All published articles of this journal are available on ScienceDirect.
The Evidence-Based Principles of Negative Pressure Wound Therapy in Trauma & Orthopedics
Abstract
Negative pressure wound therapy is a popular treatment for the management of both acute and chronic wounds. Its use in trauma and orthopedics is diverse and includes the acute traumatic setting as well as chronic troublesome wounds associated with pressure sores and diabetic foot surgery. Efforts have been made to provide an evidence base to guide its use however this has been limited by a lack of good quality evidence. The following review article explores the available evidence and describes future developments for its use in trauma and orthopaedic practice.
INTRODUCTION
Negative pressure wound therapy (NPWT) is a popular treatment for the management of both acute and chronic wounds. Its use is widespread amongst surgical specialties many of which employ NPWT to varying degrees as part of their armamentarium against challenging wounds. Its use in orthopedics is diverse and includes the acute traumatic setting as well as chronic troublesome wounds associated with pressure sores and diabetic foot surgery.
NPWT is commonly used in place of more traditional dressing techniques using cotton gauze. Whilst cotton gauze is an inexpensive product which is able to keep wounds clean and covered whilst absorbing exudate, many clinicians feel that NPWT is a more dynamic alternative that reduces infection and promotes early closure. Despite this growing popularity there is a paucity of evidence behind its use [1]. Contributing to this is the fact that wound healing is a complex process affected by both local and systemic factors. Identifying the effect of NPWT amidst so many potential confounding factors is not without its difficulties.
There is a definite need for safe and effective wound management treatments. Injuries and wounds, especially when open, put a significant strain on both the patient and clinician alike. In the USA the market of advanced wound care is worth about $4.4 billion and is expected to continue to rise [2]. Evidently improvements in wound healing, while offering health benefits to the patient, would offer significant financial incentives to healthcare providers.
This review of the evidence based principles aims to review the extent to which NPWT has contributed to improvements in the management of wounds in the trauma and orthopaedic setting. A particular emphasis has been placed on the evidence supporting its use and its overall cost-effectiveness.
BACKGROUND
NPWT was first promoted in 1989 by Charker et al. [3] who described a nascent suction drainage system for the management of incisional and cutaneous fistulae. The system they described was different to today’s devices as it used a gauze-filled dressing connected to walled suction at pressures of 60-80 mmHg. They believed that their system was effective in promoting fluid drainage, helping the formation of granulation tissue and reducing skin damage [3]. As their system required fewer dressing changes compared to traditional methods they concluded it would be more cost-effective in the long-term [3].
In 1993, Fleischmann et al [4], described a more familiar version of NPWT using a polyurethane sponge. They noted that such a method had a positive effect on granulation tissue in the management of open fractures [4]. Most of the devices currently on the market contain a similar open pore polyurethane dressing.
The use of an open pore polyurethane dressing exposed to a subatmospheric pressure (125 mmHg below ambient pressure) was promoted by Argenta et al. [5] and forms the basis of todays contemporary dressings. They subjected 300 wounds of varying chronicity to their ‘vacuum-assisted closure’ technique until the wounds were either completely closed or covered by a split thickness skin graft or local flap [5]. They reported that 296 of the wounds responded favourably stating that the technique led to the removal of chronic oedema, increased blood flow and increased granulation tissue [5].
MECHANISM OF ACTION
NPWT has evolved from the basic principles of wound healing [6]. The initial phase of wound healing involves both haemostasis and inflammation. During this period local vasoconstriction occurs and platelets collect at the damaged endothelium. Following a brief period of vasoconstriction the local vasculature dilates to allow an influx of polymorphonuclear leukocytes and other blood cells to the site of injury. This process is mediated by a host of growth factors and cytokines. The inflammatory phase lasts approximately 4-6 days and is followed by the proliferative phase lasting for around 21 days. This phase is dominated by fibroblast activity and is characterized by the formation of granulation tissue, neo-angiogenesis and re-epithelialisation. The final stage in wound healing is tissue remodeling and involves the renewal of collagen fibres and contraction of the wound through the activity of myofibroblasts.
Local factors at the wound bed can have a negative effect on the wound healing process 7. The presence of infection, oedema, high flowing exudates [7] and ischaemia can delay the healing process. Using negative pressure on the wound is thought to reduce these negative effects by promoting a lower bacterial count [4], increasing vascularity and cell proliferation [8] as well as promoting removal of exudate from the wound, promoting granulation tissue and encouraging the wound edges to come together [7].
Orgill et al. [9] described four primary effects of NPWT on wound healing:
- Macro-deformation - drawing the wound edges together leading to contraction.
- Stabilisation of the wound environment - ensuring it is protected from outside microorganisms in a warm and moist environment.
- Reduced oedema - with removal of soft tissue exudates.
- Micro-deformation- leading to cellular proliferation on the wound surface.
Multiple secondary effects were noted to result from this including cell proliferation, increased blood flow and angiogenesis, reduction of inflammation, granulomatous tissue formation and the possibility of a decrease in bacterial load at the wound bed [7].
Two main theories prevail regarding the mechanism of action of NPWT used in conjunction with a reticulated open cell foam [10]. The first is based on the theory that tissue strain caused by NPWT has a stimulatory effect on cellular proliferation. This theory is supported by the fact that tissues have been shown to undergo a 5-20% strain when subjected to NPWT [11]. This level of strain is hypothesized to proactively cause cell division and angiogenesis in a process analogous to Ilizarovian distraction [8].
The second theory is focused on the effects of NPWT on the mechanical evacuation of excessive interstitial fluid and oedema [10]. By actively removing fluid at the wound bed it is felt that the local micro-circulation is improved and secondary necrosis is reduced. This hypothesis is supported by studies which have shown reduced need for debridement at the time of ‘second look’ for wounds treated with NPWT [12]. It is also supported by in vivo studies using a porcine model which have demonstrated acceleration in capillary formation and increase in luminal area in wounds treated with NPWT [13].
DEVICES & COMPONENTS
There are many NPWT devices currently in circulation. Arguably the most well-known is the V.A.C.® Therapy System (KCI, San Antonio, Texas). Most of the published literature refers to this model and its name has become synonymous with NPWT.
Regardless of the manufacturer, the fundamental components of the different devices are comparable.
Foam Dressing
The reticulated open pore sponges are commonly made from polyurethane or polyvinyl alcohol. These are easily cut and can be shaped to fit the wound bed closely. Their open pore structure enables negative pressure to be transferred across the entire sponge. As such an even suction is transferred across the sponge regardless of where the source is located. The pore size has been developed to maximize tissue regrowth and is generally sized to around 400-600 um [14]. In wounds with exposed tendons, ligaments or nerves many manufacturers recommend a smaller pore size in order to limit tissue ingrowth and reduce the pain and bleeding associated with dressing changes.
Dressing and Tube
Adhesive dressings are required to seal the wound and allow the negative pressure to create an effective vacuum with minimal leak. The dressing is usually made of polyurethane and can be cut to size to contour the local anatomy. Its occlusive properties not only enable a vacuum to be generated but also prevent external contaminants from fouling the wound. Many traditional gauze dressings are less able to do this. Some studies have suggested a benefit to the use of idophore-alcohol adhesive dressings which may both stick better to the skin and also help prevent bacterial colonisation of the skin [15], however, this is not common-place in NPWT. The suction tube is connected by cutting an opening into the surface of the adhesive tape to expose the sponge before being sealed onto the opening.
Negative Pressure Source
The suction tube is connected to a negative pressure source or unit. These units may require an external power supply but there are many models which are portable and run on rechargeable batteries (see Fig. 1-2). They usually generate negative pressures of around -125 mmHg. Early studies showed that applying this amount of pressure to a wound bed had the greatest effect on tissue regrowth and granulation formation [15]. This improved further with the cyclical method that is most often used today. Manufacturers have tried to develop treatment regimes in an attempt to tailor NPWT to the type of wound that is being dressed (see Table 1). Whilst there is some evidence behind these regimens [16-18] they are by no means definitive.
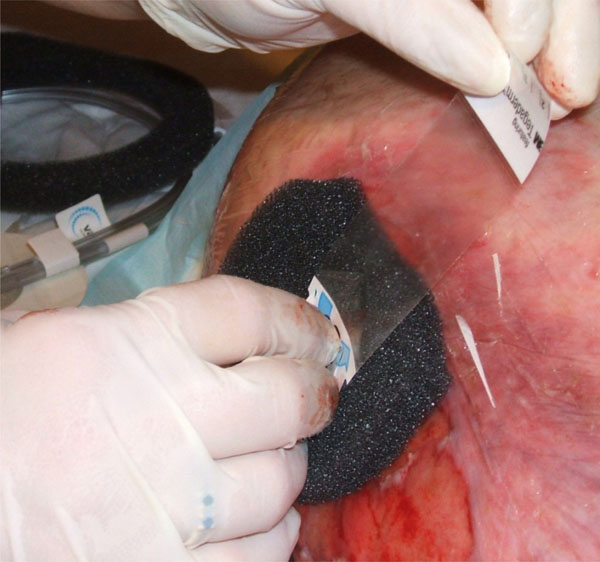
Foam and adhesive dressing [33].

Pressure source and canister [34].
Different negative pressure regimes [20].
| Type of Wound | Initial Cycle | Following Cycle | Target Pressure Granufo V.A.C. ® Granufoam Dressing | Target Pressure V.A.C. ® WhiteFoam Dressing | Dressing Change Interval |
|---|---|---|---|---|---|
| Acute/traumatic wounds/partial thickness burns | Continuous 48 hours | Consider intermittent 5min/2min cycles | 125 mmHg | 125-175 mmHg titrating upwards for increased drainage | 48-72 hours and more than 3 times per week, more if infected. |
| Lower extremity vascular surgical wounds | Continous | Consider Continuous | 125 mmHg can be reduced for patient comfort of condition of wound. | 125 mmHg | 48-72 hours, more than 3 times per week. |
| Dehisced wounds | Continuous | Continuous | 125 mmHg | 125-175 mmHg | 48-72 hours, more than 3 times per week |
| Meshed grafts and bioengineered tissue | Continuous | Continuous | 75-125 mmHg | 124 mmHg titrate up for more drainage | Remove dressing after 4-5 days |
| Pressure ulcers | Continuous 48 hours | Consider intermittent 5min/2min cycles | 125 mmHg | 125-175 mmHg titrate up for more drainage | 48-72 hours, more if infected. |
| Diabetic foot ulcer | Continuous 48 hours | Consider intermittent 5min/2min cycles | 50-125 mmHg (higher range preferred but limited by intolerance) | 125-175 mmHg titrate up for increased drainage. | 48-72 hours, more than 3 times per week. |
| Flaps | Continuous | Continuous | 125-150 mmHg | 125-175 mmHg titrate up for increased drainage | Remove dressing 72 hours post op. |
Canisters
These are usually attached to the negative pressure source and are required to collect the exudate that is drawn from the wound bed. The portable pressure sources usually have smaller canisters and as such may not be suitable for large wounds with a heavy exudate.
APPLICATION
NPWT dressings are very adaptable and can be contoured to fit wounds of varying shape, size and location. The indications for use are diverse and include acute, subacute, chronic, traumatic and dehisced wounds, partial thickness burns, ulcers (pressure, venous, diabetic), flaps and grafts. The first step is to cut the foam dressing to the shape of the wound bed. A simple method for doing this is to press the sponge against the wound and then use scissors to cut round the imprint made on the sponge by the raw wound surface. The sponge is then placed into the wound and secured below the adhesive clear dressing.
The adhesive dressings must form a complete seal around the wound margin in order to enable a vacuum to be created. The use of Compound Benzoin TinctureTM has been suggested to try and augment the quality of the seal created [19]. The mechanism by which the suction tube is connected to the adhesive dressing varies amongst manufacturers but commonly involves making a small hole in the dressing to expose the sponge and securing the tube over this hole with its own adhesive dressing. The non collapsible tube is then connected to the negative pressure source and collection system. Dressings will commonly need to be changed every 2-3 days. Different regimes described in the literature are summarized in Table 1.
CONTRAINDICATIONS
NPWT is generally considered safe and effective for a diverse range of wounds. However, there are certain situations for which NPWT is not recommended. Many of these are highlighted by the manufacturer’s themselves. It is essential that any clinician involved in the application of NPWT devices is familiar with these contra-indications [20]:
- Contact - NPWT foam dressing should not be in direct contact with exposed blood vessels, nerves, organs or anastomotic sites. There is an increase risk of fistulae formation in the presence of exposed organs, or hemorrhage with exposed blood vessels.
- Infection - Infections (including osteomyelitis) should be treated or debrided fully before the application of NPWT.
- Bleeding - Bleeding should be well controlled prior to application of NPWT. If bleeding occurs then the negative pressure should be disconnected and the dressing switched to a compression type.
- Malignancy - NPWT should not be used in the presence of malignant tissue. Its stimulatory effect on tissue growth is undesirable in the context of malignancy. Furthermore, malignant tissue is to prone to hemorrhage due to its disorganized vasculature.
- Allergy - To adhesive dressing or silver (for silver based foam).
- Other - Ischaemic wounds, fragile skin, non enteric and unexplored fistula.
COMPLICATIONS
Provided the clinician involved has been appropriately trained and is aware of the contra-indications, NPWT is generally considered to be a safe process. A common problem is the pain associated with dressing changes. Dressings can usually be changed at the bedside but this process can be painful particularly if significant granulation tissue has anchored on to the foam. Whilst this may be distressing for the patient there is no convincing evidence to suggest that the pain associated with VAC dressings is any worse than that for conventional dressings. Instilling lignocaine into the tube prior to removal of the sponge or laying a non-adhesive dressing between the sponge and the wound bed at the time of application may help [19].
Although not common place, bleeding is probably the most serious complication that can occur with NPWT. Minor bleeding is allowed for at dressing changes but any significant bleeding must be addressed. Whilst there have been some reports of significant bleeding it is not clear that NPWT was causal in all cases [21, 22]. In one instance placement of a NPWT device adjacent to an exposed anterior tibial artery led to erosion and hemorrhage from that vessel [22]. If significant hemorrhage occurs this must be addressed immediately. The negative pressure should be disconnected and direct pressure should be applied to the wound bed. If bleeding is not controlled promptly then surgical exploration may be required.
Some authors have described chronic wound sepsis caused by retention of the foam dressing [23]. Such cases highlight the importance of employing trained clinicians to manage NPWT devices. In particular it is critical that adequate documentation is completed for each change of dressing including information with regards to the number of sponges removed and inserted.
EVIDENCE FOR NPWT IN TRAUMA & ORTHOPEDICS
The evidence supporting the use of NPWT in orthopaedic surgery is relatively sparse. The majority of the published literature focuses on case reports and case series which are unable to give definitive answers regarding the benefits of NPWT. Attempts have been made to use more robust research methods but many randomized controlled trials suffer from being underpowered or poorly designed.
One of the key problems with research in this field is that wounds are very difficult to standardise. They vary in size, shape, position and chronicity. Objective assessments of wound healing are not easy to define and labeling wounds as chronic, acute or sub-acute based on arbitrary timescales has little evidence base. Furthermore, good wound healing is reliant on multiple local and systemic factors and consequently each wound is very different to the next. In order to identify the independent effect of NPWT, large numbers of cases are required to reduce the effect of potential confounding factors.
WOUND COLONISATION
The effect of NPWT on bacterial colonization of wounds is controversial. The sealed environment created by NPWT goes against traditional wisdom with regard to the management of wound sepsis which advocates free drainage. Early animal studies, using a porcine model, suggested that despite the sealed environment created by NPWT it was associated with reduced bacterial counts when compared to standard dressing [16]. This evidence has not been reliably reproduced in the human model.
A study looking at the colonization of sponge in chronic wounds treated with NPWT demonstrated persistently high bacterial loads which failed to improve with subsequent sponge changes [24]. A further study which took biopsies from open wounds treated with NPWT showed no significant difference in bacterial load when compared to similar wounds treated with standard dressings [25].
Despite this many researchers have reported remarkably low infection rates following the administration of NPWT in high risk traumatic wounds. Leininger et al. based in a field hospital in Iraq, looked at 77 patients with high energy soft tissue injuries [26]. They used NPWT as an adjunct to serial surgical debridements and reported no subsequent infections. Others have shown similar low rates of infection in patients suffering from complex blast injuries treated with prolonged NPWT (avg. 70 days) [27].
The role of NPWT in treating wound infections is not clearly defined and proposed mechanisms of action have not been proven. There is certainly no strong evidence to suggest that NPWT reduces bacterial colonization as a direct result of negative pressure at the wound bed.
TRAUMA
One of the earliest uses of NPWT was in Trauma patients [28]. Debridement to healthy bleeding tissue is a basic principle in the management of traumatic wounds. This should include exploring the entire area of injury, debriding all areas of possible contamination, excising necrotic tissue and administering a thorough washout with normal saline. Primary closure is often undesirable as it may enable infection to propagate beneath the wound surface.
In cases of significant tissue loss or when there is concerns about residual infection or tissue viability due to vascular compromise then NPWT can be valuable as a bridge to secondary closure or flap coverage [29]. Proposed mechanisms by which NPWT supports wound healing include; controlling oedema, promoting granulation tissue, improving micro-vascular circulation and decreasing bacterial load at the wound bed.
Gomoll et al. [30] published a series of 35 cases where NPWT was used for a variety of orthopaedic wounds. The cases included wounds associated with tibial fractures, foot and ankle trauma and hip revision surgery. All cases were identified by the authors as being high risk for post-operative complications with prolonged wound drainage or swelling anticipated. NPWT was used for a mean duration of just over 3 days. The authors sited two key benefits from the use of NPWT. Firstly, they felt that over the same time period four dressing changes would have occurred if conventional techniques were employed. As such patients were less troubled by dressing changes and nursing time required to manage the wounds was reduced. Secondly, they experienced no cases of infection in this high risk group and felt that NPWT had directly contributed to a reduction in local infection rates. The study also mentions that NPWT was useful in obese patients and in wounds that were difficult to reach. Whilst these findings are encouraging they offer no comparative analysis of NPWT with conventional dressings.
Bollero et al. [31] considered the use of NPWT in a retrospective review of a variety of wounds in over 80 patients. The authors felt that the significant granulation tissue associated with NPWT enabled them to treat a high proportion of wounds with a split skin graft as opposed to a more complex flap reconstruction. Whilst this is again a promising outcome, the scope of its effect is hard to ascertain without any comparative data or long term follow up.
Stannard et al. [32] evaluated the ability of NPWT to reduce wound drainage and aid closure following surgery for high trauma injuries. This study had two separate streams. Firstly, it considered the effect of NPWT on reducing the discharge from a post-surgical wound with an established haematoma. Secondly, it investigated the ability of NPWT to prevent haematoma formation and aid closure in high risk traumatic wounds. Unlike the previous studies mentioned, subjects were randomized to receive either NPWT or standard pressure dressings. In both studies, the use of NPWT was associated with a significantly reduced duration of wound drainage (P=.03, P=0.02).
The effect of NPWT on preventing complications in high risk surgical wounds has been further investigated by Stannard [32] and his colleagues. They published a report on 249 patients who had been enrolled in a multi-centre randomized controlled trial [32]. All patients had closed surgical incisions associated with tibial plateau, pilon or calcaneal fractures. Subjects were then randomized to receive either standard dressings or NPWT post-operatively. They found that the relative risk of developing a post-operative infection was 1.9 times greater in patients who had been managed with standard dressings compared to NPWT. The rates of wound dehiscence were also greater in the control group.
OPEN FRACTURES
Dedmond et al [35] examined the role of NPWT in the treatment of patients with open tibia fractures. High energy open tibia fractures are serious injuries that often have severe complications. The infection rate for all Grade III injuries is about 24% and increases to 50% with Grade IIIB fractures [36]. This latter group have significantly more soft tissue damage and by definition have inadequate soft tissue to cover the boney anatomy. This study retrospectively reviewed 49 adult patients who had sustained 50 Grade III open tibial shaft fractures [36]. All patients had NPWT administered following the initial debridement and stabilization of the open fracture. The study found that rates of infection and non union were remarkably similar to historical controls. The principle benefit revealed by this small retrospective study was a reduction in free flap procedures required to definitively close the wound. At the end of the initial debridement patients were predicted to either require flap coverage in the future or not. Following the application of NPWT almost 30% of those predicted to need flap coverage did not require it. About half of the wounds were closed by skin grafting, primary closure or secondary intention. This positive outcome is encouraging, however, there is no supporting evidence that describes their ability to predict the need for flap coverage when other dressing techniques are used.
Rinket et al. [37] completed a retrospective review of 111 open tibia fractures treated with free flaps. They focused on the effect of NPWT in patients who had definitive flap reconstruction performed ‘sub acutely’ (between 8-42 days). This group of patients is at an increased risk of developing complications related to flap reconstruction when compared to those who have definitive surgery acutely. They found that the NPWT group had a lower overall complication rate (35% versus 53%), infection rate (6% versus 18%), and flap complication rate (12% versus 21%) compared with the gauze dressings group.
The NPWT group experienced significantly lower complication rates and the Authors recommended the use of NPWT as a bridging treatment in cases where definitive flap reconstruction was delayed. The patients were not randomized to the two treatment options and the decision to use NPWT was made by the attending surgeon. There was also no agreed end-point for stopping NPWT. A similar retrospective case series of 38 patients with Gustilo IIIB open tibia fractures also supports the use of NPWT as a bridging therapy for wounds that cannot be closed acutely [38]. However, they found that the use of NPWT beyond 7 days was associated with a concomitant rise in deep infection rates. Suggesting that NPWT as a bridging therapy is time-limited and does not allow surgeons to delay surgery indefinitely.
A case series of 37 open fractures supports the use of NPWT in paediatric trauma. Whilst they reported relatively low deep infection rates (5%) it is difficult to draw conclusions from this study as they did not grade the severity of injury or offer a control group that could be used to cross-reference their outcomes [39].
FASCIOTOMY WOUNDS
In cases of compartment syndrome, the treatment goal is to decrease compartment pressure and maintain tissue perfusion. Open fasciotomies permit this and involve the creation of large surgical wounds. Primary closure of these wounds is not appropriate and achieving a delayed primary closure is not always possible. Serial dressing changes are often needed until definitive closure is possible which puts the wound at risk of infection. Primary coverage with NPWT creates a closed environment which in theory protects the wound from outside infection, reduces local oedema and reduces the need for frequent dressing changes.
A large retrospective study of 458 patients demonstrated a significantly higher rate of primary closure in fasciotomy wounds treated with NPWT compared to standard wet-to-dry dressings [40]. Primary closure was also performed earlier in the NPWT group leading to reduction in length of hospital stay. Similar results were not seen in a prospective randomized study comparing NPWT with the shoelace technique [41]. This study involved 50 patients with 82 fasciotomy wounds to the leg who were randomized to receive either NPWT or a gradual suture approximation technique to facilitate wound closure. Wound closure time was significantly prolonged in the NPWT group (P=<0.001) and the cost of treatment was increased.
SPLIT SKIN GRAFTS
Split skin grafts (SSGs) are used to cover open wounds cause by trauma, burns, infection and malignancy. The success of such grafts relies on good integration at the recipient site. This integration can be significantly impaired by local infection, mechanical shearing and the accumulation of fluid between the graft and wound bed. NPWT has been used in the application of skin grafts both to prepare the recipient wound bed prior to grafting and to try and improve the chances of subsequent graft integration. The proposed mechanism by which it achieves this is by providing mechanical stability and preventing the accumulation of fluid beneath the graft.
Two RCTs have investigated the efficacy of NPWT as an adjunct to split skin grafting [42, 43]. The first study involved the use of SSGs in burns injuries [42]. Sixty patients were prospectively recruited and the use of NPWT was associated with improved graft integration post-operatively (p<0.01) [42]. Importantly, the clinicians responsible for assessing graft integration were blinded to the treatment received. However, the wound assessment was done on day four post-operatively with no long-term follow up.
The study by Chio et al [43] did not have similar outcomes. Their patients had all undergone free radial forearm flaps and the donor sites were treated with SSGs. They were unable to demonstrate any reduction in wound complications in the NPWT group. Wound assessment was not blinded, however, the use of a consistent donor site wound is arguably a more robust model than that used to treat potentially unpredictable burns injuries described above.
Neither study is able to give definitive evidence for the use of NPWT as an adjunct to split skin grafting.
REVISION HIP SURGERY FOR INFECTION
Infection is an uncommon but problematic complication of total hip arthroplasty (THA). Its consequences can be challenging to both patient and surgeon alike. The aim of surgery is to eliminate infection whilst retaining a functional and symptom free joint. In a review of 28 cases where surgical debridement and washout had failed, NPWT was employed to try and control local sepsis [44]. A peri-prosthetic sponge was inserted following meticulous debridement and exchange of the cup inlay and prosthetic head. Once the acute sepsis had resolved, based on clinical and biochemical markers, the sponge was removed. If at the time of sponge removal there were concerns of residual infection, the sponge was exchanged and the wound closed. The authors were able to control infection with negative cultures taken from the sponges in 26 out of 28 cases. There was no clinical recurrence of infection in the long-term with a mean follow up of 3yrs [44]. The study was not able to define how frequently the sponges should be exchanged and certainly more research is required to delineate which patients are most likely to benefit from NWPT.
CHRONIC WOUNDS
Chronic wounds are difficult to treat. They often affect the elderly with co-morbidities such as diabetes and chronic venous or arterial insufficiency. A cochrane review, including 7 randomized controlled trials (RCTs) and 205 participants, examined the benefits of NPWT in the management of chronic wounds [45]. NPWT was variously compared to other treatment modalities including gauze soaked in saline or Ringers solution or gauze in combination with a hydrocolloid gel. Primary outcomes included time to complete healing, reduction in wound area, time to surgical readiness and survival rate of subsequent grafts. Infection, pain, quality of life and economical aspects were secondary outcomes also considered. Out of the 7 RCTs only one managed to find a significant improvement in wound healing associated with NPWT [46]. They reported a significant reduction in wound volume in diabetic foot wounds treated with NPWT. The study was small and only included seven wounds from six patients. It is important to note that a reduction in wound volume does not necessarily relate to more rapid wound closure or any sustained benefit in the long term.
This Cochrane review highlights the paucity of evidence available for the management of chronic wounds with NPWT. A large, well-designed, prospective RCT is required to support the increasing use of NPWT in this area.
DIABETIC FOOT AMPUTATIONS
A large multi-centre RCT was developed to clarify the role of NPWT in the management of diabetic foot wounds following partial amputation up to the trans-metatarsal level [47]. 162 patients were enrolled and randomly assigned to either NPWT or moist wound therapy post-operatively. Results revealed that significantly more patients healed in the NPWT group (56% vs 39%, p=0.04). The time to heal was also faster in the NPWT group (p<0.005), and was associated with faster granulation tissue formation (p=0.002). The authors concluded that NPWT is a safe and effective treatment option for complex diabetic foot wounds. This study has some methodological weaknesses. It was for instance not possible to blind the patients from their treatment group allocation and the decision to close wounds surgically was physician dependent and not a randomized step in the management pathway. It is also important to note that this study was funded by KCI (San Antonio, Texas) who manufacture the V.A.C.® Therapy System. Despite these shortcomings the study certainly offers compelling evidence supporting the use of NPWT. Whether or not this relates to economic benefits or improvements in quality of life remains to be proven.
NEW ADVANCES IN NPWT
A summary of research of NPWT in orthopaedic surgery is presented in Table 2.
Summary of research of NPWT in orthopaedic surgery.
| Study | Method | Results | Author’s Conclusion/Comment |
|---|---|---|---|
| Morykwas MHL et al. [16] | Animal studies using porcine model | Increase in, granulation tissue (p<0.05), increased blood flow, decreased bacteria counts (p<0.05) and improved flap survival (p<0.05) compared to standard dressings | |
| Yusef E et al. [24] | 68 foams were collected from 17 patients with chronic wounds | In 65 foams over 2 bacteria types were found. The bacterial load remained high. This was higher in polyvinyl alcohol foam (6.1 +- 0.5 CFU/ml) than polyurethane (5.5+-0.8) p=0.02. | Bacterial load remains high in NPWT foam. Routine changing does not reduce the bacterial load. |
| Moulës CM et al. [25] | Prospective randomized trial. 54 patients with open wounds were randomized to NPWT or conventional moist gauze therapy. | No significant difference in time needed to reach “ready for surgery” stage or bacterial load at the wound bed. Wound surface area was significantly reduced in the NPWT group (p<0.05) | Improvements in wound healing associated with NPWT cannot be explained by reduced bacterial load at the wound bed. |
| Leininger BE et al. [26] | Retrospective case series of 88 high energy soft tissue wounds in 77 patients from wartime missile injuries. | Wound infection rate 0%, overall complication rate 0%. | First report of use of NPWT in high energy soft tissue wounds in a deployed wartime environment. |
| Labler L et al. [29] | Case comparison of type IIIB fractures treated with NPWT or Epiguard ®. | The rate of infection was substantially higher in the epiguard group (6/11 vs 2/13). | NPWT reduced the rate of infection and is an alternative choice for the management of type IIIB fractures. |
| Bollero D et al. [31] | Retrospective review of 35 patients with 37 acute traumatic wounds treated with VAC therapy. | All patients quickly developed healthy granulation tissue and enabled many wounds to be treated with split skin graft | NPWT may represent a good alternative to immediate reconstruction in selected cases of acute complex trauma, with a stable result using a minimally invasive approach. |
| Bhattacharyya T et al. [38] | Retrospective study of 38 patients with Gustillo IIIB tibia fractures | Patients who underwent definitive coverage within 7 days had significantly lower rates of infection (12.5% versus 57% p<0.008). | Routine use of NPWT does not allow delay of soft tissue coverage past day 7 without a rise in infection rates. |
| Stannard JP et al. [32] | Multi-centre, prospective randomized trial comparing NPWT to standard dressings in high risk surgical wounds (249 patients) | Relative risk of developing a post-operative infection was 1.9 times greater in patients managed with standard dressings. The rates of wound dehiscence were also greater in the control group. | Demonstrates the use of NPWT as a prophylactic device in preventing incisional wound complications in high risk surgery. |
| Chio et al. [42] | RCT including 54 patients. Patients undergoing split skin grafting of radial forearm free flap donor site were randomized to receive NPWT or pressure dressing. | There was no significant difference in percentage of graft failure (4.5% versus 7.2% p=0.361) between the two groups | NPWT does not offer a significant improvement over pressure dressings for the management of skin grafts. |
| Llanos S et al. [43] | RCT including 60 patients with thermal injuries treated with split skin grafts randomized to receive NPWT or pressure dressing. | NPWT was associated with significantly reduced loss of the skin graft and a reduced hospital stay (avg. 13.5 days vs 17 in the control group, p=0.001). | NPWT improves split skin graft uptake in thermal injuries. |
| Armstrong DG et al. [43] | Multicenter RCT including 162 patients. NPWT was compared to standard dressings in the management of diabetic foot wounds resulting from partial amputation surgery | More patients healed in the (p=0.04) and the rate of healing was faster in the NPWT group (p=0.005). The frequency and severity of adverse events were similar in both groups. | NPWT seems to be safe an effective treatment of complex diabetic foot wounds. Study funded by KCI (San Antonio, Texas) who manufacture the V.A.C.® Therapy System. |
NPWT WITH INSTILLATION
Recent studies have trialled NPWT with the addition of instillation (NPWTi). A heavy bacterial load in a wound increases the metabolic requirements causing an inflammatory response and slowing wound healing. NPWT with instillation combines the negative pressure dressing with topical irrigation solutions that enable mechanical debridement at the wound bed.
One prospective pilot study of 15 patients with complex infected wounds treated with NPWT instillation were compared to a retrospective control group treated with stand moist wound care [48]. The treatment group experienced a more rapid resolution of infection and fewer days in hospital. The control group had more chronic wounds such as pressure ulcers than the treatment group and the authors defended this stating that initial surgical debridement converted these chronic wounds into acute ones. The authors used saline, sterile water or silver nitrate as the irrigation fluid and were not able to specify which of these, if any, was best.
A prospective case series of 131 patient from three centres in France has also shown positive results using this method [49]. NPWTi was used in a variety of wounds and in 98% of cases the wounds could be closed and did not recur. A proportion of these patients (46 subjects) were failing to progress with standard NPWT and NPWTi was identified as the mechanism which ‘jump-started’ wound healing. This study offers no direct comparison with other treatment modalities and as such the conclusions that can be drawn from it are limited.
A retrospective case-control study [50] compared NPWTi to gentamicin beads in the treatment of post-traumatic osteomyelitis. The study demonstrated significantly reduced rates of infection recurrence in the NPWTi group (10%) compared to the control group (59%). Whilst preliminary reports regarding this novel therapy are encouraging, there is a distinct lack of robust evidence supporting it. A prospective randomized trial including NPWT, NPWTi and standard dressings has yet to be completed.
SINGLE USE NPWT
Recent advances in NPWT have focussed on the practical aspects of its use and the development of ultraportable, single use devices such as the PICOTM system (Smith & Nephew Medical Ltd, Hull, UK). This modality is very new and the research supporting it is sparse. One prospective non-comparative study of 20 patients with high risk surgical wounds was undertaken to see if such devices were safe and effective [51]. All but one patient was recorded as having a wound that had healed or was healing at two weeks. The authors emphasise the preliminary nature of this study.
Whilst more research needs to be done this system is attractive for many reasons. It is easy to use, has less impact on patient mobility and once applied it requires less nursing care than traditional NPWT devices.
SUMMARY
NPWT continues to gain popularity within the field of Orthopedics and the list of indications for which surgeons have explored its use has grown exponentially since its conception. It is used to treat wounds that are acute and chronic, closed and open, infected and non-infected and any combination therein. Initially, NPWT was used as an adjunct to promote wound healing in problematic open wounds. More recently studies have focused on using NPWT as a prophylactic measure to prevent high risk wounds from becoming infected or breaking down [32]. Whilst efforts have been made to provide an evidence base to guide its use, this has been limited by a lack of good quality evidence.
The majority of support for NPWT comes from retrospective studies that fail to compare it to other wound management techniques. The most robust research to date includes the use of NPWT following amputations for diabetic feet [46]. It is disappointing that this prospective RCT was funded by the manufacturer’s themselves. The bulk of the published literature concludes that NPWT is a safe technique but requires more prospective research to support its use. Having extensively reviewed the subject this review article can only repeat these sentiments.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
Declared none.


