All published articles of this journal are available on ScienceDirect.
Desmoplastic Fibroma: A Case Report with Three Years of Clinical and Radiographic Observation and Review of the Literature
Abstract
Background:
Desmoplastic fibroma (DF) is an extremely rare locally aggressive bone tumor with an incidence of 0.11% of all primary bone tumors. The typical clinical presentation is pain and swelling above the affected area. The most common sites of involvement are the mandible and the metaphysis of long bones. Histologically and biologically, desmoplastic fibroma mimics extra-abdominal desmoid tumor of soft tissue.
Case Presentation and Literature Review:
A case of a 27-year old man with DF in the ilium, including the clinical, radiological and histological findings over a 4-year period is presented here. CT scans performed in 3-year intervals prior to surgical intervention were compared with respect to tumor extension and cortical breakthrough. The patient was treated with curettage and grafting based on anatomical considerations. Follow-up CT scans over 18-months are also documented here. Additionally, a review and analysis of 271 cases including the presented case with particular emphasis on imaging patterns in MRI and CT as well as treatment modalities and outcomes are presented.
Conclusion:
In patients with desmoplastic fibroma, CT is the preferred imaging technique for both the diagnosis of intraosseus tumor extension and assessment of cortical involvement, whereas MRI is favored for the assessment of extraosseus tumor growth and preoperative planning. While tumor resection remains the preferred treatment for DF, curettage and grafting prove to be an acceptable alternative treatment modality with close follow-up when resection is not possible. Curettage and grafting have been shown to provide good clinical results and are associated with long recurrence free intervals.
1. INTRODUCTION
Desmoplastic fibroma (DF) is a benign intraosseus neoplasia with locally aggressive and infiltrative characteristics. DF was first described by Jaffe in 1958 [1]. In 2002 DF was defined by the WHO as a rare benign bone tumor composed of spindle-shaped cells with minimal cytological atypia and abundant collagen production [2]. DF represents one of the most uncommon bone diseases as it has an incidence of only 0.11% of all primary bone tumors [3]. While the potential for malignant transformation in DF has been mentioned, it has not been reported in cases [4].
In 1996 Böhm et al. [5] reviewed 184 published cases of DF and in a metaanalysis reported the mean age of DF patients at the time of diagnosis as 23 years with a range from 15 months to 75 years. Sex predilection remains unclear while some authors report higher rates of DF in female patients [6] and others report no detectable sex predilection [7]. DF can theoretically affect any bone, but the majority of cases occur in the mandible (22%) and the metaphysis of long bones (56%) [8]. The pelvis accounts for 12% of cases. To the best of our knowledge 15 cases of DF in the iliac bone have been reported to date.
The clinical presentation of DF is non-specific and usually presents as pain over the affected area and occasionally as a palpable mass. DF has also been detected incidentally [9]. On a plain radiograph DF is not always easily differentiated from other benign or malignant tumors.
Because DF has a recurrence rate of 37% to 72% [5, 10], resection of the affected bone is the preferred therapy, and ideally an en bloc resection [11]. Cases in which anatomical considerations limit resection, alternative therapy has been curettage and reported to achieve recurrence free survival up to 9 years [12]. The average rate of recurrence after curettage has been reported to be 55% [5]. The imaging features of DF have been characterized in a few studies [11, 13] but to our knowledge no study has been published that presents the cross-section radiographic patterns and development of DF over a time period of three years.
This study presents the case of a 27-year old man, who complained of increasing pain in the sacroiliac joint for over 3 years. The patient has given informed consent for publication of his case.
2. CASE PRESENTATION
A 27-year old man presented to our outpatient department with increasing pain in the region of the right sacroiliac joint. The pain started three years earlier, at which point the patient sought help from a local orthopaedic surgeon. At this initial presentation it was thought that his pain was attributable to his physically demanding job. A CT-scan and MRI-scan were performed and revealed a bone cyst with sclerotic borders without destruction of the cortical bone (Figs. 1, 2). Signs of malignancy were not present. Accordingly, the patient was treated with physical therapy, chiropractic treatment, and pain control.
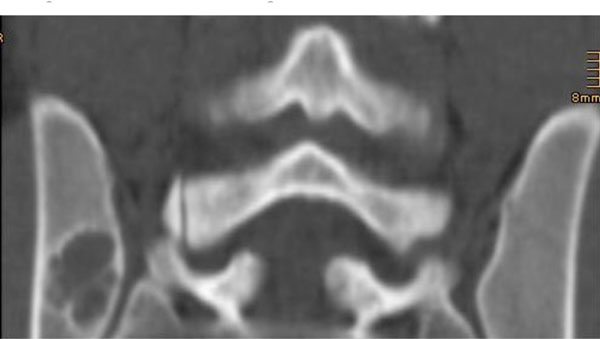
CT-scan three years preoperative. The corticalis is preserved. Within the lesion pseudo-trabeculation is visible.
T1-weighted MRI three years preoperative. Note that the lesion presents with low signal intensity and no infiltration of the surrounding tissue.
Over time his pain increased to the point of requiring opioids for pain control and presenting to our hospital. In the interim, he changed his job and decreased his physical activity, neither of which alleviated his pain.
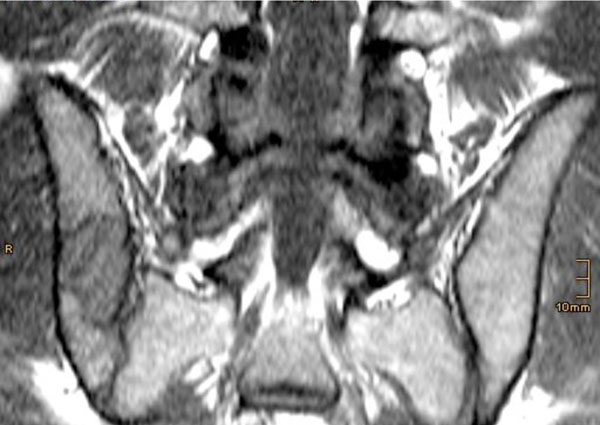
On physical examination the patient had tenderness to palpation and vibration over his right sacroiliac joint. Peripheral motor and sensory functions were intact, no pathologic reflexes were present. The patient was in a good general state of health. A CT-scan was repeated and showed a slightly enlarged bone cyst compared to the previous CT-scan three years ago. However, no cortical breakthrough was present (Fig. 3).
CT-scan 2 weeks before the operation. There is cortical breakthrough of the tumor. The size of the lesion has slightly increased in comparison the previous CT-scan.
For further diagnostic workup an open biopsy of the lesion was performed one week later. The histological examination of the tissue demonstrated the formation of abundant collagen fibers and spindle shaped cells with medium to low cell density. The nuclei were without pleomorphism and mitotic activity. In immuno-staining the nuclei of the tumor cells were positively labeled for the adhesion protein β-catenin. In addition there was a partially positive staining recognized for S-100. Morphology and immunophenotype were consistent with either aggressive fibromatosis or desmoplastic fibroma, both of which express β-catenin (Fig. 4). Because of its intraosseus location the working diagnosis of desmoplastic fibroma was made. Accordingly the indication for operative excision via curettage and grafting was established.
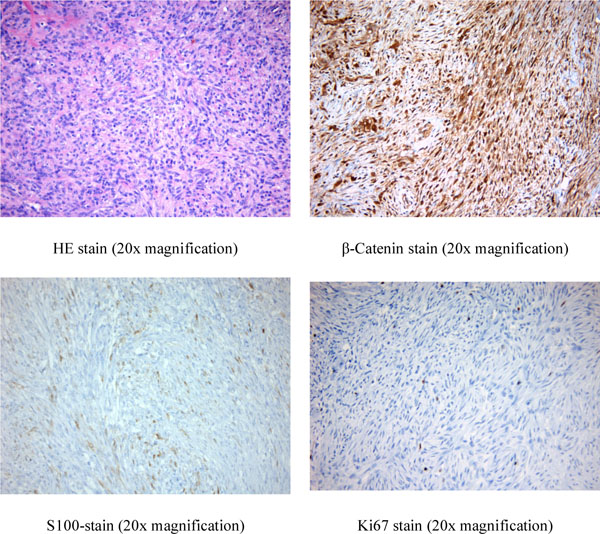
Histological examination of biopsy shows abundant collagen fibers, strong positive labelling for the adhesion protein β-Catenin, weak positive labelling for S-100 and almost no positive stain for the proliferation marker Ki67.
The operation was performed with the patient under general anaesthesia. The tumor was removed in several parts. The tissue was beige in color with hard and soft areas of consistency (Fig. 5). The removed tumor was sent for further histological examination to the department of pathology.
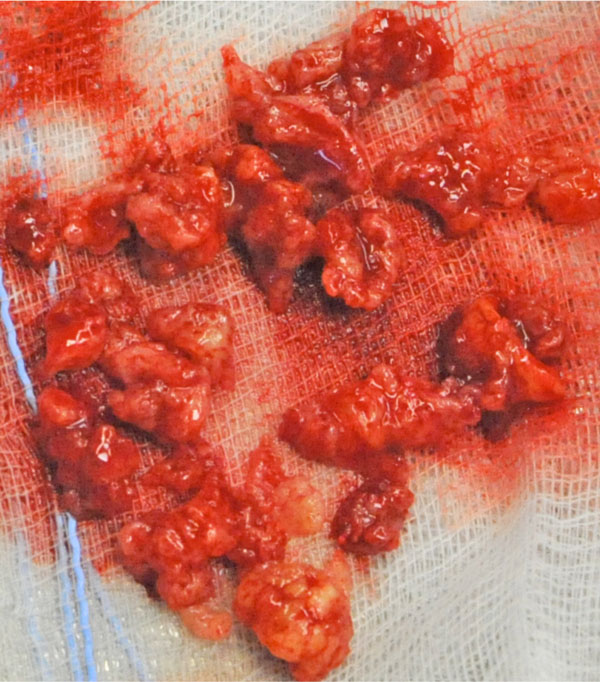
Intraoperative photograph showing the removed tissue. It has a beige color and hard as well as soft areas of consistency.
The lesion was filled with autologous bone graft from the iliac crest that was mixed with Cerasorb® (Curasan, Kleinostheim, Germany) and blood from the patient. Operating time was 105 minutes. After surgery the patient was restricted to no weight bearing on the operated side until complete wound healing. Compared to a more invasive treatment option, this therapeutic approach has the advantage of reduced operating time and earlier recovery.
No postoperative radiation of the lesion was performed to enable adequate integration of the bone graft.
Histological workup of the removed tissue demonstrated the identical picture as the biopsy and accordingly no signs of malignancy.
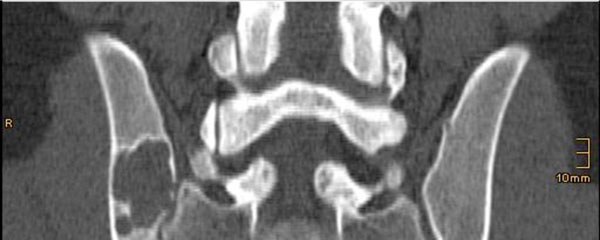
Postoperative follow-up examinations as well as CT-scans 6 and 18 months after surgery showed good integration of the graft (Figs. 6, 7). There were no signs of recurrence.
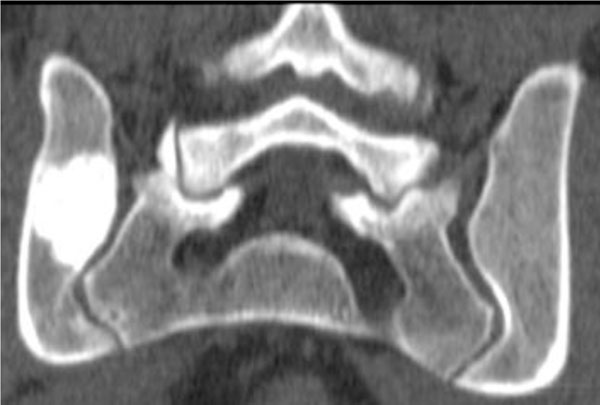
CT-scan 6 months after the operation. No residual tumor is present.
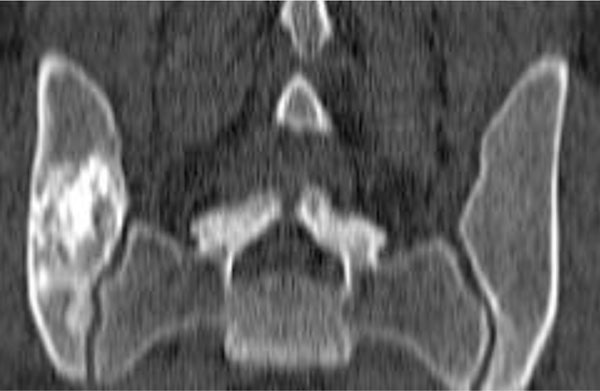
CT-scan 18 months after the operation. The bone graft is well integrated in pelvic bone. No discontinuity of the cortical bone. No recurrence of the tumor is present.
3. DISCUSSION
This case represents a typical presentation of desmoplastic fibroma clinically, radiographically and histologically.
Desmoplastic fibroma is a very rare bone tumor, affecting the mandible, long bones, and pelvis. Morphologically a distinction between desmoplastic fibroma and aggressive fibromatosis is not possible. Aggressive fibromatosis, also called desmoid tumor, was described before desmoplastic fibroma and is defined as rare mesenchymal neoplasia composed of collagenous tissue and spindle-shaped cells [14]. Desmoid tumors can be localized intraabdominally, within the abdominal wall and also extraabdominally. Most desmoid tumors are abdominal (to 69%) [14]. The intraosseus variant is the desmoplastic fibroma.
The characteristics of DF in MR and CT imaging have been well described in a case report by Stefanidis et al. [11]. The tumor reported in this case was an osteolytic lesion with destruction of the cortical bone, minor pseudotrabeculation, and marginal sclerosis. In T1-weighted sequences the tumor showed low signal intensity with signal enhancement after contrast agent administration. These findings were also present in our case. The initial CT scan three years prior to surgery shows an osteolytic lesion with marginal sclerosis and slight pseudotrabeculation. A cortical breakthrough was not present initially. In both T2-weighted and contrast enhanced T1 SPIR sequences the tumor exhibited high signal intensity.
The preoperative CT-scans showed increased tumor size compared to initial scans three years prior with cortical breakthrough in several areas, indicating bone instability. An isolated extraosseus growth was not present at any time.
Crim et al. [15], who analyzed the radiographic characteristics of DF in 83 patients from case reports, reported that only in 29% of patients cortical breakthrough was present. If cortical damage is suspected a CT-scan should be performed to better visualize the bone structure and to quantify cortical bone destruction.
If extraosseus tumor growth is suspected a MRI is preferred. MR-scans are also helpful for preoperative planning as the extent of the tumor and its relation to anatomical landmarks can be evaluated.
The diagnosis of DF is difficult by imaging studies alone. In both MR and CT imaging, many tumors resemble imaging of DF. These tumors are: fibrous dysplasia, giant cell tumor, simple bone cyst, aneurysmatic bone cyst, chondromyxofibroma, non-ossifying fibroma, eosinophilic granuloma, adamantinoma, and metastases.
The histological picture of DF is dominated by abundant collagen fibers and low density of cells. The nuclei are long and spindle shaped. Signs of mitosis are usually not present. The histological differential diagnosis includes the spindle cell tumors and tumor like lesions, including juvenile bone cyst and aneurysmatic bone cyst. The most important histological differential diagnosis is low-grade fibrosarcoma. Fibrosarcoma typically presents with high cell density, high grade of polymorphism, and a high rate of mitosis. In rare cases of low-grade fibrosarcoma mitotic activity is not present and the tissue is collagen rich with a low cell count, such that differentiation between desmoplastic fibroma and fibrosarcoma is not possible. In these cases the postoperative clinical development helps to establish the definite diagnosis.
For further diagnostical workup several immunohistochemical biomarkers are applicable to narrow down the differential diagnosis. In the presented case the tumor stained highly positive for β-catenin (Table 1), which is in accordance with the results of Hauben et al. [16], who examined the significance of β-catenin expression in DF in comparison to aggressive fibromatosis. In aggressive fibromatis β-catenin is sporadically associated with familial adenomatous polyposis coli (FAP) by a shared APC/ β-catenin pathway. This link however is not present in desmoplastic fibroma.
Immunhistochemical Marker Used to Narrow Down the Differential Diagnosis
| Antigen | Present Patient | Interpretation |
|---|---|---|
| β-Catenin | ++ | up regulated in desmoplastic fibroma [16] and aggressive fibromatosis [17] |
| S-100 | + | ubiquitary present protein (glia cells, adipocytes, melancytes, chondrocytes) |
| actin | (+) nucleus | ubiquitary present protein (mainly myocytes). Together with Desmin up regulated in leiomyosarcoma |
| KiM1P | (+) nucleus | present in histiocytes |
| MDM 2 | (+) nucleus | Together with MDM 2 and CDK4 upregulated in liposarcoma |
| CD31/CD34 | - | up regulated in angiogenesis |
| MyoD1 | - | up regulated in rhabdomysarcoma |
| myogenin | - | up regulated in rhabdomysarcoma |
| desmin | - | up regulated in leiomysarcoma |
| CDK4 | - | up regulated in liposarcoma |
| DOG1 | - | up regulated in GIST |
| Ki67 | positive in 2% of cells | up regulated in proliferating cells |
In this presented case the patient was treated with curettage and grafting. Given that DF has a recurrence rate of up to 72% after non-resection procedures (e.g. curettage) and 17% after resection (Table 2), the literature advocates resection (marginal or wide) as the treatment of choice [5]. Alternatively Nishida et al. [12] reported about 5 cases that were treated with curettage and did not show any sign of recurrence for the follow-up period of 5.5 to 9 years. In 2 of the 5 patients the pelvis was affected by DF. Similar to those cases, no isolated extraosseus growth of tumor was present in our case. This characteristic along with slow tumor growth, documented by CT-scans for 3 years, were determining factors to keep the surgical therapy as less invasive as possible.
Treatment of DF and Corresponding Rate of Recurrence
| Primary Therapy | Number of Cases | Number of Recurrences | Percentage of Recurrence |
|---|---|---|---|
| Biopsy | 3 | 2 | 66% |
| Curettage | 52 | 27 | 52% |
| Curettage and radiotherapy | 2 | 0 | 0% |
| Excision | 20 | 13 | 65% |
| Resection | 48 | 4 | 8% |
| Amputation | 3 | 0 | 0% |
| Cryosurgery | 1 | 0 | 0% |
| Total | 129 | 46 | 37% |
In 2011, a case was published by Stefanidis et al. [11], reporting the clinical and radiographic development of DF located in the ilium with a follow-up period of 6 years. The tumor was removed by curettage and replaced by bone graft. The patient was free of recurrence for 6 years.
Another case of DF in the ilium by Gebhardt et al. [10] was reported with a wider extension of tumor growth and involved the acetabulum, pubic bone and ischium. This was also a case of extraosseus tumor expansion. Isolated extraosseus filiae, however, were not present. The treatment in this case consisted of a hemipelvectomy and reconstruction using a cadaver hemipelvic allograft. As reported by Sanfilippo et al. and Nag et al. [18, 19] radiotherapy has also been shown to be a therapeutic option. Sanfiliippo et al. [19] treated one patient who had DF in the ilium with radiotherapy of 60 Gy in 30 fractions. Similarily, Nag et al. [18] treated one patient with DF in the distal end of the femur with radiotherapy of 45 Gy in 25 fractions. Both authors reported deceleration of disease progression over a time period of 30 and 36 months, respectively. Even though a decrease in pain sensations was achieved in both patients, radiotherapy did not lead to complete pain relief.
To date, 271 cases of DF, including the presented case have been published in the literature. Of these 270 cases, 30 (11%) were localized in the pelvis and 15 (50%/ 5,5%) were localized in the ilium (Fig. 8). In 6 of the 15 ilium involved cases therapy was curettage, in 5 cases it was resection, in 1 case radiotherapy was the definitive treatment [19], in 2 cases the therapy is not known [20, 21] and in one case no operation or other treatment was performed [22]. In 2 of the 15 cases a recurrence has been observed. These 2 patients were treated with curettage (Table 3). Compared to other regions where DF is common (e.g. mandible, humerus, femur) the treatment options in the ilium are more limited. Partial resection of the pelvis with subsequent reconstruction can lead to prolonged immobilization and persistence of pain [10]. Based on this review of the literature and on the experience with the presented case the authors recommend the following approach for the treatment of DF of the ilium: If no isolated extraosseus tumor growth is present and the stability of the pelvis is intact, curettage and grafting is a possible treatment modality. This modality should be offered to young and active patients, who comply with the requirement of yearly postoperative follow-up exams. In other cases resection and reconstruction seems to be the preferred treatment as it has a lower rate of recurrence.
Summary of Cases Reported About DF in the Ilium; NED: No Evidence of Recurrent Disease
| Reference | Age/Gender | Site | Radiographic Pattern | History | Initial Treatment | Follow-Up Period | Comment |
|---|---|---|---|---|---|---|---|
| Rabhan [24] | 22y/M | Right | Initial diagnosis: fibrous dysplasia | 2 months history of pain w/o hx of trauma | wide local excision | < 1y | NED |
| 10y/M | Right | Initial diagnosis: Legg-Perthes disease | 3-4 months of pain w/o hx of trauma | Curretage and graft | 7ys | Recurrence, wide local excision | |
| 24y/M | Right | Initial diagnosis: aneurysmal bone cyst | 4,5 years of pain in right groin | Curretage and graft | 4ys | Recurrence, second curretage and graft | |
| Whitesides [25] | 8y/M | Right | initial diagnosis: aneurysmal bone cyst or hemingioma | 4 months of pain. Limp for 3 weeks | Curretage and graft | 21 months | NED |
| Nilsonne [26] | 30y/F | Right | Initial diagnosis: metastasis of uterine cancer | 8 months of pain | Curretage | 12ys | NED |
| Stadler [27] | 15y/M | Left | N/A | Three weeks of pain | Curretage | N/A | N/A |
| Gebhardt [10] | 28y/M | Right | N/A | 6ys of intermittend pain and weakness | Marginal resection with hemipelvic allograft | 5,5ys | NED |
| Goldman [28] | 23y/M | Right | N/A | 8 months of pain in right hip w/o trauma | en bloc resection | N/A | N/A |
| Inwards [20] | N/A | N/A | N/A | N/A | observation | 11y | slow progression |
| N/A | N/A | N/A | N/A | N/A | N/A | N/A | |
| Nishida [12] | 35y/M | Left | Benign bone tumor | N/A | Curretage and graft | 9ys | NED |
| Zhang [29] | 49y/F | Right | Benign bone tumor | mild tenderness | Wide resection | N/A | N/A |
| Stefanidis [11] | 56y/M | Left | Cortical breakthrough on CT-scan | Lumbar pain. No hx of trauma | Curettage and graft | 6ys | NED |
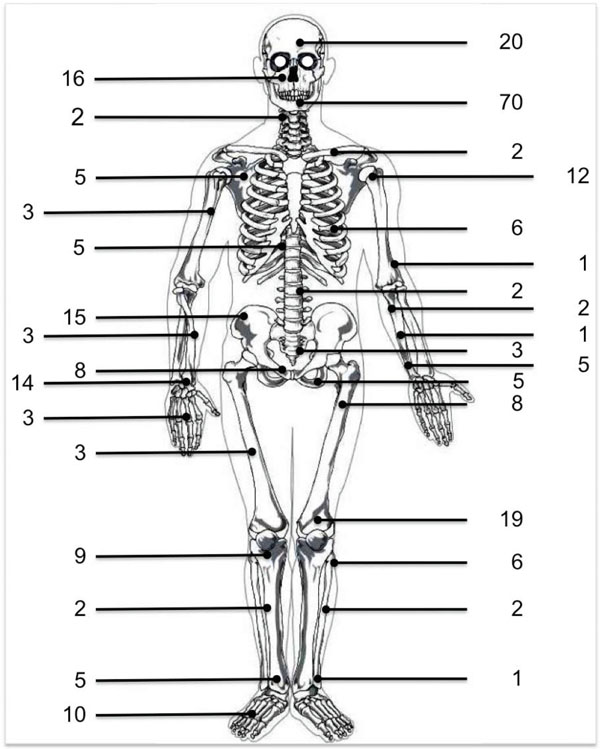
Distribution of desmoplastic fibroma in 266 patients.
Of note there seems to be a predilection for male patients being affected by DF in the ilium (male: 9, female: 2) while female patients seem to be more affected by DF in the jaw [23].
In conclusion, the therapy of DF is strongly dependent on the region of the body that it affects and on the aggressive nature of the lesion. If a resection is feasible, as it is in the mostly affected bones (mandible and long bones), it should be performed, as resection has the lowest rate of recurrence [5]. In isolated intraosseus lesions without evidence of extension in adjacent soft tissue and when resection is associated with a higher risk because of anatomical conditions, a considerate risk-benefit-analysis should be carried out and thorough curettage may be an adequate management as it reduces operation time with a lower risk of infection and facilitates faster recovery. Once the decision has been made in accordance with the patient to perform a curettage, close postoperative observation, including clinical and radiographic examinations, is necessary to detect a recurrent lesion as early as possible. The higher risk of recurrence has to be well communicated with the patient preoperatively and has to be included in the decision finding process.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflicts of interest.
ACKNOWLEDGEMENTS
This publication was funded by the German Research Foundation (DFG) and the University of Wuerzburg in the funding programme Open Access Publishing.


