All published articles of this journal are available on ScienceDirect.
Transcutaneous Treatment with Vetdrop® Sustains the Adjacent Cartilage in a Microfracturing Joint Defect Model in Sheep
Abstract
The significance of the adjacent cartilage in cartilage defect healing is not yet completely understood. Furthermore, it is unknown if the adjacent cartilage can somehow be influenced into responding after cartilage damage. The present study was undertaken to investigate whether the adjacent cartilage can be better sustained after microfracturing in a cartilage defect model in the stifle joint of sheep using a transcutaneous treatment concept (Vetdrop®).
Carprofen and chito-oligosaccharids were added either as single components or as a mixture to a vehicle suspension consisting of a herbal carrier oil in a water-in-oil phase. This mixture was administered onto the skin with the aid of a specific applicator during 6 weeks in 28 sheep, allocated into 6 different groups, that underwent microfracturing surgery either on the left or the right medial femoral condyle. Two groups served as control and were either treated intravenously or sham treated with oxygen only. Sheep were sacrificed and their medial condyle histologically evaluated qualitatively and semi-quantitatively according to 4 different scoring systems (Mankin, ICRS, Little and O’Driscoll).
The adjacent cartilage of animals of group 4 treated transcutaneously with vehicle, chito-oligosaccharids and carprofen had better histological scores compared to all the other groups (Mankin 3.3±0.8, ICRS 15.7±0.7, Little 9.0±1.4).
Complete defect filling was absent from the transcutaneous treatment groups.
The experiment suggests that the adjacent cartilage is susceptible to treatment and that the combination of vehicle, chitooligosaccharids and carprofen may sustain the adjacent cartilage during the recovery period.
INTRODUCTION
Current cartilage repair concepts such as autologous cartilage implantation (ACI) [1], matrix-associated autologous chondrocyte implantation (MACI) [2] and/or tissue engineered cartilage [3, 4] defect filling face the problem of deficient bonding and integrating of the newly formed tissue to the adjacent cartilage. This phenomenon not only occurs in defect filling concepts, but is also seen in marrow stimulating techniques such as microfracture [5, 6] and Pridie drilling [7, 8]. In histological sections, the adjacent cartilage is commonly shown to undergo degradation indicated by diminished metachromatic staining because of proteoglycan loss, clustering of chondrocytes and superficial fibrillation [9-11]. Shapiro et al. considered that micromotion through mechanical shear stress between the new and the host tissue could lead to displacement and microfissures and initiate a degenerative process [9].
The significance of the adjacent cartilage in defect repair is not yet fully understood and studies investigating the adjacent cartilage are rare [9, 11-13]. It is unknown whether the viability of the adjacent cartilage can be sustained after a lesion and thus, improves the integration and bonding of new tissue.
The present animal study focused on the sustainability of the adjacent cartilage and whether the properties of the adjacent cartilage can be influenced pharmaceutically during healing. To do so, a novel transcutaneous treatment concept (Vetdrop®), was used. The concept is based on liposomal vesicles and oxygen delivery of cartilage-modifying medications that can be administered transcutaneously onto the affected joint area. So far, this application was used for the treatment of joint diseases in horses with excellent clinical outcomes. Apart from applications used in this study, another mixture based on hyaluronic acid (0.5%) is also available for horses. Furthermore, liposomal transcutaneous therapy has recently gained popularity for human applications [14, 15] with good patient compliance resulting from easy application, with only minor side effects [16].
The aim of the present study was to show whether the adjacent cartilage in a defect is susceptible to the transcutaneously delivered pharmaceuticals and therefore, can be manipulated for better defect healing. The study was based on the hypothesis that the adjacent cartilage of a primary defect could be better preserved through transcutaneously administered anti-inflammatory medication and thus, result in better bonding to the newly formed tissue. It was expected that the transcutaneously treated groups show a better histological outcome than the control, systemically or sham treated groups. To test the hypothesis, we utilized a sheep microfracturing joint defect model.
MATERIALS & METHODS
Transcutaneous Treatment Concept
The transcutaneous treatment system (Vetdrop®) was developed by MedDrop Technology AG (Thundorf, Switzerland) for transcutaneous application of natural and synthetic pharmaceutical ingredients (Fig. 1). The system consists of an oxygen generator and an application system, which is used in connection with specially developed vehicles. The oxygen generator is extracting oxygen from the atmosphere and this high concentrated oxygen serves as a propellant. The oxygen is first stored in a pressure container and during treatment the oxygen flows through a pressure reducing valve and a treatment tube to the application device.

Transcutaneous Treatment Concept. Schematic design of the transcutaneous application with Vetdrop®.
The applicator serves as a nano-dispersion-device consisting of a drug reservoir, which lies within a gas tank. The pharmaceutical ingredients are filled into the drug reservoir through a port. Oxygen arrives in the gas tank through an adapter. The drug reservoir is surrounded by the gas tank and ends in a delivery spout that encloses a diffuser. The oxygen propels and transports the carrier substance under pressure through the diffuser while also mixing with it. The assembling of the diffuser utilizes the so-called Venturi-Effect (fluid pressure decreases in response to a constricted area of flow), in order to atomise the micro emulsion. The size of the droplets lies in the range of nanometers. The size is regulated via a needle lace, which is altering the width of the port.
The employed micro emulsions are a proprietary product of Arvine Pharma AG (MedVital Serum, Arvine Pharma AG, Thundorf Switzerland). The vehicles are based on an oil-in-water or water-in-oil micro emulsion. The active ingredients are incorporated into their watery phase. To emulsify, the ingredients are bond in adducts with adjuvants and their surface tension is broken. In this way, the pharmaceutically active ingredients are dissolved and no particle binding occurs, which allows a safe use in the micro- and nano sizes.
Animals
Twenty-eight Swiss alpine sheep between 2 and 3 years (average 2.5 years) of age and body weights between 48 – 76 kg (average 59.4 kg) were randomly allocated into 6 groups. Four groups with 6 animals each were used for transcutaneous treatment and 2 groups with 2 animals each were used as controls.
Ten days before surgery, the sheep were brought to our facilities and adapted to their new environment. Prior to surgery, the sheep were fasted for 24 h with water available ad libitum. All experiments were conducted according to the Swiss regulations of animal welfare and were authorized by the local authorities (application No 193/2010).
Anaesthesia
The animals were sedated with xylazine (Xylazine, 0.1mg/kg BW; Streuli Pharma AG, Uznach, Switzerland) and buprenorphine (Temgesic®, 0.01mg/kg BW; Essex Chemie AG, Luzern, Switzerland) both i.m.. Anaesthesia was induced with diazepam (Valium®, 0.1mg/kg BW; Roche Pharma AG, Reinach, Switzerland), ketamine i.v. (Ketasol®, 2mg/kg BW; Dr. E. Graeub AG, Bern, Switzerland) and propofol i.v. (Propofol 1% Fresenius Kabi®, 0.4mg/kg BW; Fresenius Kabi (Schweiz) AG, Stans, Switzerland. The animals’ tracheas were intubated and anaesthesia was maintained with 1- 1.5% isoflurane (Isoflo®, Abbott AG, Baar, Switzerland) and propofol as a constant rate infusion (0.01mg/kg BW/min). Furthermore, the animals received an infusion of Ringer’s lactate solution at a rate of 10ml/kg BW/hr.
Equine tetanus serum, s.c. (Tetanus serum 3000 IU, Veterinaria AG, Zürich, Switzerland), was given as a single dose. For pre- and post-operative antibiotic prophylaxis Benzylpenicillin (Procain-Penicillin Streuli® ad us. vet., 30’000 IU/kg BW, BID; Streuli Pharma AG, Uznach, Switzerland) and Gentamicin (Vetagent® ad us. vet., 6mg/kg BW, SID; Veterinaria AG, Zürich, Switzerland) were given i.v. for 4 days.
Surgical Procedure
Each sheep was placed in dorsal recumbence with the limb to be operated upright and fixed in maximal flexion. In this position, the weight-bearing condyle of the femur is exposed in an optimal way and when the limb is back in extension the created defect lies very central to the axis of weight bearing forces.
The approach to the joint was achieved through a medial para-patellar approach extending from the medial patellar ligament distally to the tibial tuberosity. The subcutaneous tissue and the superficial fascia were cut and the joint was opened just above the weight-bearing part of the medial femur condyle. The adipose tissue was partially taken away to gain a better insight into the joint. The surgery site was kept open with the help of a Weitlaner retractor. A punch-biopsy trocar (Ø 6mm) was used to place a round defect into the hyaline cartilage in the middle of the condyle. The so-called “mango-technique” was used to cut the cartilage within the circle with a scalpel blade size 11 like a map grid. Afterwards, the cartilage pieces were easily removed from the subchondral bone without further damaging the cartilage wound edges. The calcified cartilage zone was removed with a head burr, being careful not to the subchondral bone plate. Four (4) small holes were placed with a micro-pic instrument and evenly distributed within the defect through the tidemark of the subchondral bone as earlier described by Steadman and Frisbie [5]. After flushing, the joint was closed with an interlocking suture through the fascia and the joint capsule (Vicryl 2-0) and a simple continuous suture for the subcutis. The cutis was closed with staples.
After recovery, the sheep were kept in groups of three or four in the stable with free access to food and water.
Postoperative Management
The sheep were treated 3 times per week with the transcutaneous treatment system Vetdrop® for 15 minutes during 6 weeks. The first treatment took part immediately after recovery from surgery and then every 2-3 days for a total of 18 applications. The area around the stifle was kept free from hair and the skin was cleansed with ethanol before administering the therapy. The applicator was held at a distance of approximately 1cm from the treated area in an angle of 90 degrees. The areas of skin around the stifle joint were treated with a square area of approximately 10 cm2 on the medial side of the limb, medial to the surgical wound and an equally sized area lateral to the surgical wound. The employed ingredients in the mixture varied depending on the group allocation. For treatment of groups 1 – 3, the supplier (MedDrop Technology AG) provided the pre-assembled formulations. For the treatment of group 4 (VECHCA), two components were mixed together just before application. Component A consisted of vehicle and carprofen and component B consisted of vehicle only. The carprofen concentration in component A was 13.39%. Components A & B were mixed in a 1:1 ratio. Due to possible fluctuations during the procedure of mixing a variation of ± 15% was probable, such that a carprofen concentration between 5.7% and 7.7% (mean 6.7%) was assumed. Group 1 (VE; 6 sheep) was treated with vehicle only, group 2 (VECH; 6 sheep) with vehicle and chito-oliogosaccharids (2%), group 3 (VECA; 6 sheep) with vehicle and carprofen (5.74%), group 4 (VECHCA; 6 sheep) with vehicle, chito-oligosaccharids (2%) and carprofen (6.7 ± 15%), group 5 (S; 2 sheep) served as control and was sham treated with oxygen only and group 6 (CA; 2 sheep) was the 2nd control group, which was intravenously treated with only carprofen (5%). This group received once a day 4mg/kg BW carprofen (Rimadyl®, Pfizer AG, Zürich, Switzerland) during 4 days intravenously without transcutaneous application.
After the 6 week treatment period, the sheep were allowed to roam on the pastures for another 6 weeks until sacrifice. They were sacrificed at the university-owned slaughterhouse in the animal hospital.
Harvesting and Preparation of the Bone Samples
The femur condyles were harvested and examined for macroscopically visible changes, such as inflammation and degradation of the defect area (Fig. 2). The contralateral side served as control. The defect area was photographically documented (Digital-Foto Sony DSC–R1, Sony Corporation) as well as qualitatively evaluated. Afterwards, lateral and dorsal radiographs of the femural condyles were taken (49kV, 1 s, 3 mA; Faxitron X-ray systems, Hewlett Packard, Mc Minnville Division, Oregon, USA).
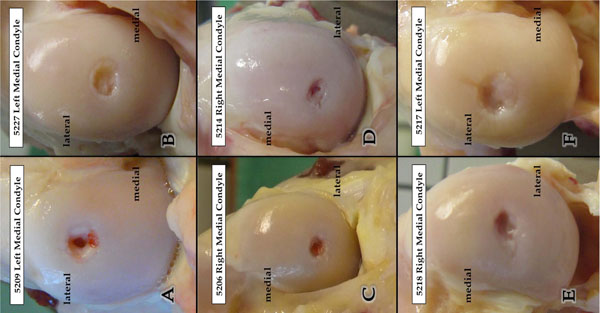
Appearance of the defects after slaughtering: Pictures A – F show an example of the appearance of the defects after slaughtering of each group. A Group1 (VE), B Group 2 (VECH), C Group 3 (VECA), D Group 4 (VECHCA), E Group 5 (S), F Group 6 (CA). The visual appearance was also within the groups very variable and showed no uniform picture.
The defects were excised from the condyles using a special band saw (Kolbe Maschinentechnik GmbH, Elchingen, Germany) and samples prepared for histology. Bone blocks were fixed in 4% buffered formaldehyde for 1 week. Thereafter, the samples were dehydrated in a series of ethanol, defatted in xylene under vacuum, and embedded in methylmethacrylate (methacrylacid–methylester; dibuthyl-phtalate and perkadox in a proportion 89.5:10:0.5). Ground sections (30–40 μm) and thin sections (5 μm) were cut in the longitudinal axis with a precision saw (Leica1 SP1600) resp. microtome (Leica1 RM 2155; Leica Instruments GmbH, Nussloch, Germany). Before the ground sections were glued to the Acropal slides (Cementit CA 12; Merz Benteli AG, Niederwangen, Switzerland), microradiographs were taken (55kV and time frame was adapted to individual specimen, Faxitron X- Ray System, Hewlett & Packard, Kodak X-OMAT MA Film, Kodak, France).
Ground sections were surface-stained with toluidine blue, whereas thin sections were deplastified with methoxyethyl-acetate (Merck AG, Zug, Switzerland) and then stained with either toluidine blue or von Kossa/McNeal staining.
Histological Evaluation
Four different scoring systems were chosen for evaluation of the histology sections: Mankin [17], ICRS I [18-20], O’Driscoll [21] and a grading system described by Yoshimi et al. [22] and modified by Little et al. [23].
The various defect sections were scored in order to assess differences between treatment groups. Furthermore, comparisons were made between the defect area and the adjacent cartilage sections. For scoring the defect part, the Mankin, ICRS and the O’Driscoll score systems were used while the adjacent cartilage was scored according to the Mankin, ICRS and the Little score system.
Before scoring, each section was divided into five parts. The sections were divided from left to right with the orientation scaling-up. The first part was called “before defect”. This cartilage part was within the normal cartilage without pathological changes and away from the defect and served as control. The second part was called “defect margin I”. This part started from the area of first pathological changes towards the cartilage defect and ended just before the defect itself. The third part consisted of the defect itself. After this, there was a second “defect margin II”, which started just after the defect part and went as far as pathological changes in the cartilage were present. The last part was called “after defect” consisting of normal cartilage again.
The parts before and after defect were not scored as their cartilage showed no pathological changes; they served as controls, respectively references to assess changes in the other defect parts. Scored were the defect part and defect margins I and II.
Statistical Analysis
Mean values were calculated and quantitative data generated from the histological grading systems were analyzed by two factor analysis of variance (ANOVA) to assess overall differences. Post hoc tests (Scheffe and Bonferroni) were conducted to determine differences between individual groups. All statistical tests were performed with a commercially available software (PASW Stats for MacIntosh, Version 19, IBM Corporation, Armonk, USA).
RESULTS
Surgical Procedure
The surgery was well tolerated and all animals showed immediate and good weight bearing postoperatively. Some of the sheep developed a slight to moderate subcutaneous hematoma on the medial side of the operated limb (8/28 sheep). The effusion was removed with a syringe once a week if the animals were disabled during ambulation. All effusions disappeared after 6 weeks. The surgical technique was well standardized and also the transcutaneous treatment itself was easy to carry out as scheduled. All animals tolerated the 18 applications without complications.
Qualitative Evaluation of Adjacent Cartilage
All typical changes known to appear in the adjacent part of osteoarthritic cartilage or surgically treated cartilage defects, respectively, were found. This included disruption of the tissue, loss of metachromatic staining and increased cluster formation of chondrocytes [11]. Furthermore, bone remodelling in the subchondral bone was abundant in all groups. The calcified cartilage layer was appropriately maintained in all groups. Cyst-like lesions were detected only in one case each of group 1 (VE) and 5 (S), and in two cases of group 3 (VECA). They appeared oblique and lateral of the defect part and stretched into the defect margin. In group 1 (VE) few but extensive clusters of chondrocytes were observed, and similarly in group 2 (VECH) whereas in group 4 (VECHCA) mainly cell duplets were recorded.
In groups 3 (VECA) and 5 (S), we recorded many single chondrocytes in the adjacent cartilage and an intense increase of metachromatic staining (Fig. 3).
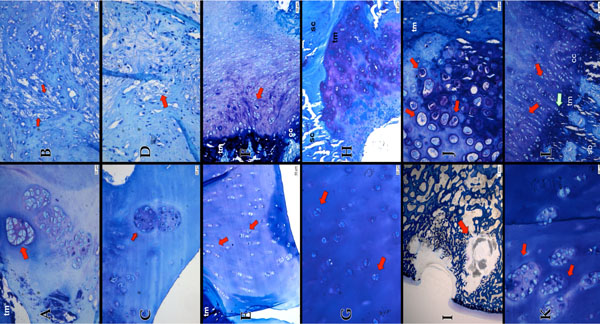
Group 1 (VE) (Pictures A & B) huge cell nests (red arrow) in the adjacent cartilage and a slight loss of matrix staining visible. The viability of the matrix is reduced. (tm=tidemark). (A) The defect part mainly consist of fibrous tissue (red arrows) (B) Group 2 (VECH) (Pictures C & D): huge cell nests, but the viability of the matrix in the adjacent cartilage is maintained (C). Similar to Group 1 the Defect in Group 2 is filled with fibrous tissue. Though it appears not as dense as in Group 1 (D) Group 3 (VECA) (Pictures E & F): The clusters in the adjacent cartilage are visible in colums (red arrows) and there are only very few big cell nests (E). The defect part is mainly filled with a mixture of fibro- cartilage (red arrow) and fibrous tissue (tm=tidemark, cc=calcified cartilage) (F) Group 4 (VECHCA) (Pictures G & H): The adjacent cartilage appears almost normal hyaline like. Cluster appears as douplets (red arrow) huge cell nests are rare (not shown) (G) Migration of cells from the subchondral bone into the defect part (sc=subchondral bone, tm=tidemark) (H) Group 5 (S) (Pictures I & J): Cyste like lesion stretching into the adjacent cartilage (ground section 30-40 µm, staining with toluidine blue) (I) Hypercellular chondrocytes (red arrows) at the defect basis (tm=tidemark) (J) Group 6 (CA) (Pictures K & L): Huge and numerous cell nests in the adjacent cartilage (red arrows) (K) hyaline-like appearance in the defect part (red arrows). Chondrocytes grow from the subchondral bone into the defect part (green arrow), similar as in Group 4 (cc= calcified cartilage, sb=subchondral bone, tm=tidemark) (L) (If not elsewise indicated: 5 µm section, staining with toluidine blue).
The adjacent cartilage showed signs of degeneration in all groups and no statistically significant differences between the groups were found in the adjacent cartilage, except for when using the Little scoring system.
Group 4 (VECHCA) had less surface irregularities visible in the adjacent cartilage as compared to the other groups when scored according to Little, and was statistically significant (p= 0.01) as compared to group 2 (VECH). Cluster formation of chondrocytes was higher in group 6 (CA). Although this was common in all groups, in group 6, we recorded more multiple clusters. Clusters in groups 3 (VECA) and 4 (VECHCA) consisted predominately of duplets and a few triplets. Adjacent Cartilage in groups 1 (VE), 2 (VECH) and 5 (S) had more triplets than duplets.
Groups 1 (VE), 2 (VECH) and 6 (CA) showed a greater decrease in toluidine blue staining, however, differences were not statistically significant. Nevertheless, group 4 (VECHCA) showed an obvious trend for improved histological scores as compared to all the other groups using all three score systems
Cell population viability of the adjacent cartilage was maintained in all six groups, and the cartilage mineralization was regarded as normal within the calcified zone. Nevertheless, the tidemark and subchondral bone revealed statistically significant differences: groups 4 (VECHCA) and 6 (CA) showed lower scores than the other groups (p= 0.0001, p=0.008 respectively). When comparing the overall score group 4 (VECHCA) to groups 1 (VE) and 2 (VECH) it was statistically significantly lower (p=0.006, p=0.002 respectively) (Tables 1-4).
Mankin Scores of the Margin & Defect Part
| Mankin | Margin | Defect | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Structure | Cells | Toluidine-Blue Staining | Tidemark Integrity | Total | Structure | Cells | Toluidine-Blue Staining | Tidemark Integrity | Total | |
| Group 1 | 0.42 ±0.7 | 1.5 ±0.8 | 0.9 ±0.5 | 0.7 ±0.5 | 3.3 ±1.2 | 6.0 ±0.0 | 2.0 ±0.9 | 2.2 ±1.0 | 1.0 ±0.0 | 11.2 ±2.0 |
| Group 2 | 1.0 ±1.0 | 2.0 ±0.0 | 1.0 ±0.5 | 0.8 ±0.5 | 4.8 ±2.0 | 5.2 ±2.0 | 2.5 ±0.5 | 2.5 ±0.5 | 1.0 ±0.0 | 11.2 ±2.6 |
| Group 3 | 1.1 ±0.9 | 2.0 ±0.0 | 0.9 ±0.3 | 0.5 ±0.3 | 3.5 ±2.5 | 6.0 ±0.0 | 2.5 ±0.8 | 2.3 ±0.8 | 1.0 ±0.0 | 11.8 ±1.6 |
| Group 4 | 0.2 ±0.3 | 1.8 ±0.6 | 0.7 ±0.37 | 0.8 ±0.7 | 3.3 ±0.8 | 6.0 ±0.0 | 2.3 ±0.8 | 1.8 ±1.0 | 1.0 ±0.0 | 11.2 ±2.0 |
| Group 5 | 0.5 ±0.7 | 2.0 ±0.0 | 0.5 ±0.7 | 0.8 ±0.4 | 4.0 ±0.0 | 6.0 ±0.0 | 2.0 ±0.0 | 2.0 ±0.0 | 1.0 ±0.0 | 11.0 ±0.0 |
| Group 6 | 0.8 ±0.4 | 2.0 ±0.0 | 1.3 ±0.4 | 0.8 ±0.4 | 4.5 ±0.7 | 3.5 ±3.5 | 2.0 ±0.0 | 1.0 ±0.0 | 1.0 ±0.0 | 7.5 ±3.5 |
ICRS Scores of the Margin & Defect Part
| ICRS | Margin | Defect | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Surface | Matrix | Cell Distribution | Cell Population Viability | Subchondral Bone | Cartilage Mineralization | Total | Surface | Matrix | Cell Distribution | Cell Population Viability | Subchondral Bone | Cartilage Mineralization | Total | |
| Group 1 | 2.3 ±0.9 | 2.8 ±0.4 | 1.7 ±0.6 | 3.0 ±0.0 | 2.2 ±0.6 | 3.0 ±0.0 | 14.8 ±0.7 | 0.0 ±0.0 | 1.0 ±1.0 | 0.3 ±0.8 | 3.0 ±0.0 | 2.0 ±0.6 | 3.0 ±0.0 | 9.3 ±1.8 |
| Group 2 | 1.5 ±1.0 | 2.8 ±0.4 | 1.8 ±0.4 | 3.0 ±0.0 | 2.1 ±0.2 | 3.0 ±0.0 | 14.2 ±0.7 | 0.0 ±0.0 | 0.2 ±0.4 | 0.0 ±0.0 | 2.0 ±1.0 | 1.8 ±0.4 | 2.5 ±1.2 | 6.5 ±1.2 |
| Group 3 | 1.1 ±1.1 | 3.0 ±0.0 | 1.8 ±0.5 | 3.0 ±0.0 | 1.9 ±0.6 | 3.0 ±0.0 | 12.5 ±0.8 | 0.0 ±0.0 | 1.0 ±0.9 | 0.0 ±0.0 | 3.0 ±0.0 | 1.8 ±0.4 | 3.0 ±0.0 | 8.8 ±1.0 |
| Group 4 | 2.5 ±0.9 | 2.9 ±0.2 | 1.9 ±0.2 | 3.0 ±0.0 | 2.2 ±0.4 | 3.0 ±0.0 | 15.7 ±0.7 | 0.0 ±0.0 | 0.3 ±0.8 | 0.0 ±0.0 | 3.0 ±0.0 | 1.8 ±0.4 | 3.0 ±0.0 | 8.2 ±1.0 |
| Group 5 | 3.0 ±0.0 | 2.5 ±0.7 | 1.5 ±0.7 | 3.0 ±0.0 | 2.0 ±0.0 | 3.0 ±0.0 | 15 ±1.2 | 0.0 ±0.0 | 0.0 ±0.0 | 0.0 ±0.0 | 3.0 ±0.0 | 2.0 ±0.0 | 3.0 ±0.0 | 8.0 ±0.0 |
| Group 6 | 0.0 ±0.0 | 3.0 ±0.0 | 2.0 ±0.0 | 3.0 ±0.0 | 2.0 ±0.0 | 3.0 ±0.0 | 13 ±1.2 | 0.0 ±0.0 | 1.5 ±0.7 | 0.0 ±0.0 | 3.0 ±0.0 | 2.0 ±0.0 | 3.0 ±0.0 | 9.5 ±0.7 |
Scores According to Little of the Margin Part
| Little | Margin | ||||||
|---|---|---|---|---|---|---|---|
| Structure | Cellularity | Cell Cloning | Territorial Toluidine Blue | Interterritorial Toluidine Blue | Tidemark/Calcified Cartilage/Subchondral Bone | Total | |
| Group 1 | 1.1 ±0.5 | 2.3 ±0.8 | 3.3 ±0.8 | 1.4 ±0.6 | 1.3 ±0.4 | 3.0 ±0.0 | 12.3 ±1.5 |
| Group 2 | 1.7 ±0.3 | 1.8 0.4 | 3.4 ±0.4 | 1.5 ±0.6 | 1.3 ±0.4 | 3.0 ±0.0 | 12.7 ±1.0 |
| Group 3 | 1.2 ±0.6 | 1.4 ±0.6 | 2.8 ±1 | 1.4 ±0.2 | 0.9 ±0.6 | 3.0 ±0.0 | 10.8 ±1.8 |
| Group 4 | 0.7 ±0.3 | 1.3 ±0.5 | 3.0 ±0.9 | 0.9 ±0.2 | 1.1 ±0.7 | 2.0 ±0.0 | 9.0 ±1.4 |
| Group 5 | 1.0 ±0.7 | 1.8 0.4 | 3.0 ±0 | 1.3 ±0.4 | 1.3 ±0.4 | 3.0 ±0.0 | 11.3 ±0.4 |
| Group 6 | 1.0 ±0.0 | 2.3 ±1.1 | 3.5 ±0.0 | 2.0 ±0.0 | 1.3 ±0.4 | 2.0 ±0.0 | 12.0 ±0.7 |
O’Driscoll Score of the Defect Part
| O‘Driscoll | Defect | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cellular Morphology | Staining of the Matrix | Surface Regularity | Structural Integrity | Thickness | Bonding to Adjacent Cartilage | Hypocellularity | Chondrocyt Clustering | Freedom from Degenerative Changes in Adjacent Cartilage | Total | |
| Group 1 | 1.0 ±1.0 | 1.5 ±0.8 | 1.2 ±1.0 | 0.5 ±0.5 | 0.5 ±0.8 | 1.2 ±0.7 | 1.3 ±1.0 | 1.0 ±0.0 | 2.0 ±0.0 | 10.2 ±5.0 |
| Group 2 | 0.3 ±0.8 | 1.3 ±0.5 | 1.7 ±0.8 | 0.5 ±0.5 | 0.0 ±0.0 | 0.5 ±0.5 | 0.7 ±0.8 | 1.0 ±0.0 | 2.0 ±0.0 | 8.0 ±1.5 |
| Group 3 | 0.3 ±0.8 | 1.3 ±0.5 | 2.0 ±0.0 | 0.7 ±0.5 | 0.2 ±0.4 | 0.6 ±0.5 | 1.2 ±1.3 | 1.0 ±0.0 | 2.0 ±0.0 | 8.5 ±3.8 |
| Group 4 | 0.3 ±0.8 | 1.3 ±0.8 | 0.3 ±0.8 | 0.0 ±0.0 | 0.2 ±0.4 | 0.8 ±0.4 | 1.2 ±1.0 | 0.8 ±0.4 | 1.8 ±0.4 | 6.8 ±3.7 |
| Group 5 | 1.0 ±1.4 | 1.0 ±0.0 | 0.0 ±0.0 | 0.0 ±0.0 | 0.0 ±0.0 | 1.0 ±0.0 | 1.5 ±0.7 | 1.0 ±0.0 | 1.5 ±0.7 | 7.0 ±1.4 |
| Group 6 | 2.0 ±0.0 | 2.5 ±0.9 | 2.0 ±0.0 | 1.0 ±0.0 | 0.5 ±0.7 | 1.0 ±0.0 | 3.0 ±0.0 | 1.0 ±0.0 | 2.0 ±0.0 | 15.0 ±1.4 |
Comparisons between the Mankin and the ICRS values showed a Pearson’s correlation of -0.638 and therefore indicated a strong relationship (p>0.001) (Fig. 4).

ICRS versus Mankin histology scores. Comparison between the Mankin and the ICRS score showed meaningful relationship.
Semiquantitative Evaluation of Adjacent Cartilage
Qualitative Evaluation of the Defect
The defects were partly or only marginally filled with a mixture of fibro-cartilaginous, fibrous or soft tissue. A normal cartilaginous structure within the defect was not identifiable in any group, although differences between groups were noticed. Group 6 (CA) had the most cartilage-like defect filling with a mixture of hyaline-like and fibrocartilage, whereas in group 5 (S), the defect was mainly filled with soft tissue but no fibrocartilage was visible. The soft tissue in group 5 (S), however, was bonding to the adjacent cartilage and at the basis of the defect next to the subchondral bone, some hypercellular nests of chondrocytes were visible. In group 4 (VECHCA) a large number of chondrocytes were detected at the base of the defect, but otherwise the defect was poorly filled and the replacement tissue did not reach the normal surface level.
The defect in group 3 (VECA) was mainly filled with a mixture of fibrous tissue and fibrocartilage, which also did not reach the normal surface level. In group 2 (VECH), the defect was filled to approximately 2/3 with a mixture of fibrous and soft tissues. Group 1 (VE) showed more fibrous tissue than group 2 (VECH) and had some spots with an intense metachromatic staining in the defect area (Fig. 3).
Semiquantitative Evaluation
Defect
The defect part in group 6 (CA) demonstrated the best results in total score for all three scoring systems used. Whereas the differences between the groups after Mankin Score were not statistically significant, the O’Driscoll score showed a statistically clear trend (group 6 (CA) > Group 1 (VE) > group 3 (VECA) > group 2 (VECH) > group 5 (S) > group 4 (VECHCA)). Furthermore, group 6 (CA) had a significantly better overall score than group 4 (VECHCA; p=0.017). In the category “hypocellularity”, group 6 (CA) had a significantly better score than group 2 (VECH; p=0.017) and in “structural integrity” group 6 (CA) was significantly better than group 3 (VECA) and group 4 (VECHCA; p=0.042, p=0.021 respectively). “Surface regularity” showed the most statistically significant differences: group 2 (VECH) > group 5 (S) (p=0.0019) and group 4 (VECHCA; p=0.004), group 3 (VECA) > group 4 (VECHCA; p=0.0001), group 5 (S) > group 3 (VECA; p=0.002), group 6 (CA) > groups 4 (VECHCA) and 5 (S; p=0.019, p=0024, respectively). The most favourable ICRS Score was demonstrated for group 6 (CA), although it was not statistically significant.. The following statistically significant differences were recorded for the ICRS score: In the category “cell population viability” group 2 (VECH) showed inferior scores to groups 1 (VE), 3 (VECA) and 4 (VECHCA) (all p=0.002). Group 2 (VECH) was overall significantly inferior to groups 1 (VE; p=0.002) and 3 (VECA; p=0.024). Whereas the overall total Mankin score showed no differences between the transcutaneously treated groups, we found a clearly better trend for group 6 (CA) (Tables 1-4).
Comparison between the Mankin and ICRS scores indicated a Pearson’s correlation of -0.370, which came close to attaining statistical significance (p=0.053) (Fig. 4).
DISCUSSION
In the present study, we tested the hypothesis that adjacent cartilage and/or a cartilage defect can be influenced positively for healing by transcutaneous treatment with cartilage preserving ingredients. To do so, four different scoring systems were used and it was demonstrated that the adjacent cartilage in the transcutaneously-treated group with VECHCA (group 4) obtained better histological scores than all the other groups. Although not statistically significant, except for the Little score, a clear trend was observed for different variables. In particular, the metachromatic staining was better maintained in group 4 (VECHCA). This indicated that the extracellular matrix in this group was better preserved compared to the other treatment groups. Furthermore, the chondrocyte clusters found were small and consisted mostly of duplets or triplets. Clusters are generally regarded as being a sign of cartilage degeneration, but can also represent an indicator of attempted cartilage repair through chondrocyte proliferation [12, 24]. Therefore, only big cell nests and a decrease in metachromatic staining were considered as negative indicators.
The ingredients used in the transcutaneous treatment group 4 (VECHCA) were vehicle, chito-oligosaccharids and carprofen. Chito-oligosaccharids are derivates of chitosan, which has recently gained popularity with cartilage research as it proved to be suitable for improving cartilage repair as scaffold material [25-33]. Chitosan consists of glucosamine and N-acetylglucosamine monomers and has many interesting properties in a wide range of biomedical applications. Besides its biocompatible and biodegradable properties it has a good affinity for proteins, displays haemostatic characteristics due to its ability to aggregate erythrocytes and activate platelets [34, 35], enhances bacterial resistance and also improves wound-healing activities. Ueno et al. [36] observed that chitosan derivatives have a stimulatory effect on macrophages and are also chemoattractants for neutrophils. Furthermore, they stimulate granulation tissue formation due to growth factor accumulation. Fukui et al. [37] raised the hypothesis that skin and cartilage have many features in common and they proposed to consider elements of techniques used in wound healing, as also being present in cartilage repair. The use of chitosan in cartilage repair certainly supports this idea. The only constraint in biomedical application of chitosan is that it is insoluble under physiological pH conditions. Hydrolyzed products such as chito-oligosaccharids, have a lower viscosity and have a better solubility at neutral pH due to their shorter chain lengths and free amino groups in D-glucosamine units [38]. This made chito-oligosaccharids interesting for many researchers.
Our results suggest that the chito-oligosaccharids included in our treatment mixture helped to sustain the adjacent cartilage. As the same good histological results were missing in animals treated with vehicle and chito-oligosaccharids only, we assume that the combination of vehicle and chito-oligosaccharids with carprofen led to this better outcome. Carprofen is a widely used non- steroidal anti-inflammatory drug (NSAID) in veterinary medicine and has strong analgesic, antipyretic and antiphlogistic effects [39-41]. Apart from inhibiting the enzyme cyclooxygenase (COX), Benton et al. [42] discovered that carprofen also stimulated the rate of glucosaminoglycan production in dogs and therefore had a positive influence on the synthesis of the chondral extracellular matrix. Nevertheless, a concentration higher than 20μg/ml had a negative effect on the glycosaminoglycan synthesis. We hypothesize that this could be the reason why animals treated intravenously with only carprofen, had the worst histological scoring outcome in the adjacent cartilage. The high amount of carprofen achieved in the joint in our study may have inhibited the glucosamino-glycan synthesis. Although the synovial carprofen concentration measured (Fouché et al., 2012) was much lower than 20 μg/ml, the histological scores were not as good as in group 4 (VECHCA). This may indicate that the combination of chito-oligosaccharids and carprofen give rise to the better histological outcome since the two ingredients seem to have a positive impact on each other. However, the meaning of concentrations and combination of ingredients needs to be further explored.
An observation period of 12 weeks was chosen in the current study. We therefore neither expected a complete healing of the defect site nor did we intend it. The surgical experimental design aimed only to provide and set up the cartilage defect with the best possible intrinsic healing properties. The focus was set on the early alterations of the adjacent cartilage after different transcutaneous pharmaceutical treatment. However, the defect itself was also examined in order to obtain a more complete impression of the individual groups and to detect potential relationships between adjacent cartilage and the defect part. Therefore, every section was divided into 5 parts and scored separately with 4 different histological scores to get as much information as possible about the individual sections.
Today, many different scoring systems in cartilage repair exist. Some were validated and others were aligned. But there is still no general consensus about which scores should be applied in a specific experimental or clinical setup [43]. This means that so far there is no existing valid grading system acknowledged as the gold standard [44]. The Mankin score is often used to score osteoarthritic cartilage in human beings. The score was developed to investigate advanced osteoarthritis and is therefore only restrictively applicable for mild and early phases of osteoarthritis [45]. Furthermore, whereas Van der Sluijs et al. [46] validated the reliability of the Mankin score as good, Custers et al. [45] declared that the inter- and intraobserver variability of this system is rather high. Nevertheless, Ostergaard et al. [44] stated that although the Mankin score has some weaknesses, it is still valuable for systematic assessment of articular hyaline cartilage. Additionally, the Mankin score was applied to histological evaluations for many years due to missing alternatives, and therefore its outcome is well known [45]. Based on these criteria, we included the Mankin score in our evaluations.
The ICRS score was additionally chosen because it incorporates additional features. Whereas the Mankin score has only one category for cell scoring, the ICRS includes one about cell distribution and another about cell viability. Furthermore, the ICRS score also provides special categories for the subchondral bone and the calcified cartilage. However, the ICRS score does not allow for evaluation of staining alterations (metachromasia) or detailed comparison of the structure as does the Mankin score. Therefore, combining both scoring systems provided more detailed information. Besides, Moussavi-Harami et al. [47] compared the ICRS to the Mankin score and demonstrated a uniform relationship. Comparisons of the Mankin and ICRS scores in the margin parts I and II in the current study demonstrated a good relationship whereas the comparisons of the respective two scores in the defect part did not. This could be due to the fact that both, Mankin and ICRS scores were developed to score osteoarthritic cartilage and not to score repaired cartilage after experimentally created articular defects. While the margin part is reflecting more an osteoarthritic cartilage the defect part clearly does not.
The Little score was selected because it includes all features of the Mankin and the ICRS score in one scoring system.
Finally, the O’ Driscoll score [21] was the first score developed specially for cartilage repair and it is recommended for assessing an in vivo repaired cartilage defect [48]. This score is very specific for cartilage repair and includes the integration of the repair tissue into the adjacent cartilage. Therefore, the O’Driscoll score could only be applied within the defect part itself.
When examining our results, it becomes apparent that none of the scoring systems led to the same grading sequence of the individual groups. Interestingly, there was a clear trend in all score systems for best results in group 4 (VECHCA) for adjacent cartilage and for group 6 (CA) for the defect part. As this trend was observed in every scoring system it seems reasonable to rely on it. However, when looking at the other groups,, the overall picture was not as clear as expected, since between groups 1 (VE), 2 (VECH), 3 (VECA) and 5 (S) the different scoring systems revealed dissimilar results. Therefore, an explicit conclusion may be only found for groups 4 (VECHCA) and 6 (CA) and leaving the results for the other groups ambiguous. The reasons for these different outcomes in the various scorings are probably multifaceted: long lasting experience and both, the reliability and the validity of the semi-qualitative scores may be diversely assessed by investigators and be subjective [20, 44, 46-51]. In addition, focus was placed specifically on the adjacent cartilage. So far, this has not been very common and thus more general histological grading scores are developed to score either osteoarthritic, tissue engineered or repaired cartilage defect. They may not be simply transferable to our specific case. Nevertheless, we chose common histological grading systems because we assumed more objective and acceptable results than with an own, newly created scoring scale. On the other hand, sections were divided into different parts to maximise standardization. Nevertheless, it could be shown with all 4 score systems that with the transcutaneous application of cartilage protecting medication, the adjacent cartilage of a defect could be better preserved.
The significance of this study for clinical applications is such that the transcutaneous treatment system could be an alternative or additional treatment option for patients suffering from a cartilage defect. As the transcutaneously applied ingredients seem to sustain the viability of the adjacent cartilage, the immediate environment of the graft-host interface and the integration of bone marrow derived repair tissue, namely the native cartilage, could be improved. In addition, the transcutaneous treatment system is in accordnace with high patient compliance since the treatment is free of pain.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
We would like to thank Kommission für Technik und Innovation (KTI), who supported this project (#10212.1 PFLS-LS). Furthermore, we thank Meddrop Technology AG, Brandschaft 6 CH-8512 Thundorf, Switzerland for the free of charge supply of the material and substances. We are grateful to Dr. Katja Nuss and Dr. Karina Klein for surgical assistance, Dr. Stefan Stübinger for taking excellent pictures.


