All published articles of this journal are available on ScienceDirect.
Minimum Two-Year Follow-Up of Cases with Recurrent Disc Herniation Treated with Microdiscectomy and Posterior Dynamic Transpedicular Stabilisation
Abstract
The objective of this article is to evaluate two-year clinical and radiological follow-up results for patients who were treated with microdiscectomy and posterior dynamic transpedicular stabilisation (PDTS) due to recurrent disc herniation. This article is a prospective clinical study. We conducted microdiscectomy and PDTS (using a cosmic dynamic screw-rod system) in 40 cases (23 males, 17 females) with a diagnosis of recurrent disc herniation. Mean age of included patients was 48.92 ± 12.18 years (range: 21-73 years). Patients were clinically and radiologically evaluated for follow-up for at least two years. Patients’ postoperative clinical results and radiological outcomes were evaluated during the 3rd, 12th, and 24th months after surgery. Forty patients who underwent microdiscectomy and PDTS were followed for a mean of 41 months (range: 24-63 months). Both the Oswestry and VAS scores showed significant improvements two years postoperatively in comparison to preoperative scores (p<0.01). There were no significant differences between any of the three measured radiological parameters (α, LL, IVS) after two years of follow-up (p > 0.05). New recurrent disc herniations were not observed during follow-up in any of the patients. We observed complications in two patients. Performing microdiscectomy and PDTS after recurrent disc herniation can decrease the risk of postoperative segmental instability. This approach reduces the frequency of failed back syndrome with low back pain and sciatica.
INTRODUCTION
Lumbar disc herniation is one of the most common spinal conditions and causes widespread medical problems. Unsatisfactory results are reported in 38% of patients who undergo lumbar disc surgery [1-3]. Recurrent disc herniation is one of the most important reasons for unsatisfactory results and, consequently, failed back syndrome. Reherniation rates have been reported as 5-26% in previously published studies [4-6].
The observed rate of recurrent sciatica after lumbar disc herniation can be as high as 37% [4]. Sciatica due to recurrent disc hernia after lumbar disc surgery, which does not respond to medical treatment and neurological deficit, requires reoperation. In such reoperations wider decompression is typically performed and more disc tissue is excised. It is a well-known fact that aggressive disc tissue removal reduces disc altitude and increases the load on facet joints; therefore segmental instability and spondylosis may develop as a result [7-9]. Such situations are one of the most important causes of failed back syndrome after lumbar discectomy and associated with poor clinical results [9-11].
Our objectives in this prospective clinical study were to prevent segmental instability and to reduce the incidence of poor clinical results with failed back syndrome after reoperation for recurrent disc herniation. In order to prevent failed back syndrome after reoperation in recurrent disc herniation cases we performed PDTS with microdiscectomy. In this paper we present our clinical results after a minimum two-year follow-up period.
MATERIALS AND METHODS
A total of 40 recurrent disc herniation cases performed between 2004 and 2007 were selected for this study. Criteria for inclusion in the study were (a) previous operation due to lumbar disc herniation (b) recurrent disc herniation on the operated side and (c) lack of a response to medical treatment at six weeks. Cases with different spinal pathology, spondylolisthesis, traumatic vertebral fracture, scoliosis, infection and serious systemic disease were excluded from the study.
All patients had leg and/or low back pain. All patients were diagnosed by performing preoperative contrast and non-contrast lumbar MRI. All patients were examined with lumbar antero-posterior and lateral X-Rays, as well as lumbar hyperflexion and hyperextension dynamic radiographies.
Cosmic (Ulrich GmbH & Co. KG, Ulm, Germany) dynamic pedicle screws and rigid rod system were used together with the microdiscectomy procedure in all patients (Figs. 1, 2).

Cosmic (Ulrich GmbH & Co. KG, Ulm, Germany) dynamic transpedicular screw.
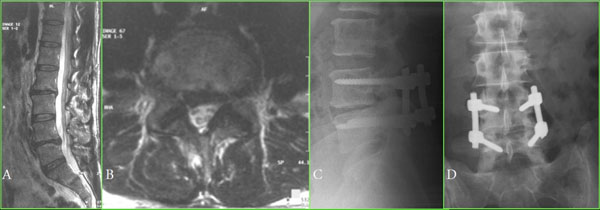
A 34-year-old male patient with microdiscectomy was operated upon two years previously due to an L4-L5 herniated lumbar disc. Fifteen days after the first operation, the patient was operated upon again due to recurrent disc herniation; only microdiscectomy was performed. Three months after the second operation, the patient was operated on again due to a second recurrent herniated lumbar disc. PDTS (Cosmic dynamic screw/ rod system) and microdiscectomy were performed. All microdiscectomies were performed at the same level and side. A) Sagittal T2-weighted MR imaging of the patient after second recurrent disc herniation. B) Axial T2-weighted MR imaging of the patient after second recurrent disc herniation. C) Lateral radiographic view after the third operation. D) Antero-posterior radiographic view after the third operation.
Clinical results were evaluated by VAS and Oswestry disability index. Measurements of the segmental lordosis angle (α), lumbar lordosis angle (LL) and intervertebral space (IVS) were used in the evaluation of patients’ radiological results. Both clinical results and radiological outcomes were recorded at 3, 6, 12, and 24 months postoperatively.
Operative Technique
All patients were taken into the operating room under general anaesthesia in the prone position. Prophylactic antibiotics were given to all patients before the operation. All operations were performed using operational microscopy and standard surgical technique. The level of operation was determined via intraoperative fluoroscopy. When the interlaminar level with recurrent disc herniation was approached from the medial aspect, existing laminotomy was widened with the help of a high-speed drill and the facet joints’ medial portions were removed. After identifying the correct nerve root, free disc fragments under the nerve root and passageway were removed. Decompression was finished by performing the required for laminotomy. After carrying out the microdecompression procedure, we also executed posterior dynamic transpedicular stabilisation from the same incision, with the help of lateral intraoperative fluoroscopy by Wiltse approach via inside lateral paravertebral muscle. The dynamic pedicle hinged screws used in our cases were Cosmic (Ulrich Gmbh & Co. KG, Ulm, Germany). Dynamic pedicle screws were used in combination with rigid rods.
Statistical Methods
The NCSS 2007 & PASS 2008 Statistical Software (Utah, USA) program was used to analyse data. Aside from descriptive statistics (average, standard deviation), repeated measures analysis of variance (repeated measures test) was used for the quantitative comparison of data showing a normal distribution. Additionally, the Bonferroni test was used in post-hoc evaluations. The Friedman test was used for the comparison of VAS parameters, which did not show a normal distribution. The Wilcoxon Signed-Rank test was used for post-hoc evaluation. The significance level was p < 0.05 in all evaluations.
RESULTS
The mean follow-up period for all 40 patients was 41 months (range: 24-63 months). The VAS and Oswestry scores showed significant improvements at 3, 12 and 24 months postoperatively as compared to preoperative scores (p < 0.01). Variation in Oswestry measurements was found to be highly significant (p < 0.01) during the follow-up period. Post-hoc Bonferroni test evaluations revealed highly significant decreases in post-operative 3rd-, 12th- and 24th-month measurements (p < 0.01) (Table 1). Variation in VAS scores during the follow-up period was also found to be highly significant (p < 0.01). According to post-hoc Wilcoxon signed-rank test, decreases in the 3rd, 12th and 24th post-operative months were highly significant (p < 0.01) (Table 1).
The Evaluation of Oswestry Disability Index and VAS Measurements
| Oswestry+ | VAS++ | |
|---|---|---|
| Means ± SD | Means ± SD | |
| Pre-op | 67,30 ± 9,04 | 7,22 ± 0,89 (7) |
| Post-op mo 3 | 26,35 ± 9,26 | 3,0 ± 1,28 (3) |
| Post-op mo 12 | 12,40 ± 6,50 | 1,47 ± 0,93 (2) |
| Post-op mo 24 | 7,70 ± 3,55 | 0,97 ± 0,73 (1) |
| P-values | 0,001** | 0,001** |
| Post-hoc | Pre-op > Post-op mo 3 (0,001**) Pre-op > Post-op mo 12 (0,001**) Pre-op > Post-op mo 24(0,001**) |
Pre-op > Post-op mo 3 (0,001**) Pre-op > Post-op mo 12 (0,001**) Pre-op > Post-op mo 24(0,001**) |
+ Repeated measures test; Post-hoc Bonferroni test was used.
++ Friedman test; Post-hoc Wilcoxon signed-rank test was used.
SD: Standard deviation; p: Significance level
** p < 0,01.
Variation in Lumbar Lordosis (LL) measurements taken during pre-operative, early post-operative, and post-operative months 3, 12, and 24 were not statistically significant (p > 0.05) (Table 2).
The Evaluation of LL, α and IVS Measurements
| Lumbar Lordosis (LL) | Segmental Lordosis Angle (α ) | Intervertebral Space (IVS) | |
|---|---|---|---|
| Means ± SD | Means ± SD | Means ± SD | |
| Pre-op | 43,52 ± 12,99 | 8,67 ± 4,98 | 0,27 ± 0,06 |
| Early post-op | 42,92 ± 12,65 | 8,42 ± 4,91 | 0,28 ± 0,06 |
| Post-op mo 3 | 42,72 ± 12,15 | 8,47 ± 4,10 | 0,27 ± 0,06 |
| Post-op mo 12 | 42,70 ± 10,94 | 8,50 ± 3,38 | 0,27 ± 0,06 |
| Post-op mo 24 | 42,95 ± 11,08 | 8,60 ± 3,67 | 0,26 ± 0,06 |
| +p | 0,969 | 0,970 | 0,640 |
| Post-hoc | N.S. | N.S. | N.S. |
+ Repeated measures test.
Post-hoc Bonferroni test was used. SD: Standard deviation.
NS: Non-significant (p > 0.05); p: Significance level
** p<0,01.
Changes in segmental lordosis angle (α) measurements taken during pre-operative, early post-operative, and post-operative months 3, 12, and 24 were not statistically significant (p > 0.05) (Table 2).
Changes in intervertebral space (IVS) measurements in pre-operative, early post-operative, and post-operative months 3, 12, and 24 were also not statistically significant (p > 0.05) (Table 2).
Data averages are summarised in Table 3.
Averages of Preoperative and Postoperative Data Points
| VAS | ODI | LL | α | IVS | |
|---|---|---|---|---|---|
| Preoperative | 7.23 | 67.30 | 43.53 | 8.68 | 0.272 |
| Early postoperative (3rd day) | - | - | 42.93 | 8.43 | 0.279 |
| 3 Month follow-up | 3.00 | 26.35 | 42.73 | 8.48 | 0.270 |
| 12 Month follow-up | 1.48 | 12.40 | 42.70 | 8.50 | 0.270 |
| 24 Month follow-up | 0.98 | 7.70 | 42.95 | 8.60 | 0.275 |
VAS: Visual Analog Scale, ODI: Oswestry Disability Index, LL: Lumbar Lordosis Angle
α Segmental Lordosis Angle, IVS: Intervertebral Space.
No novel recurrent disc herniation was observed during the follow-up period.
We observed complications in two patients. Foreign body reaction was observed in the first patient. The patient was reoperated upon and the dynamic stabilisation system was removed. In the other patient low back pain and sciatica due to PDTS continued. Therefore, the dynamic system was removed and fusion with rigid stabilisation was performed.
DISCUSSION
Recurrent disc herniation accounts for the most common problematic situations after lumbar disc surgery. Recurrent disc herniations are radiologically visualised lumbar disc herniations, which are non-responsive to medical treatments other than surgery [12-16]. The rate of reoperation due to recurrent disc herniation after lumbar disc surgery is approximately 5-15% [3]. The ethyopathogenesis of recurrent disc herniation is a controversial issue. The rate of recurrent disc herniation is significantly higher in preoperative MRI analyses of patients with modic changes in end-plates of the vertebra corpus [17, 18]. Cinotti reported that recurrent disc herniation patients’ pre-operative MRI analyses revealed higher degrees of disc degeneration than were observed in the control group [12]. Cinotti also reported that mechanic loading causes sciatica in 42% of recurrent disc herniation patients [12]. Some experimental studies showed that the frequency of recurrent disc herniation after lumbar disc surgery is higher due to insufficient recovery in the annuli of discs with a high degree of degeneration [15, 19].
A recent study by Barth et al. compared clinical and radiological results of lumbar microdiscectomy and microscopic sequestrectomy after a two-year follow-up period. The microdiscectomy patient group presented deteriorative functional results, whereas the sequestrectomy group presented stable results. There was not any difference in reherniation rate between the two groups [20]. Radiological evaluation of the same study and correlation with clinical results showed that diagnoses of post-operative disc degenerations such as decreases in disc altitude and end-plate degeneration were much less frequent among patients who underwent sequestrectomy as compared to microdiscectomy. Furthermore, modic-type end-plate changes were associated with negative clinical results [21]. Recently, Carragee described a lumbar disc herniation classification system based upon continuity of the annulus and the presence of extruding/free disc fragments [4]. In his study, the reherniation rate after limited discectomy in the fragment-defect (wide annular defect) patient group was as high as 27.3%. Similarly, the reoperation rate in this group was significantly high at 21.2% in comparison to the fragment-fissure (small annular defect) patient group, for which the reherniation and reoperation rate was 1.1%. Moreover, when patients in fragment-defect group, with more than 6 mm annular defect in Carragee classification, were questioned in the postoperative period with 27.3% the complaint rate of persistent/recurrent sciatica after microdiscectomy was much higher than expected [4]. We think that in these patients both clinical and radiological recurrence of lumbar disc herniation would be higher regardless of the surgical treatment is performed or not. On this account, considering reherniation rate of 27.3% and reoperation rate of 21.2% in cases with wide annular defect in Carragee classification, in fragment-defect group; in order to decrease the rates of both reherniation and failed back syndrome we do not think that is it wrong or unnecessary to propose performing PDTS even in the first surgery. Carragee et al. then performed aggressive discectomy in patients with wide annular defects in order to decrease the rate of reherniation [22]. However, after a one-year follow-up period these patients reported persistent low back pain problems. Recurrent disc herniation is one of the most important reasons for unsatisfactory results after lumbar disc surgery and, consequently, failed back syndrome. In contrast, successful results are reported when recurrent disc herniation is treated with another microdiscectomy [23]. However, the risk of developing new recurrences after the first recurrence is 15-20% [24]. The patient’s chances for successful recovery gradually decrease after each simple discectomy and the risk of other spinal operations increases accordingly [24]. Re-performed discectomies do not stop continuous segmental degeneration; moreover, they may aggravate the degeneration process [15, 25-27]. Declines in intra-disc pressure and disc elevation increase loading on facet joints; as a result segmental instability may develop due to laxity in articular capsules and ligaments [7, 8, 19, 28]. Low back pain and/or segmental instability with sciatica significantly contribute to the development of failed back syndrome after lumbar disc surgery. According to the relevant literature, the incidence of spinal instability in patients with low back pain is around 20-30% [29, 30]. Segmental instability is diagnosed in 20% of patients with lumbar disc herniation [31, 32]. Instability after lumbar disc surgery is secondary segmental instability, described as ‘status-post discectomy’ by Frymoyer [33]. In fact this situation is not an overt instability; as described by Benzel, it is a chronic instability [34]. Studies have shown that when performed on segmental degeneration cases, discectomy may cause segmental instability and accounts for 38% of unsatisfactory results [2, 35].
Segmental fusion operations are performed frequently as treatment for recurrent disc herniation. Nevertheless, fusion also carries various risks such as adjacent segment degeneration, bone graft donor place pain, and pseudoarthrosis [26,27,35,36]. Today, dysfunctional segmental movement and chronic instability due to recurrent disc herniation are usually treated with posterior dynamic stabilisation. In chronic instability cases it is very important to make sure that the stabilisation surgery is suitable for every age group and certain to yield satisfactory results. Recently, several clinical studies reported that posterior dynamic transpedicular stabilisation yielded good clinical results and represents a safe and effective alternative technique to spine arthrodesis in selected cases of degenerative lumbar spine instability [37-39]. In fact, posterior dynamic stabilisation appears to be essential in order to achieve the desired outcomes. Dynamic stabilisation controls abnormal movements in an unstable, painful segment and facilitates healthy load transfer, in order to prevent degeneration of the adjacent segment [40]. Thus spinal stabilisation is achieved while alleviating unnecessary pain. Some studies reported that posterior dynamic transpedicular stabilisation biomechanically provides stabilisation that is similar to that provided by rigid systems [41-43]. Moreover, dynamic stabilisation systems appear to have advantages over rigid spinal implants. Putzier et al. reported that after 34 months of follow-up, disc degenerations showed far less progression in patients who had nucleotomy with posterior dynamic system applications as compared to patients who did not have dynamic stabilisation [35]. Schaeren et al. reported very successful clinical and radiological results after performing dynamic stabilisation for cases of degenerative spondylolisthesis. However, they also reported that adjacent segment degeneration remained problematic [44]. In another new systematic review study, Barrey et al. reported that there is not a large difference between the effects of rigid stabilisation systems and dynamic systems on adjacent spinal segments [45]. Several studies in the literature reported that recurrent disc herniation with the probability of developing instability and isolated spinal canal stenosis are new indications for dynamic stabilisation systems during the postoperative period [35, 46, 47]. Nevertheless, in recent years dynamic stabilisation devices are being inserted to treat the segmental instability due to iatrogenic decompression or segmental degeneration [48,49].
In this study we detected satisfactory improvements in VAS and Oswestry scores after a minimum of two years’ follow-up. We achieved encouraging clinical and radiological results after performing microdiscectomy together with PDTS in our recurrent disc herniation patients. Furthermore, no novel recurrent disc herniation was seen during the follow-up period (mean: 41 months) in any of the patients included. Consequently, we can recommend performing PDTS along with microdiscectomy in cases of recurrent disc herniation with risk of segmental instability and failed back syndrome. We think that posterior dynamic stabilisation is an effective alternative to fusion in the treatment of chronic instability and degenerative diseases of the lumbar spine.


