All published articles of this journal are available on ScienceDirect.
Intramedullary Nail-Guided Correction of Lower Extremity Long Bone Deformity: Technique and Case Series
Abstract
Introduction
Limb deformities of the lower extremity are frequently managed with gradual correction using external fixators. Some authors have described utilizing computer-assisted hexapod external fixators for precise intraoperative Deformity Correction (DC). However, external fixator correction is both costly and time-intensive, and intramedullary nail-guided correction (IMNG) may be an acceptable alternative. The purpose of this study was to assess the efficacy of IMNG as a non-inferior cost-effective alternative approach to lower extremity long bone DC.
Methods
All patients who underwent femoral or tibial IMNG at a single institution from 2006 to 2022 were retrospectively reviewed. Pre- and postoperative deformity measurements, including Mechanical Axis Deviation (MAD), Lateral Distal Femoral Angle (LDFA), Medial Proximal Tibial Angle (MPTA), and Lateral Distal Tibial Angle (LDTA), were recorded using standing hip-to-ankle radiographs. Radiographic and clinical outcomes were recorded.
Results
Twenty-four IMNG Deformity Correction (DC) procedures performed on 18 patients were included [10 (56%) female, mean age 30 (15-62) years, mean body mass index 31 (21-67) kg/m2, mean follow-up 36 (9-111) months]. There were 10 (45%) isolated femoral DC, 10 (45%) isolated tibial DC, and 2 (9%) ipsilateral femoral and tibial DC in a single operation. All patients were followed until clinical and radiographic union. Mean MAD was corrected from 42.5 ± 35.0 mm preoperatively to 10.2 ± 4.8 mm from the targeted MAD. Complications occurred in 9 (50%) patients, predominantly related to symptomatic hardware and delayed healing. The rate of soft tissue complications was 5.6%.
Discussion
The IMNG achieved accurate correction with lower rates of soft tissue complications compared to computer-assisted hexapod fixators. Furthermore, the rate of hardware removal with IMNG was approximately half of that seen with external fixator methods.
Conclusion
IMNG correction is an effective and cost-conscious technique to correct complex long bone deformities of the femur and tibia.
1. INTRODUCTION
Long bone deformity is commonly seen in patients with metabolic bone disease, congenital deformity, growth disturbance, or impaired osteogenesis following traumatic injury. In the lower limb, deformity may lead to pain, fractures, and impaired function [1-3]. The natural history of untreated deformities often includes early arthrosis due to an abnormal mechanical axis [4-9]. Surgical intervention aims to correct the mechanical axis of the lower extremity, leading to a lower risk of subsequent fracture and arthrosis, as well as aiding patient mobility and independence. Traditionally, treatment of long bone deformity with external fixation has achieved positive clinical outcomes via gradual correction. However, this demands a significant financial and time commitment from both patient and provider. Additionally, external fixators carry a risk of pin-site infections, and prolonged soft-tissue transfixation can cause scarring and pain and may even impair the range of motion, affecting postoperative rehabilitation [10-12].
More recently, computer-assisted hexapod external fixators have been used in Deformity correction (DC) [13-15]. This type of frame (two or more rings, connected by six telescopic struts) allows for precise three-dimensional DC, as well as for gradual correction over time. One of this technology’s main advantages is that it allows the surgeon to input defined parameters for the current deformed bone state, as well as the desired correction, leaving the software to determine precise strut adjustments required to accomplish the correction. This use of computerized software to assist in complex DC has been further expanded to assist with acute DC. Authors have described the use of Computer Hexapod-Assisted Orthopedic Surgery (CHAOS) as an alternative method to achieve precise DC intraoperatively [12,16,17]. CHAOS applies a hexapod external fixator only intraoperatively, utilizing strut settings and computer software to assist with DC. After the desired DC is achieved, definitive internal fixation via plate or intramedullary nail (IMN) is performed, with removal of the hexapod frame intraoperatively. Proponents of CHAOS indicate that it enables precise, minimally invasive multiplanar DC intraoperatively. Multiple studies report highly accurate correction with CHAOS for a variety of lower extremity deformities, accompanied by a low complication rate [12,16-18].
However, utilization of the hexapod external fixator is both costly and time-intensive. The quoted cost of the frame, struts, and necessary pins can vary, but estimates are as high as $10,675 [19]. Procedure duration is also substantially increased with CHAOS, with an average of 43 minutes required for frame application intraoperatively prior to beginning internal fixation [12].
The aim of this retrospective cohort study was to assess the efficacy of intramedullary nail-guiding (IMNG) as a non-inferior cost-effective alternative approach to CHAOS for lower extremity long bone DC. It is hypothesized that the IMNG approach can achieve DC at a rate of complications equal to or lower than that of the CHAOS procedure. Surgical description of the IMNG technique is supplemented by a review of clinical and radiographic outcomes of the technique in a patient cohort treated at a single institution.
2. METHODS
2.1. Retrospective Clinical Cohort
A retrospective review was conducted of consecutive patients with long bone deformities of the lower extremity (femur and/or tibia) treated with IMNG between 2006 and 2022. During the study period, 24 corrections of metaphyseal or diaphyseal femoral and/or tibial deformities in 18 patients were performed. Medical records and serial radiographs were reviewed to quantify pre-operative and post-operative deformity and to assess for the development of postoperative complications (i.e., infection, nonunion, malunion, reoperations). Preoperative deformity was measured from radiographs taken on the closest date prior to surgery. Postoperative deformity was measured using the most recent radiographs. Standing hip-to-ankle radiographs were utilized for pre- and postoperative Mechanical Axis Deviation (MAD). When unavailable, anatomic measurements were recorded from femoral or tibial radiographs that included both proximal and distal joints. All deformity measurements, including MAD, lateral distal femoral angle (LDFA), Medial Proximal Tibial Angle (MPTA), and Lateral Distal Tibial Angle (LDTA), were measured in accordance with the method outlined by Paley et al. [20]. Patients were excluded if they were lost to follow-up prior to clinical and radiologic evidence of union.
Data were summarized by reporting counts and percentages for categorical covariates and means and standard deviation or range for continuous covariates.
2.2. Preoperative Planning
Identifying the proper osteotomy site requires an understanding of the three-dimensional structure of the bone contributing to the deformity, as well as the correction required to restore alignment of the patient’s innate mechanical axis. The pre-operative MAD is measured with bilateral standing hip-to-ankle films with a plumbline connecting the center of the hip to the center of the talus. The MAD is measured from the center of the knee to the mechanical axis line (Fig. 1). Varus deformities have a positive MAD, while valgus deformities have a negative MAD. Joint centers are identified at the hip, knee, and ankle, and the mechanical axes and joint lines are then drawn. This information will determine the joint angles, comparing them to population norm measurements. After analyzing the joint angles, the deformity is identified at either the femur, tibia, or both. The apex of the deformity is identified, which often dictates the location of the planned osteotomy (Fig. 2). Occasionally, the apex of the lower limb deformity is at the knee joint, which requires an alternative location for the osteotomy at the expense of accepting translation after DC. In patients with clinical symptoms and radiographic evidence of unicompartmental osteoarthritis, targeted deformity correction was templated to over-correct for this deformity. Additionally, in 1 patient (patient #18), a postoperative valgus alignment was planned to restore metadiaphyseal alignment for subsequent placement of knee arthroplasty components. As a result, correction of MAD from the preoperative to postoperative state is also reported with targeted/goal correction, as not all patients’ MADs were targeted to 0 mm.
2.3. Operative Technique - Femoral Deformity
Deformities proximal to the isthmus were often managed with an antegrade femoral nail, while deformities of the distal femur were corrected with a retrograde IMN. In the case of antegrade femoral nailing, the guidewire is fluoroscopically positioned onto the appropriate starting point of the femur, based on the selected nail geometry. The type of nail, i.e., trochanteric versus piriformis, is less important than understanding the exact geometry of the nail and its relationship to the anatomy of the patient’s proximal femur. Templating software is utilized to ensure the paths of the opening reamer and IMN through the proximal femur are exactly colinear to the anatomic axis of that bone segment. After opening reaming, the intramedullary reduction tool is inserted into the femur and utilized as a surrogate for eventual nail placement. Based on preoperative planning, standard wedge osteotomy is carried out at the apex of the deformity.
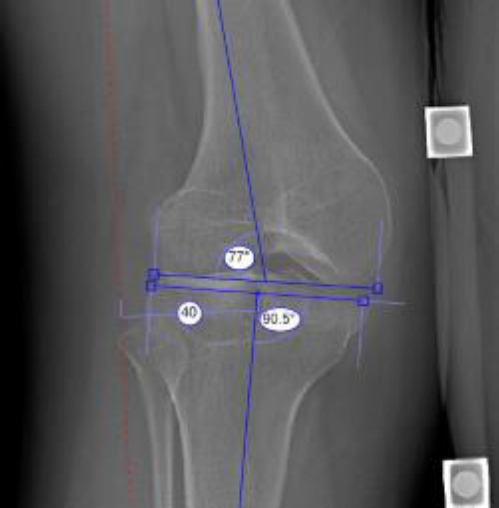
Preoperative analysis of the mechanical axis depicts 40 mm of lateral joint deviation in a patient with right femoral valgus (patient #14). The red dashed line represents the mechanical axis. Lateral distal femoral angle (77°) and medial proximal tibial angle (90.5°) are also shown for reference.
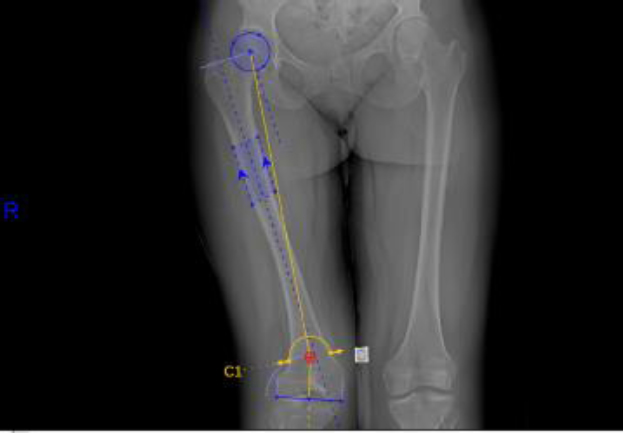
Preoperative hip-to-ankle standing radiograph with templated dome osteotomy for correction of a femoral deformity (patient #14).
For distal femoral deformities, a retrograde nail combined with supracondylar dome osteotomy is typically used. In this case, a longitudinal incision is made just proximal to the patella, splitting the quadriceps tendon in line with its fibers, to access the anterior supracondylar femur, and the dome osteotomy is performed. A single guide pin is placed at the apex of the deformity, marking the point around which the dome osteotomy should rotate. Using a parallel pin guide, multiple drill holes are made in a dome-shaped pattern and subsequently connected using an osteotome. To access the starting point for the retrograde nail, a mini-medial parapatellar approach is made just proximal to the tibial tuberosity. After dissecting through the skin and subcutaneous tissue, the patellar tendon is identified and retracted laterally. The entry portal for the retrograde IMN at the intercondylar notch is identified fluoroscopically. Fluoroscopy is used to confirm the appropriate starting point and trajectory that is exactly colinear with the anatomic axis of the distal femur. The guide pin is placed, and the cannulated opening reamer is utilized to create the initial nail pathway through the distal femur. Based on patient anatomy, an appropriately sized IMN is advanced to the point of the osteotomy. Appropriate nail positioning is confirmed via fluoroscopy throughout advancement. At the osteotomy site, the bone is manipulated to align the nail with the anatomical axis, and the nail is advanced further, correcting the deformity. After advancement of the nail, satisfactory reduction and restoration of the anatomic and mechanical axes are again confirmed fluoroscopically. The nail is then locked either proximally or distally, the osteotomy site is compressed, and the nail is secured proximally with 2 statically locked bolts. If a closing wedge osteotomy was performed, the wedge of bone is excised, any cancellous bone removed is packed back into the osteotomy site, and a standard closure is performed.
Details regarding surgical technique are supplemented by an example of closing wedge osteotomy of a distal femoral deformity (Fig. 3).
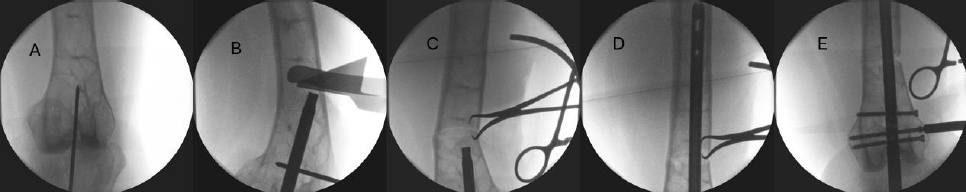
A) Based on preoperative templating, the retrograde starting point is obtained. B) After reaming the entry point to a diameter 1mm larger than the nail diameter, a tubular reduction device is advanced to the apex of the deformity. Osteotomy triangles are used to align the sagittal saw to cut the desired-sized wedge. Circumferential protection of the surrounding soft tissues is achieved with thin, malleable retractors contoured for the specific anatomic site. C) After removal of the wedge, the reduction is obtained and compressed with a pointed reduction clamp placed through unicortical drill holes above and below the osteotomy site. D) The intramedullary nail is advanced across the compressed osteotomy site. E) The intramedullary nail is locked above and below the osteotomy site.
2.4. Operative Technique - Tibial Deformity
The patient is placed in a supine position on the operating table. An incision is made just proximal to the patella, splitting the quadriceps tendon in line with its fibers, to access the appropriate pathway for suprapatellar tibial nailing. Again, the precise point that matches the planned nail pathway after DC is utilized to determine the exact starting point for the guide pin. This must account for the exact size and shape of the planned IMN in both coronal and sagittal planes to ensure precise DC. After confirming the position of this guide pin via fluoroscopy, attention is turned to the osteotomy. An incision is made at the appropriate site on the tibia, and depending on patient-specific preoperative planning, a standard wedge osteotomy is performed. An appropriately sized nail for the patient’s medullary canal is chosen. After reaming to accommodate a secure and stable fit, the nail is advanced into the proximal tibia. As the nail reaches the point of osteotomy, the osteotomy site is manipulated to align the IMN to be parallel with the anatomical axis of the tibia. The nail is then advanced through the osteotomy, and fluoroscopy is used to confirm appropriate mechanical axis alignment and compression of the osteotomy site. After advancement of the nail to achieve a satisfactory reduction, the nail is statically locked proximally and distally. Any cancellous bone from the osteotomy site is packed back in prior to closure to accentuate bone healing. A standard closure is then performed.
3. RESULTS
Twenty-four IMNG procedures were performed in 18 patients, 10 of whom were female. At the time of surgery, the mean patient age was 30 (15 to 62) years, and the mean body mass index was 31 (21-67) kg/m2. Ten (56%) patients had metabolic bone disease, 4 (22%) had a history of congenital deformity or childhood growth disturbance, and 3 (17%) patients required DC for malunion following trauma (Table 1). The mean time of follow-up after surgery was 36 months (range 9 to 111 months). Eight patients underwent correction of isolated tibial deformities, of whom two had contralateral tibial deformities corrected in separate operations. Ten patients underwent isolated femoral DC, and two patients also underwent multiple DC operations for both femoral deformity and tibial deformity.
| Patient | Age | Sex | Relevant Medical History | Prior Corrective Surgery | Deformity/Deformities Corrected | Complications |
|---|---|---|---|---|---|---|
| 1 | 19 | F | Alagille Syndrome | Femoral osteotomy | Left tibial varus and procurvatum | Hardware removal, bilaterally |
| 20 | - | - | Femoral osteotomy | Right tibial varus and procurvatum | - | |
| 2 | 16 | M | Childhood growth disturbance | None | Left tibial varus and procurvatum | - |
| 3 | 15 | M | Malunion | None | Right tibial varus and procurvatum | - |
| 4 | 11 | F | Right proximal femoral cyst | Three prior operations, including excision, bone grafting, hip screw, and sideplating | Right femoral varus | - |
| 5 | 47 | M | Malunion | ORIF right proximal tibia | Right tibial varus | - |
| 6 | 38 | F | Osteogenesis imperfecta | Bilateral tibial rods | Right femoral varus and procurvatum | - |
| 7 | 17 | F | Hypophosphatemic rickets | Eight plates in the right distal femur and right proximal tibia | Right tibial varus and procurvatum | Delayed union requiring nail dynamization |
| 18 | - | - | Eight-plate in the left proximal tibia | Left tibial varus and recurvatum | - | |
| 8 | 21 | F | Hypophosphatemic rickets | Bilateral lower extremity osteotomies | Right femoral valgus and procurvatum | - |
| 26 | - | - | - | Left femoral varus and procurvatum, left tibial varus | - | |
| 9 | 48 | F | Malunion | None | Right tibial varus | - |
| 10 | 22 | F | Congenital deformity, iatrogenic injury during left total hip arthroplasty |
Left total hip arthroplasty | Left femoral valgus | - |
| 11 | 40 | F | Osteogenesis imperfecta | Femoral osteotomy with ORIF plate, Rush rod fixation | Right femoral varus and procurvatum | Delayed union requiring open reduction and internal fixation with iliac crest bone grafting |
| 12 | 26 | F | Nail-patella syndrome | None | Right femoral valgus | Hardware removal |
| 13 | 53 | M | Osteogenesis imperfecta | None | Left tibial valgus and procurvatum | Hardware removal |
| 14 | 25 | F | Congenital deformity | None | Right femoral valgus, right tibial valgus | - |
| 27 | - | - | - | Left femoral valgus | - | |
| 15 | 32 | M | Osteogenesis imperfecta, Klippel-Trénaunay syndrome | Tibial osteotomy | Right tibial valgus | Hardware removal |
| 16 | 40 | M | Congenital deformity | None | Left femoral valgus and rotational deformity | - |
| 17 | 30 | M | Hypophosphatemic rickets | Tibial osteotomy | Right femoral valgus | Nonunion and infection requiring irrigation and debridement, femoral plating, and bone grafting |
| 18 | 62 | M | Osteogenesis imperfecta | Intramedullary femoral nail | Left femoral valgus | Nonunion requiring open reduction and internal fixation with iliac crest bone grafting |
Five patients had pre-existing unicompartmental osteoarthritis with a medially targeted mechanical axis. Patient 18 was indicated for staged knee arthroplasty following femoral deformity correction and, as such, was templated to have a residual valgus deformity to accommodate the anticipated total knee components.
Pre- and postoperative radiographs were available for 16/18 (89%) patients to assess changes in standard lower limb angles post-correction. The mean preoperative MAD was 42.5 mm, ranging from 3 mm to 134 mm away from the targeted postoperative MAD. The patient with preoperative MAD of 3 mm had a severe varus deformity of the femur. Postoperative average MAD was 10.2 ± 4.8 mm from the targeted MAD, with overall MAD ranging from 0.55 to 14.9 mm from the target. In patients with femoral deformities, preoperative mechanical and anatomic LDFAs averaged 87.6° (range: 74° to 111°) and 77.9° (66.6° to 100°), respectively. Postoperative mechanical and anatomic LDFAs were 89.3° (85.7° to 99°) and 82.5° (76.7° to 92.3°), respectively. In patients with tibial deformities, the average preoperative MPTA was 84.8° (77.4° to 94.1°), and postoperative MPTA was 86° (73.1° to 92.4°). LDTA averaged 85.9° (50.6° to 96.8°) preoperatively as compared with 87.1° (73.6° to 93.6°) postoperatively (Table 2).
| Preoperative | Postoperative | |
|---|---|---|
| All (n=24) | ||
| Mechanical axis deviation, from target (mm) | 42.5 ± 35.0 | 10.2 ± 4.8 |
| Tibial varus (n=9) | ||
| Medial proximal tibial angle (°) | 83.3 ± 5.5 | 86.1 ± 6.1 |
| Lateral distal tibial angle (°) | 91.2 ± 4.4 | 89.5 ± 3.8 |
| Tibial valgus (n=3) | ||
| Medial proximal tibial angle (°) | 91.6 ± 4.1 | 86.5 ± 2.3 |
| Lateral distal tibial angle (°) | 70.0 ± 17.1 | 81.0 ± 6.6 |
| Femoral varus (n=4) | ||
| Mechanical lateral distal femoral angle (°) | 94.1 ± 16.4 | 86.9 ± 0.3 |
| Anatomic lateral distal femoral angle (°) | 86.1 ± 12.2 | 78.8 ± 3.5 |
| Femoral valgus (n=8) | ||
| Mechanical lateral distal femoral angle (°) | 85.2 ± 11.5 | 90.3 ± 4.7 |
| Anatomic lateral distal femoral angle (°) | 74.8 ± 5.0 | 84.1 ± 4.9 |
Definitive fixation was achieved in each case with a statically locked IMN. Twelve procedures were performed for uniplanar DC, and ten procedures corrected biplanar deformities. Seven patients had two-level osteotomies. All corrections of tibial deformities were performed with closing wedge osteotomies. Femoral deformities were corrected with closing wedge osteotomies in five procedures and dome osteotomies in five procedures. Femoral corrections were performed with retrograde IMN in 8 cases and anterograde IMN in 4 cases. Postoperative rehabilitation protocol encouraged immediate weight-bearing as tolerated in the operative extremity.
Complications occurred in 5 patients with tibial DC, including 3 cases of hardware removal due to symptomatic prominent interlocking screws and 1 case of delayed union. Complications arose in 4 patients with femoral DC, including 3 cases of nonunion, 1 case of hardware removal due to prominent screws, and 1 case of nonunion and infection (Table 1). There were no cases of peroneal nerve palsy or compartment syndrome. All patients achieved eventual clinical and radiographic union. Pre- and postoperative radiographs of 4 cases are included to highlight the variation in presentation and operative site (Figs. 4-7).
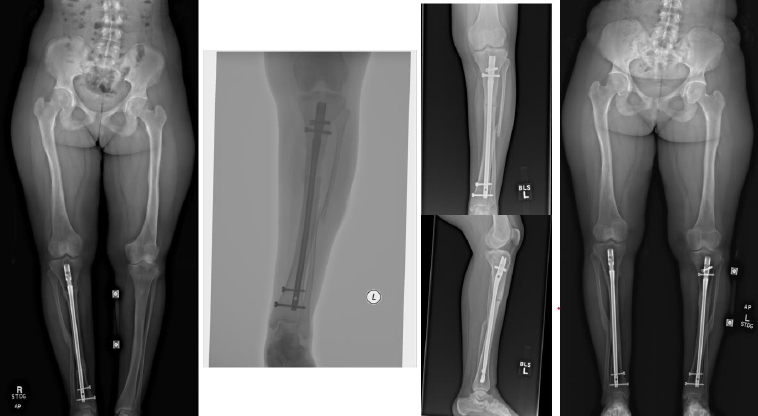
From left to right: Preoperatively, immediately postoperatively, 5 months postoperatively, and 5 years postoperatively (patient #7).
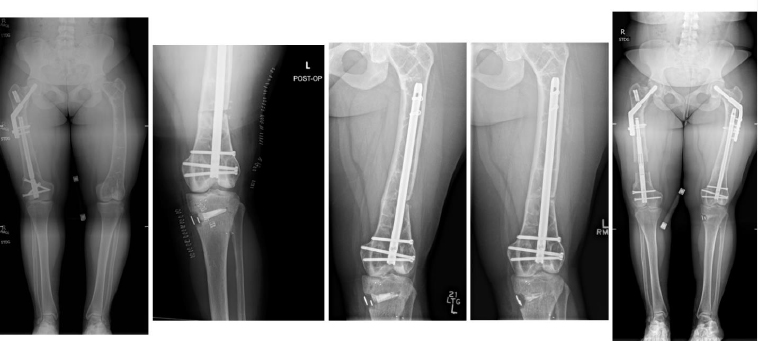
From left to right: Preoperatively, immediately postoperatively, 3 months postoperatively, 9 months postoperatively, and 3 years postoperatively (patient #8).
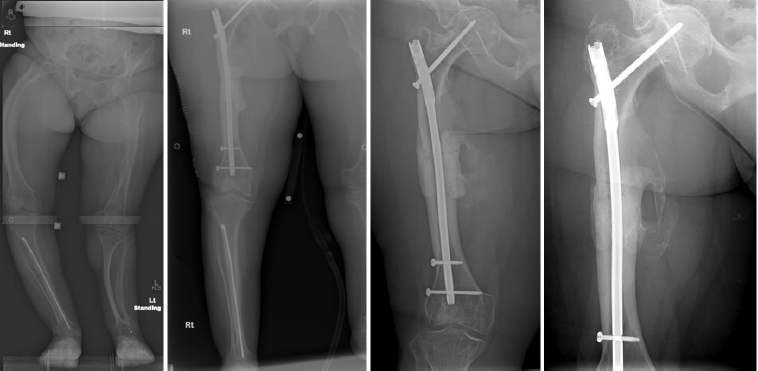
From left to right: Preoperatively, immediately postoperatively, 5 months postoperatively, and 4 years postoperatively (patient #6).
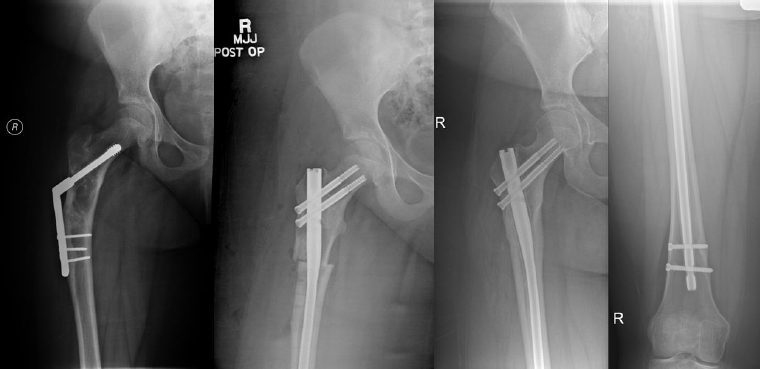
From left to right: Preoperatively, immediately postoperatively, and 4 years postoperatively (patient #4).
4. DISCUSSION
Results of this patient series demonstrate that the IMNG approach to complex tibial and femoral deformities is reliable and effective at correcting large deviations in limb alignment. The patients in this cohort had a high degree of variation in deformity types and preoperative MAD, including an MAD that measured 134 mm in one patient. Postoperative MAD was corrected to an average of 10.2 mm from the target, demonstrating that IMNG is well-suited for realigning the lower extremity biomechanical axis within the normal range of MAD, previously defined by Paley et al. as 9.7 ± 6.8 mm medial to the center of the knee joint line [20].
Previous studies conducted on patients who underwent DC with the CHAOS technique report restoration of anatomic alignment, though there has been variation in the data points reported. In the initial CHAOS paper by Rogers et al., MAD was represented as a ratio of distance from the center of the knee joint to the width of the tibial plateau, rather than measured in millimeters, to account for variation in the size of radiographs available [12]. Though an MAD ratio correction of 0.42 and a postoperative MAD ratio of 0.11 were reported, it is difficult to quantify the exact postoperative MAD. In a 2016 study by Hughes et al. on tibial DC in 12 patients using CHAOS, postoperative tibial mechanical axis was reported as 0.7° varus, which is in line with historical normal anatomical measurements [17,21,22]. In the most recent CHAOS study to report MAD, French et al. reported mean postoperative MADs of 4 mm and 7.4 mm for valgus and varus deformities, respectively, though the maximum MAD postoperatively ranged up to 27 mm[18]. In comparison to previous studies on CHAOS, IMNG demonstrates a similar level of precision in overall DC as measured by postoperative MAD.
Compared with CHAOS, IMNG is accompanied by a comparable or lower rate of complications, especially relating to soft tissues. The additional pin-sites necessary for hexapod frame attachment to various segments of the deformed bone expose the patient to a greater risk of soft tissue injury or infection, especially in the setting of multilevel osteotomies. Compared with IMNG, which resulted in a relatively low infection rate (5.6%) and did not result in any nerve injuries or vascular complications, CHAOS studies report varying soft tissue complication rates ranging up to 33%, including common peroneal nerve palsies, tibial pseudoaneurysms, and superficial wound infections [12,16-18]. The rate of nonunion or delayed healing in this study was 22%, which is slightly higher compared to the rate of nonunion and malunion with CHAOS (0 - 14%). The rate of hardware removal in patients treated with IMNG was 22%; in the two largest cohorts of patients treated with CHAOS, subsequent hardware removal was required in 41 - 58% of cases, though the studies did not further delineate which of these patients required removal for soft tissue irritation versus secondary surgery [16,18]. Nevertheless, the rate of hardware removal in patients in this IMNG cohort is approximately half of that reported with CHAOS and is an important consideration when reducing the overall morbidity associated with DC.
Perhaps the most significant advantages of the IMNG approach are the simplicity of the surgical technique and decreased financial burden. Surgical procedures are a major expense for the healthcare system, and operating room costs make up the second-largest portion of surgical care spending [23]. Review of the manufacturer’s suggested retail price of one example hexapod frame system (Taylor Spatial Frame, Smith and Nephew, Memphis, TN) compared with intramedullary nailing with interlocking screws revealed a four-fold difference in cost; the hexapod frame retail price is approximately $39,000, whereas the cost for IMNG is roughly $10,000. This gap in price widens further after accounting for the fact that 20-33% of patients undergoing CHAOS required multilevel osteotomies, necessitating additional hexapod frames [16,18]. However, it must be noted that these price estimates assume the hexapod frame to be a single-use item, which has been reinforced in existing literature [16,18]. At the study institution, reuse of any part of the hexapod frame system is similarly prohibited. This discrepancy in cost may be potentially reduced at certain institutions that permit the reuse of struts or ring components for temporary intraoperative use. Institutional single-use versus reuse of hexapod frame system components is not well-described, and data from other institutions are needed to better characterize variation in equipment expenses. Importantly, the savings in operating room time with IMNG in comparison to CHAOS are also substantial. Rogers et al. reported that application of the hexapod frame intraoperatively required between 35 and 60 minutes prior to initiation of internal fixation [12]. A recent study by Moody et al. on the cost of orthopedic trauma surgery operating time reported an average cost of $16 per minute, which can translate to roughly $1,000 in additional cost solely from pre-fixation frame application time [24]. While this study does not report the exact time savings that the IMNG approach affords, this technique definitely requires fewer time and steps than CHAOS.
This study is limited by its retrospective nature and its small sample size. Furthermore, because not all patients in this cohort had hip-to-ankle standing radiographs available for review, some anatomical measurements, such as LDFA, MPTA, and LDTA, were recorded using long bone radiographs. Given that most patients did not have lateral hip-to-ankle standing radiographs postoperatively, it was not possible to quantify the degree of correction of procurvatum and recurvatum deformities. Future studies should consider standardizing postoperative lateral hip-to-ankle standing films, in addition to anteroposterior, for review by the orthopedic surgeon to better assess these deformities.
CONCLUSION
This study demonstrates accurate correction of complex multiplanar tibial and femoral deformities using the IMNG approach, while maintaining a low rate of complications in comparison to other DC techniques. IMNG should be considered a cost-efficient correction option with readily available implants for restoring alignment and function in patients with complex deformities of the femur and tibia.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: S.A.S., W.W.C., B.J.Y., J.T., W.W.C., K.A.H.: Study conception and design; M.L.A., A.L.T., A.K.: Data collection; M.L.A., A.L.T., B.J.Y., S.A.S.: Analysis and interpretation of results; M.L.A., A.L.T.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| IMNG | = Intramedullary Nail-Guided |
| DC | = Deformity Correction |
| MAD | = Mechanical Axis Deviation |
| CHAOS | = Computer Hexapod-Assisted Orthopedic Surgery |
| IMN | = Intramedullary nail |
| LDFA | = Lateral Distal Femoral Angle |
| MPTA | = Medial Proximal Tibial Angle |
| LDTA | = Lateral Distal Tibial Angle |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The Mayo Clinic Institutional Review Board in Rochester, MN, United States of America, reviewed this study and approved it under IRB number 21-009039.
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2000.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this published article.
FUNDING
This research was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases for the Musculoskeletal Research Training Program (T32 AR56950).
ACKNOWLEDGEMENTS
The authors would like to acknowledge Gavin H. Ward for his contributions toward the generation of figures for this manuscript.


