All published articles of this journal are available on ScienceDirect.
A Case Report: Custom Made Porous Titanium Implants in Revision: A New Option for Complex Issues
Abstract
Background:
Bone loss management is considered one of the most difficult challenges for orthopaedic surgeon. In massive bone defects, few surgical options are available and they do not offer a reliable or optimal solution for knee reconstruction.
Objective:
The aim of this paper is to present and justify a new custom-made approach for complex metadiaphyseal bone defects management in knee revision surgery.
Methods:
We report a case of a 66-year-old woman who underwent a staged left total knee arthroplasty revision for infection with Anderson Orthopaedic Research Institute classification III uncontained femoral and tibial metadiaphyseal bone defects following five prior surgeries. Along with a case discussion, we describe clinical and radiological outcomes of 3 similar patients treated with this new custom-made device.
Results:
To manage these problems, we developed new, custom porous titanium devices for both femoral and tibial reconstruction tailored to a patient’s specific bone loss. Since, 2014, we treated four cases using custom-made porous titanium cones and we had optimal clinical and radiological results, with no instances of loosening, component migration, or mismatches between preoperative planning and intraoperative findings.
Conclusion:
In extremely selected cases, this new device can be considered a possible and viable surgical step between “off the shelf” reconstruction implants and knee substitution with a tumor megaprosthesis.
1. INTRODUCTION
Since 2003, the number of Total Knee Arthroplasties (TKAs) in the United States has increased by 130%, and the number is projected to increase worldwide. Concomitantly, the demand for revision TKA is increasing and expected to grow proportionally [1-6]. The main reasons for knee revision are aseptic loosening, periprosthetic joint infection, periprosthetic fractures, major osteolysis, extensor mechanism problems, instability, and stiffness [7, 8]. These conditions pose several challenges to orthopaedic surgeon in the area of restoration of knee biomechanics, bone loss management, and durable implant fixation. According to the recent literature, minor bone defects (smaller than 5 mm) should be managed with cement and morcellized bone graft for optimal outcomes [9-11]. By contrast, a consensus regarding major bone loss management (for Anderson Orthopaedic Research Institute [AORI] classification [9] types II and III) has yet to be established. Monocondylar bone defects (AORI type IIA) could be managed with cement augmented with screws or Trabecular Metal (TM) modular augments, which have shown optimal mid-term to long-term clinical results [11-13]. In cases of severe bone loss (AORI type IIB or III), the possible solutions rely on structural allografts, TM cones, sleeves or wide bone resection with megaprosthesis implantation [14-18]. Disease transmission, delayed weight-bearing, late resorption, graft fracture or non-union, and collapse are well-known disadvantages of structural grafting [19, 20]. For these reasons, TM cones and sleeves gained increased popularity. Recent literature about these devices provided excellent clinical and radiological data [21-25], but very few data on long-term follow-up is available [26, 27]. Moreover, in cases of large metadiaphyseal bone defects, none of these devices can effectively solve the problem. Megaprosthesis have been proposed in large bone defects with good results at short-term follow-up [18, 28] but data on oncologic cohorts of patients at longer follow-up periods are generally poor [29]. Globally, the life expectancy of patients with severe bone loss is longer than that of oncologic patients, which is why megaprosthesis should be considered in extremely selected cases.
We report the case of a 66-year-old woman who underwent a staged left TKA revision for infection with AORI class III uncontained femoral and tibial metadiaphyseal bone loss following five prior surgeries. To manage these problems, we developed new, custom porous titanium devices for both femoral and tibial reconstruction that were tailored to the patient’s specific bone loss and matched with the hinged prosthetic design used.
2. CASE HISTORY
A 66-year-old obese woman came to our facility for consultation with progressive left knee pain (9/10 on Visual Analogue Scale) mainly unrelated to activity levels. The patient reported her first left knee surgery in 1994 for an arthroscopic partial medial meniscectomy. She underwent primary left TKA in 2010. One year later, she was diagnosed with a Periprosthetic Joint Infection (PJI) and was managed with two-stage exchange using a revision-constrained condylar knee prosthesis with tibial tuberosity osteotomy, medial tibial wedge, and offset tibial stem. A breakage of the initial spacer with subsequent spacer substitution occurred during the interstage period.
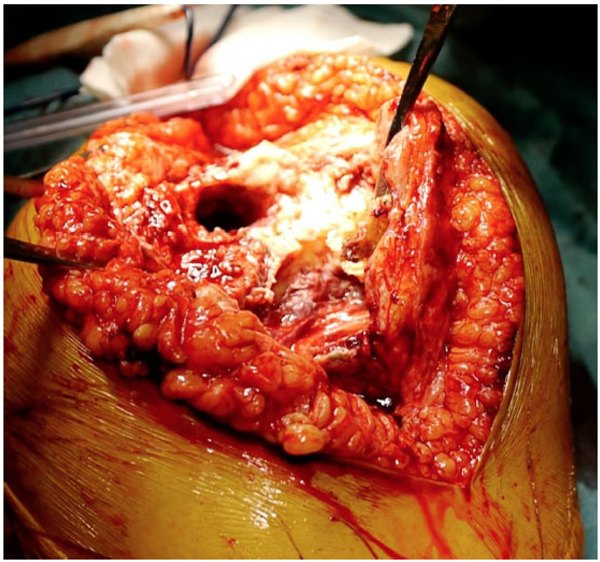
On physical examination at our facility, her knee showed a partially retracted midline scar with severe soft tissue damage (knee instability) and gross purulent drainage. Local swelling and inflammation signs were present. Her limb alignment was neutral. The left knee range of movement was painful and limited from -5° to 30°. Her hip examination results were unremarkable. Her body mass index was 34.7 kg/m2. A concomitant drug-induced (Linezolid) peripheral neuropathy was observed. Radiographic analysis performed at the time of our first evaluation showed a knee revision prosthesis with press-fit tibial and femoral stems and hybrid fixation (Fig. 1). We also detected metaphyseal tibial and femoral osteolysis consistent with AORI type III defects.
Serologic tests showed high levels of C-reactive Protein (CRP) (96 mg/L; range, 0.0 mg/L to 5 mg/L) and Erythrocyte Sedimentation Rate (ESR; 130 mm/h; range, 0 mm/h to 30 mm/h). No arthrocentesis was performed.
Knee arthrodesis was contemplated as an option, but considering its unacceptable results, a further two-stage procedure was attempted. The patient gave her informed consent before each surgical procedure and for clinical and scientific investigations.
Firstly, we performed a TKA explantation with extensive debridement of the infected tissues; then we placed an articulating cement spacer impregnated with antibiotic (Vancomicin and Gentamicin) (SpacerK® - TECRES S.p.A., Sommacampagna, Verona, Italy) fixed with antibiotic-loaded bone cement (Palacos® R+ G - Heraeus Medical GmbH, Wehrheim, Germany). For sufficient implant stability, the femoral side was completed with a handmade stem internally reinforced with a 2.2-mm K-wire (Fig. 2). Intraoperative cultures showed the presence of methicillin-resistant Staphylococcus epidermidis, which we treated with an intravenous antibiotic course of six weeks with Daptomycin and Ciprofloxacin. Accurate follow-up with serological CRP and ESR was performed in the interstage period; preoperative synovial fluid aspiration and analysis (culture, leukocyte esterase strip, synovial white blood cells count and alpha defensin lateral flow test) were performed after 2 weeks of antibiotic washout.

Due to the extremely poor bone stock and further bone loss anticipated during the subsequent surgery, very few surgical options were available. Although structural allograft showed good bone stock restoration at mid-term and long-term follow-up [30], it is a technically demanding surgery with higher septic failure risk in patients with PJI [31]. Megaprostheses have proven to be successful in patients with large amounts of bone loss but their suboptimal results are not adequate for patients with a long life expectancy. Trabecular cones and sleeves lead to reliable results in moderate and contained bone loss but in cases of wide or uncontained metadiaphyseal defects, no preformed shape is available, and high demanding double-cone constructs are required. In those cases, an adequate fit with the available residual bone is often impossible to achieve.

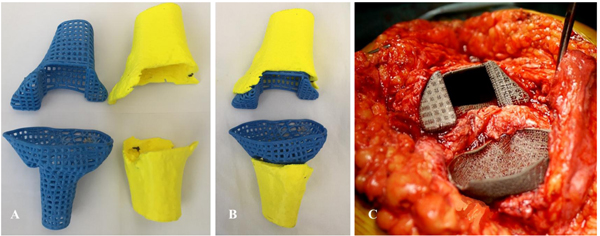
Therefore, we produced a new, custom-made porous titanium device that mimics the patient’s specific anatomy on both the femoral and tibial side. The aim was to provide adequate metadiaphyseal stability, bone ingrowth, and joint line restoration using the available residual bone (Fig. 3), and we used the contralateral knee as a reference configuration.
In cooperation with industry engineers (Adler Ortho S.P.A., Cormano, Milano, Italy), a custom-made cone for each side was tailored and designed based on the preoperative Computed Tomography (CT) scan with the spacer in situ (Fig. 4). Preoperative measurements were based on the contralateral unaffected side anatomy. The custom-made femoral device was created with an external diameter of 36 mm and had 45 mm of scratch fit interface on the metadiaphyseal region for bone ingrowth. The internal diameter was 16 mm to allow the insertion of a 15-mm x 100-mm stem and a 1-mm cement mantle between the stem and the custom-made cone. The tibial device was designed with a mean metaphyseal outer diameter of 31 mm and 40 mm of scratch fit. The inner diameter was 13 mm for a 12-mm x 75-mm stem and a 1-mm cement mantle (Fig. 5).
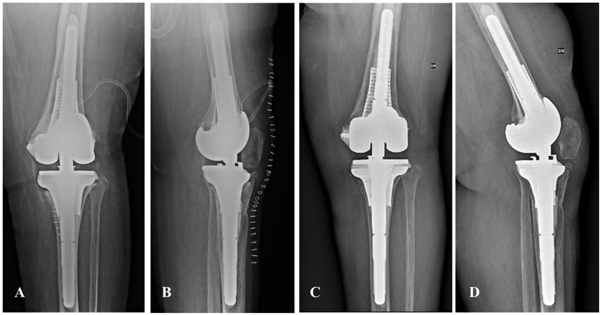
The patient underwent TKA reimplantation with a rotating hinged knee prosthesis (Zimmer Biomet, Warsaw, IN, USA) according to the preoperative planning 12 weeks after the first stage. Optimal implant primary stability and range of movement was achieved intraoperatively.

The patient was mobilized form the third postoperative day, and she was allowed toe-touch weight bearing for four weeks. According to our department protocol, her antibiotic therapy was prolonged until the results of the intraoperative cultures were obtained. Ten days after surgery, the antibiotic therapy was stopped after the intraoperative culture results were negative. After the one-month clinical and radiological evaluations, the patient started 50% weight-bearing and reached 100% weight-bearing three months after surgery. At the 2-year follow-up, she could perform her daily-life activities (Knee Society Score: 86) with 0° to 90° of range of movement, no pain (0/10 on Visual Analogue Scale), and she rated her outcome as satisfactory. The radiographic analysis performed during this follow-up showed excellent implant alignment and adequate cementation with no radiolucent lines or migration (Fig. 6).
3. DISCUSSION
Management of bone loss in a revision TKA is a cause of concern. The proposed surgical techniques in this scenario rely on structural allograft, porous metal cones, sleeves, and a megaprosthesis.
Structural allograft carries the possibility of biologic graft integration and is considered a viable option in these surgical settings. Recently, Sandiford et al. compared clinical and radiological results with complications observed in 45 knee revisions in which a femoral head allograft was used in 30 and TM cones in 15 [30]. The minimum follow-up was 5 years (mean, 9 years). According to their data, no statistically significant differences were detectable in clinical or radiological results or complication rates between the two groups. Survivorship of allograft at a mean of 5 years was 93%, which compares favourably with the same data in the TM cones group (91%). One cone was revised for aseptic loosening, and one patient sustained a periprosthetic fracture in the allograft cohort of patients. Despite these data, Beckman et al showed how a structural allograft could fail (6.5% incidence of graft failure, 3.4% of aseptic component loosening, and 55% of deep infection at 5.9 years follow-up) affirming the higher reliability of cones in these situations [32].
TM cones showed durable results in metaphyseal bone loss management at mid-term and long-term follow-up [26, 33, 34]. Some issues can occur for extensive uncontained metadiaphyseal defects where scratch fit is difficult to obtain. Some authors suggested the use of the two -cone techniques for massive bone defects. Boureau et al. proposed the use of two cones, one next to the other, in the management of massive bone defects during revision TKA [35]. At a preliminary follow of 1.4 years, they reported optimal clinical and radiological data with no loosening or implant revisions and no complications due to wear of the tantalum cones. Some concerns remain for the long-term durability of this technique, especially regarding possible tantalum wear. Ongoing new developments may help maximize the bone-metal contact. Faizan et al. performed a mechanical analysis of a new porous titanium metaphyseal cone [36]. This device was manufactured according to a CT-based anatomical database to maximize the bone-implant contact surface. Ten of the new cones were implanted and compared to seven traditional ones. The authors reported that the new implant had the stability that was equal or better than the tantalum cones. The new component demonstrated less micromotion in the medial, valgus, and posterior directions when compared to usual cones. Custom metal sleeves have had promising results, too [37]. These data are encouraging for possible new developments and reminds us that the optimal solution has not yet been found.
From a biomechanical point of view, bone stress and the relative load transferred to the bone are important factors that can be related to the life expectancy of each reconstructive technique. Ideally, stress-strain levels should be close to the control situation in which no augment is inserted [38]. As also reported by Patel et al. [39], the difference in elasticity between metal and bone may cause stress shielding and increase potential bone loss. Indeed, the presence of a wedge is a fundamental tool to treat bone defect, because, if it is untreated, it could lead to a missing mechanical support producing high bone stress variation. However, as determined by Innocenti [11], among the different augment options, the main parameter that is responsible for the change of bone stress is the material (due to its stiffness) of the augment that should be as close as possible to the one of the bone. Moreover, due to the additional benefit of the porous metal of allowing bone ingrowth, the loosening rate will be further reduced. Furthermore, as also demonstrated by El-Zayat [40], a short stem is able to distribute the stress only on the metaphyseal region of the bone, without any additional constraint on the distal tibia, that could overload the bone and, therefore, increase the risk of fracture of the bone. Accordingly, the use of trabecular cone and short stem, are beneficial for a safer metaphyseal implant fixation, avoiding any additional stress concentration and stress risers, promoting good stability.
Accordingly, since 2014 we treated four cases using custom-made porous titanium cones and we had optimal clinical and radiological results, with no instances of loosening, component migration, or mismatches between preoperative planning and intraoperative findings (Table 1).
| Case | Sex | Age | Side | Bone Defect | Reason for Revision | Number of Previous Surgeries | Side of Custom-made Device | Final Implant | Complications | Final Follow-Up (months) |
|---|---|---|---|---|---|---|---|---|---|---|
| Case 1 | F | 71 | L | AORI F3/T3 | PJI | 4 | Femur and Tibia | RHK | No | 30 |
| Case 2 (case report) |
F | 66 | L | AORI F3/T3 | PJI | 5 | Femur and Tibia | RHK | No | 24 |
| Case 3 | M | 58 | R | AORI F3/T3 | PJI | 3 | Femur | RHK | No | 19 |
| Case 4 | M | 66 | R | AORI F3/T3 | PJI | 5 | Femur | RHK | No | 14 |
CONCLUSION
Bone loss management represents one of the most challenging issues for the orthopaedic surgeon. In most cases, stems, structural allograft, TM cones, and sleeves are adequate to allow optimal implant stability and durable fixation. In selected cases of wide metadiaphyseal bone defects, these devices do not provide proper intraoperative stability. In such scenarios, further steps are needed and include complex modular reconstruction, substitution with megaprosthesis (exposing patients at high risk of early failure) or joint arthrodesis that can yield unacceptable results. A custom-made implant may be an intermediate and reliable solution in cases where the choices are allograft, cones or sleeves, and megaprosthesis or arthrodesis. Accurate surgical planning and intraoperative management of soft tissues and residual bone stock are of paramount importance.
LIST OF ABBREVIATIONS
| TKA | = Total Knee Arthroplasty |
| AORI | = Anderson Orthopaedic Research Institute |
| TM | = Trabecular Metal |
| PJI | = periprosthetic Joint Infection |
| CRP | = C-Reactive Protein |
| ESR | = Erythrocyte Sedimentation Rate |
FUNDING SOURCES
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Local Institutional review board approval was obtained before starting the study. Each patient gave written and informed consent before each surgical procedure.
HUMAN AND ANIMAL RIGHTS
The authors certificate that the performed procedures respect the ethical standards in the Helsinki Declaration of 1975, as revised in 2000, as well as the national law.
CONSENT FOR PUBLICATION
All patients represented in this study were informed that the data from their case would be de-identified and used in a journal publication.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGMENTS
Declared none.


