All published articles of this journal are available on ScienceDirect.
Low Level Laser Therapy (LLLT) for Neck Pain: A Systematic Review and Meta-Regression
Abstract
Purpose:
This systematic review update evaluated low level laser therapy (LLLT) for adults with neck pain.
Methods:
Computerized searches (root up to Feb 2012) included pain, function/disability, quality of life (QoL) and global perceived effect (GPE). GRADE, effect-sizes, heterogeneity and meta-regression were assessed.
Results:
Of 17 trials, 10 demonstrated high risk of bias. For chronic neck pain, there was moderate quality evidence (2 trials, 109 participants) supporting LLLT over placebo to improve pain/disability/QoL/GPE up to intermediate-term (IT). For acute radiculopathy, cervical osteoarthritis or acute neck pain, low quality evidence suggested LLLT improves ST pain/function/QoL over a placebo. For chronic myofascial neck pain (5 trials, 188 participants), evidence was conflicting; a meta-regression of heterogeneous trials suggests super-pulsed LLLT increases the chance of a successful pain outcome.
Conclusions:
We found diverse evidence using LLLT for neck pain. LLLT may be beneficial for chronic neck pain/function/QoL. Larger long-term dosage trials are needed.
BACKGROUND
Description of the Condition
Neck pain can be classified as simple "non-specific" neck pain (i.e. sprain/strain) described as pain without specific identifiable etiology and “specific” neck pain with identifiable etiology (i.e. radiculopathy) [1, 2]. According to the Bone and Joint Decade 2000–2010 Task Force, the incidence of neck pain was 150 to 200 per 1000 cases per year, the annual worldwide prevalence varied from 12.1% to 71.5% and neck pain limiting activities was 11.5% [3, 4]. Neck pain is costly to the patient and society. In Quebec, annual prevalence of neck pain in the working population is close to 43% in men and 54% in women [5]. Disabling neck pain is in 10% of men and 18% of women in this population. Over 11% of Ontario workers claimed lost-time benefits due to neck pain [6].
Description of the Intervention
Various treatment strategies including low level laser therapy (LLLT) are used to treat neck pain [6, 7]. The term Laser is an acronym for light amplification by stimulated emission of radiation-a form of photonic therapythat is defined by the following characteristics: collimation – it has little beam divergence over distance; convergence – the light waves are all in phase; and monochromicity – it has a single or narrow band of a particular wavelength of light [8]. Proponents of LLLT note laser devices are either high power or low power. High power laser devices, having a thermal effect, destroy tissue and are used during surgical procedures and for thermolysis. Low power laser devices have little to no thermal effects, have a stimulative effect on target tissues and are used to treat an array of musculoskeletal conditions to decrease pain and inflammation, stimulate collagen metabolism and wound healing, and promote fracture healing [8-10].
Lasers used therapeutically emit relatively low light energy [from a few milliwatts (mW) to 100 to 200 mW] for short periods of time (seconds to minutes) and produces insignificant changes in tissue temperature (measured to be around 1.0 °C). As such, this type of laser is often referred to as LLLT or photomodulation. The wavelength of the light emitted from lasers varies from >100 to >10000 nanometer (nm) in the electromagnetic spectrum [8] so only wavelength above 193 are transmitted in the atmosphere. Lasers used to stimulate biological tissues were historically produced using a Helium-Neon (HeNe) gas mixture. Light is attenuated exponentially in tissue and the physical penetration depth is given by the distance over with the initial power or energy density dropped to 1/e or ~37% of its original value. The depth over which a sufficient dose can be delivered comprises multiple physical penetration depths and is more commonly quoted in the literature. Here we adopt the clinical usage of this term as the depth to which clinical affects can be achieved. HeNe has a wavelength output of 632.8 nm that is visible red light, is continuous and can penetrate 0.8 mm into tissue with indirect effects of up to15 mm [8]. Currently low level laser devices are commonly produced from semiconductor diodes composed of crystal compounds such as Gallium-Arsenide (GaAs) or Galium-Aluminum-Arsenide (GaAlAs), designed to emit laser energy at various specific wavelengths in the infrared range of the electromagnetic spectrum (730nm to 905nm). The infrared (IR)-laser light, GaAs, laser can penetrate up to approximately 5 cm into tissue with a wavelength of 904 nm and is pulsed [8]. The IR-laser, GaAlAs, laser has a wavelength of 830 nm [11, 12], is pulsed, and can penetrate approximately 2 to 3 cm into the tissue [10]. Hence, lasers with longer wavelengths penetrate deeper into the skin tissue than lasers with shorter wavelengths. There is experimental evidence to suggest that the biological effects and physical behaviour of lasers vary with the wavelength of light used [13-15]. The wavelength of red light has been consistently shown to biostimulate cellular responses including membrane permeability, intracellular calcium influx, and ATP production [14-16]. Laser driver technology considers the delivery of the therapeutic dose (J or J/cm2) either with a constant time average or as a pulse light source with low duty cycle but very high dose rate. The pulsed delivery of light allows higher dose-rates to reach deeper tissues, particularly for very short pulsed and low repetition rates. For example, a 905 nm continuous wave infrared laser allows 2.5 cm penetration of a clinically effective dose-rate, while a 905 nm super-pulsed infrared laser allows the same dose-rate even at a10 cm depth. Super-pulsed infrared laser allows high peak power (50 W) to be delivered in bursts of very short duration (200 nanoseconds). These brief pulses of light energy are delivered at frequencies of up to 10 kHz. Thus superficial tissues will not heat up due to the very short bursts. This (high peak power of short duration and high frequency) allows a therapeutic dose-rate to reach deep tissues. It is however important to note that the dose is not effected by pulsed delivery, only its rate of delivery during the actual emissions cycle of the laser. Dual channel lasers can combine both continuous and pulsed lasers to allow superficial and deep dose-rate delivery of laser energy. Thus laser drive technology allows penetration to deep tissues or more superficial tissue promoting acceleration of healing by reducing pain and inflammation while staying below the Maximal Permissible Exposure tolerance for tissue. Because of the relative ease of producing semiconductor diodes and the relative ability of infrared light to penetrate biological tissues, infrared lasers (GaAs; GaAlAs) are most often used clinically to treat musculoskeletal conditions involving structures located deep within the joint. Dosage of a laser treatment is calculated using the power output [milliwatts (mW)], the surface area of the laser beam (cm2) and the amount of time the laser beam is in contact with the skin (seconds) [8]. The wavelength of the laser device (nm) determines the quantum energy available for photochemical processes during laser exposure. Laser energy density is measured in joules per square centimeters (J/cm2) of tissue area and laser power emitted is expressed in mW.
How the Intervention Might Work
The degree of biological reaction to decrease pain and inflammation, stimulate collagen metabolism and wound healing and promote fracture healing is believed to depend on a number of factors including power density (W/cm2), the wavelength (nm), the energy density (J/cm2), and site of application [17,18]. The three main mechanisms by which laser produces analgesic effects [19, 20] are believed to be: stimulating endogenous opioid release, elevating pain thresholds [21], and modifying the release of noxious mediators such as bradykinin [22] and histamine [23]. Pain modulation may also occur due to changes in nerve conduction velocity. There have been reports noting that administering laser to nerves resulted in altering the action potential conduction velocity [23] however, several other reports have not demonstrated a clinically important effect of laser on nerve conduction velocity [24, 25]. In addition, there have been some exciting experimental reports which suggest that laser therapy may improve recovery following nerve trauma [26]. Laser therapy has also been found to have an effect on peripheral motor nerve healing [8, 27]. One randomized controlled trial (RCT) assessing HeNe laser on rats with crush injuries found that laser treatment lead to an increased amplitude and velocity of action potentials along the injured motor nerve and an increase in the speed of nerve healing [28]. In summary, LLLT slows down the transmission of pain signals through the autonomic nervous system, regulates serotonin and norepinephrine, and increases the pain threshold [8, 23, 27].
LLLT is also used for inflammation, oedema, swelling, and tissue healing. LLLT application is believed to limit the release of inflammatory mediators, such as bradykinin and histamine, decreasing the inflammatory response [8, 22, 23]. However, it has been strongly hypothesized that a decrease in prostaglandin activity during the inflammatory process is the main anti-inflammatory effect of laser stimulation. Prostaglandins cause vasodilation at the site of inflammation, facilitating infiltration of inflammatory cells to the surrounding tissue. Studies have shown that a decrease in prostaglandin activity due to laser stimulation may promote healing [8, 29].
LLLT stimulate collagen metabolism, wound healing [30], and promotes fracture healing [8-10]. Researchers have also found an increase in collagen and elastic fibers in injured tissue post-laser treatment in animal studies [28]. Similar results were found in other studies [24, 25].
For which clinical outcomes does it work? Laser has been indicated to manage pain associated with many conditions [31] including trigeminal and post-herpetic neuralgia [32, 33], carpal tunnel syndrome [34], fibromyalgia [35], tendonitis [36], osteoarthritis [37], and rheumatoid arthritis [38]. Little information exists regarding its influence on function.
Why it is Important to Do this Review
Fourteen reviews published between 2005 and 2011 included LLLT either as the main intervention, as the comparator treatment or as part of a multi-modal intervention had a low AMSTAR ratings ranging from 4 to 8 of a possible 11 total score. Three were of higher quality (AMSTAR > 6) (Gross 2007 [39]– 8, Leaver 2010 [40] – 7, Chow 2009 [41] – 8). There are conflicting reports from these reviews. Our previous review found evidence to support the use of LLLT for pain reduction and functional improvement in the intermediate term for acute/subacute and chronic mechanical neck disorder (MND)/degenerative changes [39, 41]. Leaver et al. show unclear evidence for the effectiveness of LLLT compared to control groups [40,41]. A systematic review of 16 RCTs showed that LLLT reduces pain from immediate post-treatment in acute neck pain and up to 22 weeks following completion of treatment in those with chronic neck pain [41]. However, the clinical heterogeneity of the pooled trials in this review by Chow and colleagues was debated due to varied dosage [41]. In our current update, we utilized a meta-regression to explore this latter issue and categorized findings by specific subgroups to enhance clinical applicability and generalizability.
Objective
This systematic review update assessed the effect of LLLT on pain, function, patient satisfaction, quality of life, and global perceived effect in adults suffering from mechanical neck pain with or without cervicogenic headache or radiculopathy. Where appropriate, the influence of risk of bias, duration of the disorder and subtypes of neck disorder on the treatment effect was assessed. A meta-regression explored key dosage factors set a priori.
METHODS
Criteria for Considering Studies for this Review
Our criteria and methodology were consistent with our published protocol for our Cochrane reviews [42, 43] and followed the Cochrane Handbook [44] or PRISMA Guidelines [45]. A protocol specific to this review was not published or registered.
Types of Studies
We included any published and unpublished RCTs in any language.
Types of Participants
The subjects of included studies were adults (18 years of age or older) with acute (less than 30 days), sub-acute (30 to 90 days) or chronic (longer than 90 days) neck pain categorized as simple non-specific mechanical neck pain including sprains and strains [46], neck pain associated with myofascial pain syndrome (MPS) and degenerative changes [47], cervicogenic headache [48], whiplash [49, 50], and radiculopathy [48].
We excluded studies that addressed neck disorders with definite or possible long tract (upper motor neuron) signs; with neck pain caused by other pathological entities (i.e. systemic diseases, infections, fractures or grade IV neck pain); with headache not of cervical origin, but associated with the neck (i.e. migraine, tension-type headache) [48].
Types of Interventions
Studies using LLLT alone or in combination with other interventions were included. Acceptable comparison groups were placebo, another intervention (i.e. exercise), or other treatment added to both arms of the trial (i.e. LLLT plus exercise versus sham LLLT plus exercise). The comparisons were arranged in the results first by control intervention and then by comparison intervention for disorder subtypes.
Types of Outcome Measures
The outcomes of primary interest were pain intensity, function, and disability. Quality of life, global perceived effect and patient satisfaction were also investigated. Periods of follow-up were immediately post treatment (about one day); short-term (closest to three months); intermediate-term (closest to 6 months); and long-term (closest to 12 months).
Search Methods for Identification of Studies
We screened citation titles and abstracts using pre-piloted forms and two independent authors. We did an updated search for the following computerized databases, without language restrictions from 2006 up to Feb 2012: MEDLINE, EMBASE, Manual Alternative and Natural Therapy, Cumulative Index to Nursing and Allied Health Literature, Index to Chiropractic Literature, and CENTRAL (Cochrane Library Issue 2, 2010) (See APPENDIX A for MEDLINE search). We included the following MeSH headings and key words for physical medicine methods: phototherapy, lasers, physical therapy, combined modality therapy, exercise, exercise therapy, rehabilitation, low level laser therapy, and neck pain. We also screened reference lists, identified content experts and searched select conference proceedings for grey literature.
Data Collection and Analysis
Standard mean difference (SMD) and relative risks (RR) with 95% confidence intervals (CI) were calculated using a random effects model. For continuous outcomes reported as medians, we calculated effect sizes based on Kendall [(p 237)] [51]. In addition, we calculated number needed to treat (NNT) and the percent treatment advantage for pain; this is the difference between the changes in the treatment and control groups divided by their respective baselines.
Selection of Studies
Two independent reviewers selected articles for inclusion and data extraction; quadratic weighted kappa (Kw) [52] was used to assess agreement. Disagreement was resolved through discussion. Study authors were contacted for clarification when needed. Selection of articles in languages other than English was performed with the assistance of a translator with a medical, science or research background.
Assessment of Risk of Bias
The Cervical Overview Group used a team of assessors with at least two assessors who independently assessed each study using the 12 criteria for risk of bias (Fig. 4). The quadratic weighted Kappa (Kw) statistic was used to assess agreement on risk of bias assessment (Κw 0.23 to 1.00). Risk of bias was discussed by the broader validity assessment team to maximize inter-rater reliability [53]. A low risk of bias is defined as meeting six or more criteria.
Measures of Treatment Effect
For the purpose of this review, we utilized a 10-point change on a 100-point pain intensity scale [small: weighted mean difference (WMD) < 10%; moderate: 10% ≤ WMD < 20%; large: 20% ≤ WMD of the VAS scale] to be a minimal clinically important difference between treatments. Additionally, we considered a difference of five neck disability index units (10%) to be the minimal clinically important difference for the neck disability index [54]. In the absence of clear guidelines on the size of a clinically important effect size [measured as Standard Mean Difference (SMD)], we used a commonly applied system by Cohen; small (0.20), medium (0.50) and large (0.80) [55].
Assessment of Heterogeneity
Before calculating a pooled effect measure, we assessed the reasonableness of pooling on clinical and biological grounds and assessed statistical heterogeneity (Cochrane Q, p< 0.01; I2> 0.40).
Data Synthesis
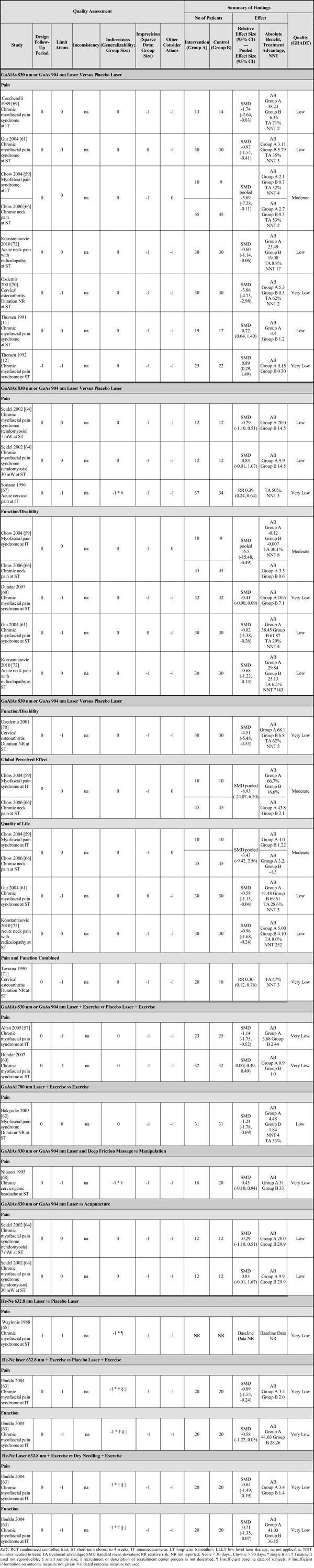
Two of our reviewers independently examined the quality of the evidence using the GRADE (Grading of Recommendations Assessment, Development and Evaluation) criteria recommended by Cochrane Collaboration [44, 56]. Domains considered in this assessment were: 1) the study design, 2) risk of bias, 3) consistency of results, 4) directness (generalizability), 5) precision (sufficient data), and 6) other considerations (i.e. publication bias). The studies were classified according to standardized published grade criteria (See Table 1) [57].
The Quality of Evidence Utilizing the GRADE Approach are Defined
| GRADE | Definition |
|---|---|
| High | Further research is very unlikely to change our confidence in the estimate of effect (all of the domains are met). |
| Moderate | Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate (one of the domains is not met). |
| Low | Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate (two of the domains are not met). |
| Very Low | We are very uncertain about the estimate (three or more of the domains are not met). |
Subgroup Analysis
Data are presented by categories of disorder subtype and duration of disorder. Subgroup analysis was explored using the funnel plot when data were available (Fig. 1).

Funnel plot for subgroup analysis.
Sensitivity Analysis
Sensitivity analyses and meta-regression were planned for variables identified a priori: methodological quality, subtype disorder, duration of symptoms, and dosage. One role of meta-regression is to explain the heterogeneity in terms of study-level covariates. Variables with a significance level < 0.10 were retained as potential prediction factors. The significance level was selected to increase the likelihood that no potential prediction variable would be overlooked. Four dosage variables were identified by team consensus [two physiotherapists (1 PhD, 1 clinician), a chiropractor (clinician), aphysicist (PhD), an engineer] a priori as being of greater clinical relevance [energy density (J/cm2), dosage per session (J), dosage per treatment program (J) and drive technology (applied technology of delivering the energy of the lasers – pulsed vs continuous)], and were entered into the regression model to determine the best set of variables to predict treatment success. It is important to note that if drive technology is a confounding factor that the total density as well as the power density (or the rate of delivering the ‘dose’) are outcome determining parameters. The MCID for pain was used to define treatment success. These variables were abstracted and reported for each trial in Table 3 – Laser Dosage and Clinical Characteristics.
Clinical Applicability
Two reviewers independently assessed all articles with a clinical applicability checklist (agreement varied from 83 to 100%; Fig. 2).
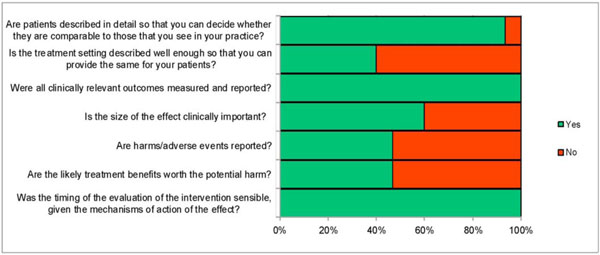
Clinical applicability across all LLLT trials for neck pain.
RESULTS
Results of the Search
For this update, we selected 17 studies from 110 citation postings related to laser from 2171 identified citations for physical medicine methods search strategy [Kw 0.85 (95%CI: 0.69 to 1.00)] (Fig. 3).
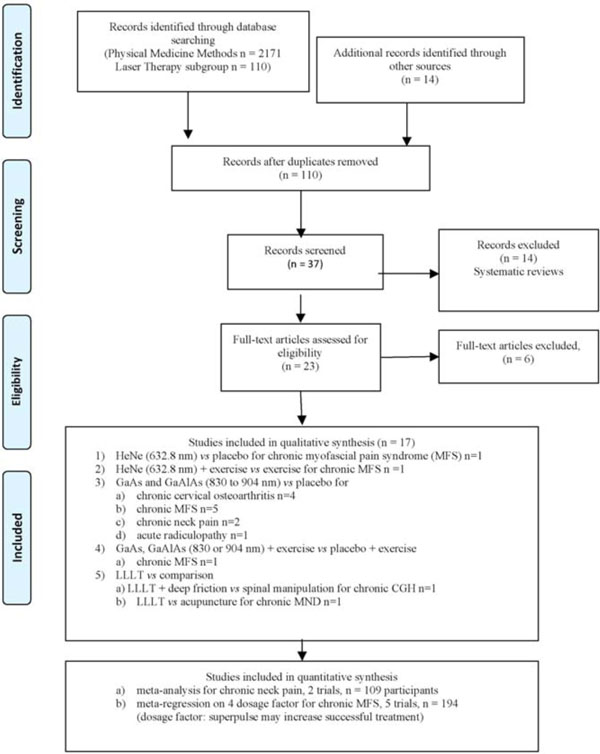
PRISMA flow diagram for study selection.
Description of Included Studies
Eleven trials examined chronic MPS [11, 12, 57-65]; one chronic [66] or one acute neck pain [67]; one chronic cervicogenic headache [68], four cervical osteoarthritis [67, 9-71] and one acute radiculopathy [72].
Risk of Bias in Included Studies
Seven of the 17 included studies were rated low risk of bias (Table 2) [11, 59, 62, 64, 66, 69, 72]. The primary methodological weaknesses were a failure to report participant compliance, to adequately describe the randomization procedure, to detail allocation concealment and to carry out intention-to-treat analysis (see Fig. 4). We assessed selective outcome reporting as being consistently unclear. Funnel plot analysis suggests a publication bias in which small, negative trials are not published (Fig. 1).
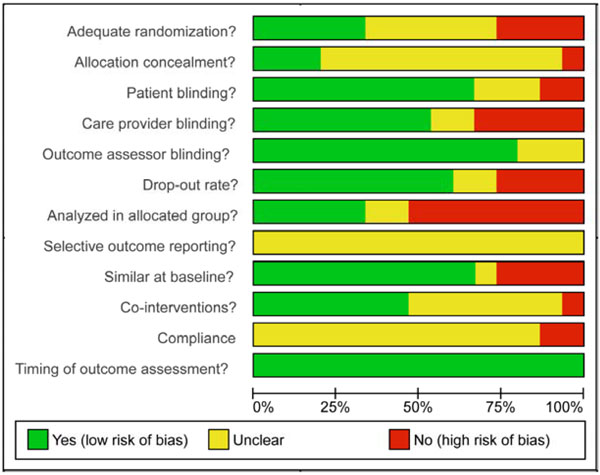
The risk of bias for all LLLT trials is depicted by each criterion.
Methodological Quality and Outcome for Each Trial
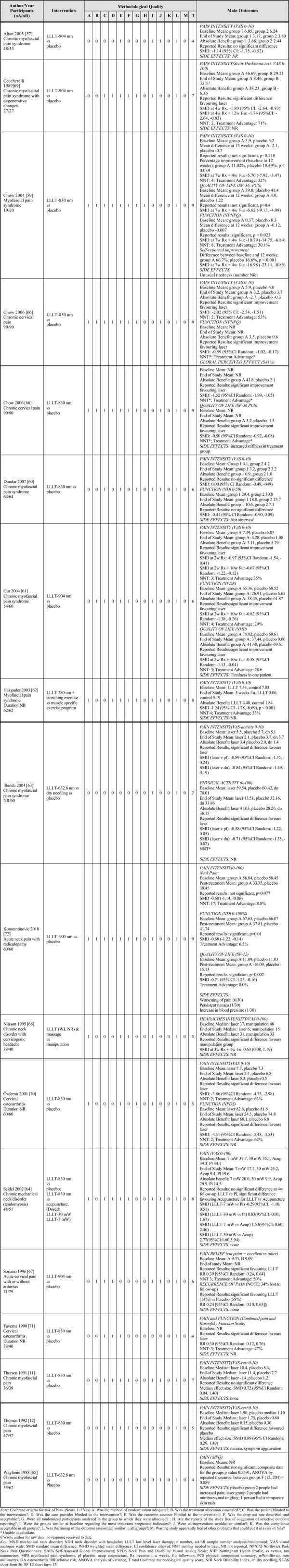 |
LLLT (HeNe 632.8 nm) Versus Placebo
Low quality evidence (1 trial, 55 participants) showed no benefit for chronic MPS in pain reduction or analgesic intake immediately post-treatment [65].
LLLT (HeNe 632.8 nm) + Exercise Versus Comparison + Exercise
Very low quality evidence (1 trial, 40 participants) that compared LLLT plus exercise to either exercise plus dry needling or exercise plus sham laser in patients with chronic MPS, showed significant improvements in pain intensity and physical activity immediately after treatment but did not maintain at six-month follow-up [63].
LLLT (830 nm or 904 nm) Versus Placebo
Eleven placebo-controlled trials evaluated LLLT in chronic MPS [11,12, 61, 64, 69] cervical osteoarthritis (OA) [69-71], acute neck pain with or without associated OA [67], acute radiculopathy [72] and chronic neck pain [59, 66]. The trials were statistically not homogenous (p < 0.00001); two trials favored placebo [11, 12], another found no difference [64] and eight trials [59, 61, 66, 67, 69-72] favored treatment. These trials were not clinically homogenous. Meta-regression was not feasible due to the small number of trials per factor. While sensitivity analysis of two factors (the duration of the disorder and methodological quality) appeared not to influence our findings, the disorder subtype did as follows (see Fig. 5).

Meta-regression for four clinically relevant dosage factors yielded the following regression equation for drive technology (SMD = - 2.70 + 1.74 DT).
Disorder Subtype 1 - Cervical Osteoarthritis
Four trials studying neck pain associated with osteoarthritis/arthrosis showed positive results for pain and function [67, 69-71]. First, when we performed the test for heterogeneity for the two trials assessing participants with osteoarthritic changes that reported continuous outcomes [69,70], they were heterogenous (p = 0.001, I2 = 94.9%). Both trials showed evidence of benefit, but we did not pool them. This difference may relate to variations in treatment characteristics and dosage; however, formal assessment needs to be conducted through meta-regression when more studies become available (See Table 3). Very low quality evidence (1 trial, 60 participants) showed a significant improvement in pain intensity and Neck Pain Disability Scale scores immediately after treatment in the LLLT group compared to placebo in subjects with cervical OA [70]. One RCT with 27 participants, some of whom had underlying cervical OA, found low quality evidence of LLLT for reducing pain immediately and in the short-term [69]. Second, one further study reported dichotomous data for participants with chronic osteoarthritic changes [71]. Very low quality evidence (1 trial, 38 participants) showed that LLLT lead to significant decreases in combined Pain and Karnofsky Function Scale scores [RR 0.30 (95%CI Random: 0.12 to 0.76)] immediately after treatment for subjects with cervical OA [71]. The NNT was 2 for pain and improved pain/disability; there was a treatment advantage of 47 to 71% for pain and 54% for pain/disability, or an absolute benefit of 47 to 50 VAS points and 68 pain/disability points (max 100). Therefore, a practitioner needs to treat two people to have one person experience about 60% pain relief and 54% pain/disability improvement. Third, one additional study evaluated acute cervical pain with or without degenerative changes [67]. Very low quality evidence (1 trial, 71 participants) showed a significant reduction in pain in the immediate term. The LLLT group had significantly lower rates of pain recurrence at six months [67].
The Laser Dosage and Clinical Characteristics are Noted for Each Trial
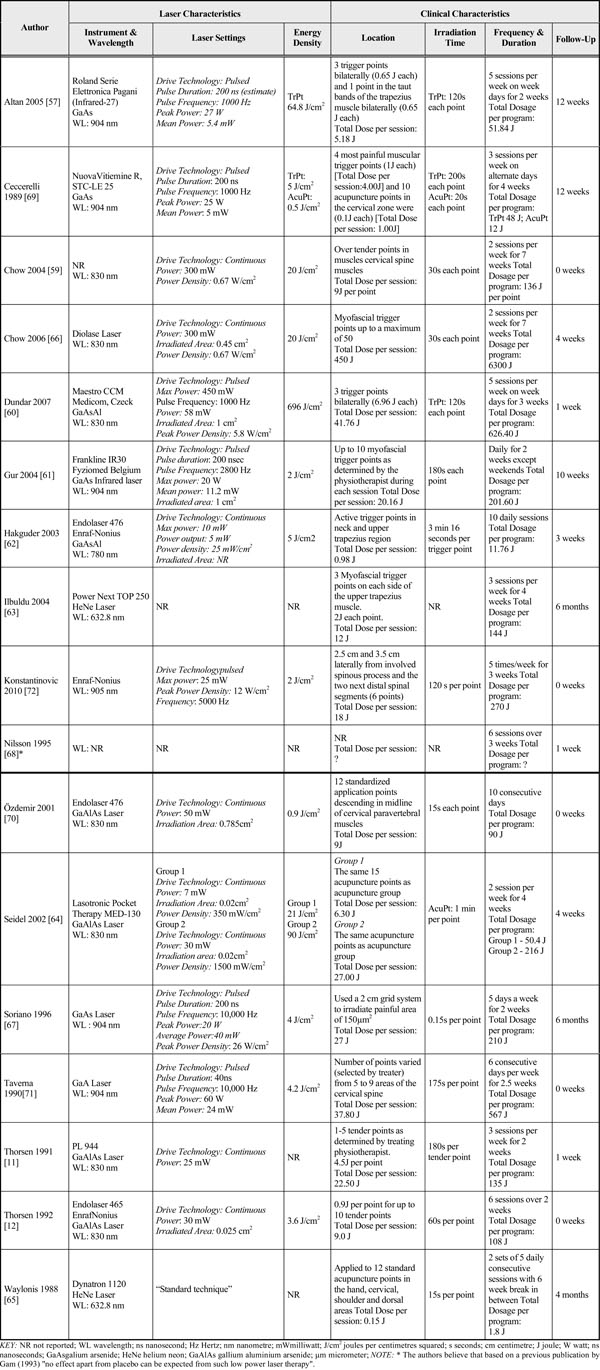 |
Disorder Subtype 2 - Myofascial Pain Syndrome
For chronic MPS there was diverse and conflicting evidence (Fig. 6). Five trials examined LLLT in patients with chronic MPS [11,12, 61, 64, 69]. The dosage of each of the trials was diverse, and therefore, due to clinical heterogeneity, we decided not to perform a meta-analysis. On the one hand, low quality evidence (1 trial, 27 participants) showed benefit of LLLT for reducing pain immediately and in the short-term [69]. Additionally, another study with 60 participants showed low quality evidence for positive immediate and short-term effects of LLLT for improving function, quality of life and reducing pain [61]. On the other hand, low quality evidence from one RCT with two separate laser arms with two distinct doses of LLLT (24 participants 12 in each arm) investigating the short-term effects of LLLT versus placebo, found no significant difference in pain levels at four weeks between the two groups in subjects with chronic mechanical neck disorder (tendomyosis) [64]. Very low (1 trial, 47 participants) [12] to low quality evidence (1 trial, 36 participants) [11] from two separate studies comparing LLLT to placebo laser showed benefits of short-term effects of placebo laser on pain. We fitted a meta-regression model to help explain the heterogeneity in these trials in terms of study-level dosage covariates. Four covariates were entered into the meta-regression model, that is, drive technology, energy density (J/cm2), dosage per session (J) and dosage per treatment program (J). Table 4 notes the analysis results. One covariate, drive technology (super-pulse), increases the chance (p=0.026) for successful outcome in clients with chronic MPS.
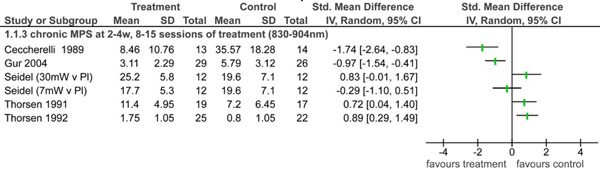
Chronic myofascial pain syndrome at 2 to 4w, 8 to15 sessions of treatment usinga830 nm or 904 nm LLLT wavelength.
GRADE Assessment of Included Studies
 |
Disorder Type 3 – Chronic Neck Pain
Two further trials investigated chronic neck pain [59, 66]. Moderate quality evidence (2 trials, 109 participants) from a meta-analysis of the effects of LLLT compared to placebo in patients with chronic cervical pain and demonstrated significantly improved pain intensity [SMDpooled -3.69 (95% CI 7.28 to -0.11)] and function [SMDpooled-5.5 (95% CI -15.48 to -4.49)] heterogeneity is significantly different p=0.00001, I2 96% in both the immediate and intermediate terms [59, 66]. Reasons to explain the heterogeneity of the populations included in these two studies were explored. However, no differences in characteristics of the patients, including age, gender and pain intensity, could be determined, and therefore, the heterogeneity that exists cannot be explained.
Despite the moderate quality of evidence that is obtained from these two trials, limitations do exist and need to be considered when interpreting the results [59, 66]. One major limitation is the presence of a selection bias, as the same research group carried out both trials. The possibility of a failure of blinding, in addition to exposure-suspicion bias, also exists, which lead to better outcomes being reported by those in the treatment group.
Disorder Subtype 4 – Acute Neck Pain with Radiculopathy
Low quality evidence (one trial, 60 participants) suggests LLLT improved pain, function and quality of life immediately post 3 weeks (15 sessions) of treatment when compared to a placebo [72].
LLLT (830 nm or 904 nm) + Exercise versus Placebo Laser + Exercise
Very low quality evidence from two trials (48 participants [57]; 64 participants [60]) examined the short-term effect of LLLT (830 nm or 904 nm) versus placebo laser with exercise in the treatment of chronic MPS. Both studies found no significant difference in pain between study groups immediately post-treatment [57, 60].
LLLT (780 nm) + Exercise Versus Exercise
Low quality evidence from one trial with 62 participants examined the short-term effect of LLLT combined with exercise versus exercise alone in the treatment of MPS [62]. The laser combined with exercise group showed a significant reduction in pain compared to the exercise alone group immediately after treatment, which was sustained for three weeks [SMD -1.24 (95%CI: -1.78 to -0.69)] [62]. Therefore, a practitioner needs to treat four people to have one person experience 33% more pain relief than exercise alone or an absolute benefit of 50 point VAS pain change from baseline.
LLLT + Deep Friction Massage Versus Manipulation
Very low quality evidence (1 trial, 38 participants) assessed the effects of massage and LLLT compared to manipulation in participants with cervicogenic headache showed no significant pain relief in the short-term [68].
LLLT (830 nm) Versus Acupuncture
There is low quality evidence (1 trial, 12 participants) of no benefit in pain intensity at four weeks follow-up when LLLT was compared to acupuncture for tenomyositis in the short-term [64].
DISCUSSION
The results of our review vary by disorder subtype, laser parameters and laser characteristics. For chronic neck pain, there was moderate quality evidence supporting the use of LLLT over placebo to improve pain, disability, QoLand GPE in the short and intermediate-term. For acute radiculopathy, low quality evidence suggested that LLLT improves short-term pain, function and QoL over a placebo. For chronic MPS, there was unclear evidence regarding the use of LLLT (HeNe 632.8 nm, GaAlAs 830 nm, GaAs 904 nm) in decreasing pain and improving function in patients. Insight from the meta-regression analysis of these trials suggests that drive technology (super-pulse) may increase the chance of successful treatment for this group of patients. When combined with exercise, laser has varied results with differing laser parameters. First, no evidence of benefit has been shown for the use of LLLT (830 nm or 904 nm) plus exercise for decreasing pain in subjects with MPS over exercise alone. Alternatively, when combined with exercise, LLLT 780 nm has been shown to decrease pain in patients with chronic MPS. When combined with deep friction massage, LLLT shows no benefit in decreasing cervicogenic headache intensity. For cervical osteoarthritis or acute neck pain, low to very low quality evidence support the use of LLLT to improve ST and IT pain and function albeit the two positive trials were clinically and statistically heterogenous and trial results could not be pooled.
Our systematic review investigates the effectiveness of LLLT in the treatment of neck pain and has several strengths. The GRADE method of analysis used in this systematic review provides information not only on the internal validity (risk of bias) of all included studies, but also external validity (design, limitations, inconsistency, indirectness, imprecision, other considerations). This, along with the clinical applicability assessment included in our review, helps to increase the generalizability and translation of our results into clinical practice. Additionally, we included studies published in languages other than English, which decreases the risk of potential language bias that could skew results. This review is not void of limitations. The doses, types of lasers, frequencies of treatment, and poor descriptions made some meta-analysis inappropriate. Many of the studies did not describe the conditions of participants in detail, thus limiting the applicability of results. The vast majority of the studies examined the effects of laser on pain and a few assessed whether there were significant changes in function, global perceived effect, or quality of life. When positive, we did translate the evidence to clinically meaningful terms including the magnitude of the effect and NNT. All of the studies had small sample sizes and few male subjects were included. Generalizability is therefore a pivotal limiting factor in our findings of predominately low and very low quality.
Our results are consistent with other current reviews investigating the effectiveness of LLLT. A systematic review by Chow and colleagues showed the benefit of LLLT for acute and chronic neck pain both in the short and intermediate terms [41]. The effects of LLLT have also been studied in patients with acute and chronic nonspecific low-back pain in a systematic review by Yousefi-Nooraie and colleagues [73]. Their study showed that LLLT leads to significant improvements in pain relief in the short and intermediate terms, with some evidence that LLLT reduces short-term disability [73].
The optimal treatment parameters for LLLT have yet to be clearly identified and have been the ‘Achilles’ heel’ to establishing sound meta-analyses [20, 74]; dosage trials for each subtype disorder are warranted. Based on the studies included in our review, combined with the results of our meta-regression, we note the following LLLT dosage characteristics for the treatment of patients with cervical pain caused by chronic MPS, chronic neck pain or osteoarthritis were used: Drive technology: super-pulse LLLT; Location: cervical trigger point(s); Time: 30 to196 seconds; Frequency: 2 to7 days per week; and Duration: 10 days to7 weeks. In future research, trials need larger sample sizes, outcomes that measure function, quality of life and global perceived effect, longer follow-up time periods, and consistent comparisons. Trials are also required to define dosage factors that lead to the most beneficial outcomes, as only one study compared dose parameters [64]. Additionally, more human trials need to be conducted to increase understanding of the underlying mechanism of action of laser on neck pain.
CONCLUSION
We found diverse evidence for the use of LLLT in the treatment of various subtypes of neck pain. We found moderate quality evidence in favour of LLLT for chronic neck painindicating further research is likely to have an important impact on our confidence in the estimate of effect and may change this estimate. Our results suggest that there is mostly very low to low quality evidence available, indicating a lot of uncertainty. Our meta-regression suggests drive technology (super-pulse) may increase the chance of success in treatment of patients with chronic MPS. Future studies with larger sample sizes are needed to explore the functional outcomes of LLLT in the treatment of neck pain, to compare different types of laser, and to further our understanding of the dosage parameters of LLLT in the treatment of neck pain.
CONFLICT OF INTEREST
Roger White, BESc., is president and CEO of Theralase Inc. His products were not assessed in any of the retrieved trials; there was no financial support received from his company.
ACKNOWLEDGEMENTS
Centric HealthLifemark is a multidisciplinary health care private service that is the Cervical Overview Group’s industry partner and provided funding administered independently through McMaster University. It has no financial interest in low level laser therapy.
We also thank Centric Health-Lifemark for being our industry partner and for their generous financial support. We thank Dr Pam Houghton (PhD) for being a consultant to this project.
APPENDIX A
COG Detailed MEDLINE Search Strategy for Physical Medicine Methods



