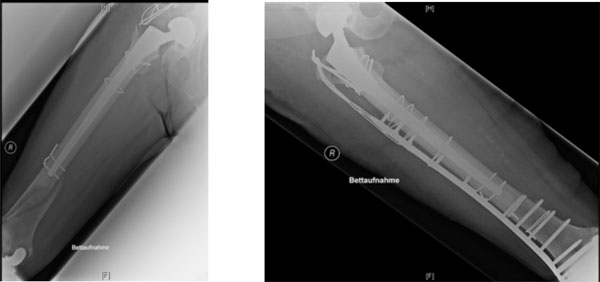
Vancouver C fracture of the right hip, managed with arthroplasty-retaining locking plate osteosynthesis.

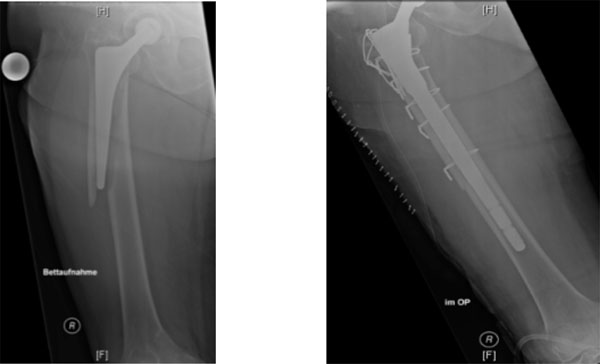
Specially designed drilling machine for intraprosthetic screw fixation.
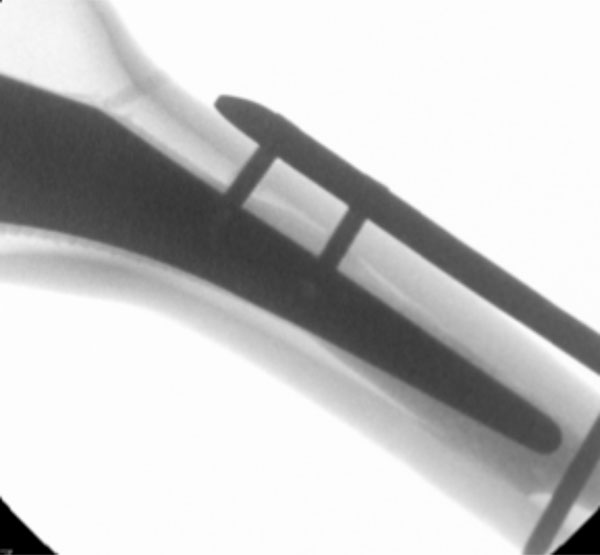
X-Ray demonstration of intraprosthetic screw fixation [42].
Vancouver classification system (VCS) of periprosthetic fractures.
| A | trochanteric region | A G: greater trochanter |
| A L: lesser trochanter | ||
| B | diaphysis | B1: stable arthroplasty |
| B2: unstable arthroplasty | ||
| B3: B2 with osseous defect | ||
| C | distal of arthroplasty |
The unified classification system (UCS) of periprosthetic fractures [23].
| Type | General Description | Description for the Femur | Description for the Pelvis |
|---|---|---|---|
| A | Apophyseal | greater or lesser trochanter | anterior superior or inferior iliac spine, or ischial tuberosity |
| B | Bed of the implant or close to it | around or close to the femoral stem (stem well fixed if B1, stem loose if B2, loose and poor bone if B3 | acetabular fracture: B1: fracture of the acetabular lip, wall or floor, which does not affect the stability of the component B2: loose acetabular component but adequate bone stock to support an uncomplicated acetabular component revision B3: loose component, and severe bone loss such that complex reconstruction or a salvage procedure is required |
| C | Clear of the implant bed | the femur distant from the implant | ilium, superior and/or infe- rior rami |
| D | Dividing one bone which supports two joint replacements | between a hip and knee replacement | a fracture of the pelvis complicating bilateral replacements |
| E | Each of two bones supporting one joint replacement | both the femur and acetabulum | both the acetabulum and femur |
| F | Facing or articulating with an implant | not applicable for the femur | fracture of the acetabulum after hemiarthroplasty |
Vancouver B Fractures
Vancouver B fractures account for the vast majority of periprosthetic proximal femur fractures. For Vancouver B1 fractures (stable primary implant), arthroplasty-retaining osteosynthesis usually is the treatment method of choice (Fig. 3) [24, 28, 29].
Locking plate systems such as the LISS (Less Invasive Stabilization System, Synthes, Zuchwil, Switzerland), or polyaxial locking plate systems such as NCB (Non-Contact Bridging, Zimmer Inc., Warsaw, IN, USA) are commonly used for this kind of management.
Sufficient bony anchoring of screws in the osteoporotic bone in the area of the arthroplasty poses a major challenge to the orthopedic surgeon [22].
A biomechanical sawbone study by Zdero et al. comparing osteosynthetic treatment options for Vancouver B1 and C periprosthetic fractures demonstrated that a combination of a nonlocking plate with an allograft strut resulted in the highest stiffness of the construct compared to unlocked and locking plates (with or without addtional cables) without strut augmentation [30].
In a human cadaveric study, Wähnert et al. compared the fixed angle locking attachment plate (LAP®, Depuy Synthes®, Solothurn, Switzerland) to the variable angle non-contact bridging plate (NCB®, Zimmer GmbH, Winterthur, Switzerland) in a Vancoucer B1 fracture model. The non-contact bridging plate system yielded significantly higher failure loads compared to the fixed angle locking plate in their study [31].
To further compare, 3 different options of proximal screw fixation using the non-contact bridging periprosthetic proximal femur plate (Zimmer Inc., Warsaw, IN) in a Vancouver B1 sawbone model, Hoffmann et al. demonstrated that bicortical screw placement achieved the highest load of failure and the highest bending stiffness compared to unicortical screws plus one cerclage cable, or three cerclage cables [32]. This was confirmed by the human cadaveric study from Lenz et al., who similarly found that bicortical screw anchorage provided the best proximal plate fixation in Vancouver B1 periprosthetic fractures. The authors also found that the cerclage-screw combination was a valuable alternative in osteoporotic bone [33]. These biomechanical findings are consistent with clinical experience, while however finding sufficient bony anchoring of screws in the osteoporotic bone in the area of the arthroplasty is one of the biggest challenges in the clinical setting.
A recent systematic review on 333 patients with Vancouver B1 periprosthetic femur fractures identified a total complication rate of 15%, with a revision surgery rate of 9%. Of interest, locking plates had higher rates of nonunion (9% vs 3%), and hardware failure (7% vs 2%) compared to cable plate/compression plate systems [34]. However, these findings need to be interpreted with caution due to lack of high-evidence studies and potential methodological flaws.
As a biological augmentation device in order to enhance healing, application of allograft struts has been suggested as an adjunct to osteosynthesis in periprosthetic fractures [35, 36].
Khashan et al. reported on 21 patients with Vancouver type B1 or C fractures treated either by plate fixation and a strut allograft or by plate fixation alone. They found similar times to fracture union (around 12 weeks in both groups), but a significantly higher failure and revision surgery in the plate alone group (45%) compared to the group with additional allograft strut (0%) [8].
On the other hand, a recent systematic review of 37 manuscripts with 682 Vancouver B1 fractures revealed an increased infection rate for patients operated with allograft augmentation (8.3 vs 3.8%) and time to union (6.6 months vs 4.4 months) compared to those operated without allograft. Union rates were similar for patients treated with or without an allograft strut (90.7% vs 91.5%). Of interest, union rate and time to union were found to be independent of the applied osteosynthesis technique [37]. In the light of the findings of this systematic review, allografts have to be considered with caution for repair augmentation in periprosthetic fractures.
Different to B1, Vancouver B2 and B3 fractures are mainly determined to revision arthroplasty. The key issue for these cases is a complete removal of the entire implant with its surrounding cement coat, followed by stable bridging of the fracture with a diaphyseally anchoring long-stemmed revision arthroplasty (Figs. 4, 5) [38].
The choice of revision arthroplasty depends on the patients biology and expectations as well as the surgeons skill and preference.
Cementless modular implants with diaphyseal anchoring are a good option to achieve an optimal restitution of length, soft-tissue lever arms, and femoral offset, with convenient adjustability by their modular structure.
Cemented modular devices allow for early weight bearing and are a good option especially for elderly patients with osteoporotic bone.
As an alternative for patients who are not fit for prolonged surgical procedures, the so called Exeter technique, retaining a well-fixed cement mantle and performing a cement-in-cement revision arthroplasty, offers the advantages of a simplified operative technique, decreased operating time, and faster recovery from surgery [39].
Vancouver C Fractures
On the other hand, Vancouver C fractures usually contain a stable primary implant and are therefore a domain of arthroplasty-retaining osteosynthesic management (Fig. 6) [40, 41]. Most commonly, long-distance bridging locking plates (such as LISS-PT (Synthes, Zuchwil, Switzerland) or polyaxial locking plate systems such as NCB (Non-Contact Bridging, Zimmer Inc., Warsaw, IN, USA)) and cerclage osteosynthetic devices are applied. Similar to the problem in Vancouver type B fractures, sufficient bony anchoring of the locking screws in order to achieve sufficient primary stability at the level of the arthroplasty remains the major concern. This is of particular interest in osteoporotic bone, which usually does not offer a sufficient bony anchoring substance [22].
POTENTIAL FOR FUTURE DEVELOPMENTS IN VANCOUVER B1 AND C FRACTURES
As a particular problem in elderly patients with pronounced osteoporosis sustaining Vancouver B1 and C fractures with a stable arthroplasty, the surgeon often encounters a thin, eggshell-like bone layer at the level of the arthroplasty. This only allows for inadequate anchoring of screws and therefore increases the risk of postoperative implant failure. With such thin bone layers, the screws have to be drilled meticulously right adjacent to the arthroplasty, or can only be drilled monocortically. This often impedes the concept of locking screws with a predetermined angulation between the screws and the plate. Adequate and sufficient mechanical fixation imperatively need to be achieved in these patients to allow for early functional recovery and return to pre-injury independence [7].
This arises the question whether high primary stability of the repair can be achieved via a special drilling device allowing to drill screws through the arthroplasty, as the most solid and reliable component in the patient (Fig. 7).
The idea of this concept is to achieve both centric drilling and longer anchoring distances, as well as maintenance of locking screw stability (Fig. 8).
Drilling through solid materials such as hip stems creates substantial temperatures. It has previously been shown that osteonecrosis and further tissue damage is already caused by temperatures of 47° C with a residence time of 30 seconds [43, 44].
As recently investigated, transprosthetic drilling without cooling leads to an immediate increase in temperature to over 200 °C. Therefore, a device to ensure cooling throughout the drilling procedure is required to allow for tissue-preserving transprosthetic drilling. Using a custom-made internal cooling device within the power drill setup reduced temperatures during drilling to tissue-preserving levels for cemented hip stems (24.7 °C) [45]. However, the same research group also demonstrated that for cementless hip stems without the insulating cement layer, the temperature during drilling increased to 47 °C, even when the internal cooling device was applied [46]. These studies indicate a potential solution for the concern of tissue-preservation during transprosthetic drilling, with however some further research being required especially for cementless hip stems.
Brand and coworkers demonstrated superior biomechanical properties for intraprosthetic screw anchorage compared to conventional monocortical locking screw fixation in both a Vancouver B1 fracture model with a cemented straight hip stem [42] and a Vancouver C fracture model [47]. This approach might represent a potential solution for osteosynthetic prodecures for patients with periprosthetic fractures with well-fixed implants, providing high primary stability and the potential of early full weight-bearing mobilization. Future clinical trials need to investigate this novel method.
CONCLUSION
Surgical decision-making for periprosthetic proximal femoral fractures mainly depends on the stability of the arthroplasty, and the quality of bone stock. As patients final outcomes mainly depend on early mobilization, a high primary stability of the construct is of particular relevance. Osteoporotic bone with insufficient anchoring substance for screws poses one major concern for cases with well-fixed arthroplasties. Transprosthetic drilling of screws through the hip stem as the most solid and reliable part in the patient might represent a promising future approach, with auspicious results in recent biomechanical studies.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
Declared none.


