All published articles of this journal are available on ScienceDirect.
One Stage Bilateral Total Hip Arthroplasty in Siblings with Larsen Syndrome
Abstract
Background:
Larsen syndrome is a rare genetic disorder caused by congenital weakness of the connective tissues. It can present with a variety of musculoskeletal and cardiovascular abnormalities. The current report describes two siblings with Larsen Syndrome who presented with severe bilateral hip arthritis and underwent one stage bilateral total hip arthroplasty (THA). The aim was to report on the clinical features of Larsen Syndrome and their implications for total hip replacement surgery.
Methods:
Two siblings, a 32 year-old female and a 30 year-old male, presented with severe bilateral hip arthritis and a history of Larsen Syndrome. Both patients underwent a detailed, multidisciplinary preoperative medical work up and radiological imaging including computer tomography. All four hips were operated using a cementless primary press-fit cup (Pinnacle, DePuy, Warsaw, IN) and a cementless modular stem system (S-ROM, DePuy, Warsaw, IN) utilizing a posterior approach. Prophylactic cerclages wires were placed proximal to the lesser trochanter before stem preparation and bone grafting of bone cysts in the greater trochanter was performed in all four hips.
Results:
After 2 years both patients reported significant improvements of function, pain and quality of life. The Oxford hip score improved from preoperative 21 (range 12-24) points up to 39 (range 38-41) points at 2-year follow up. Radiographic follow-up showed good graft incorporation and no signs of implant loosening.
Conclusion:
The current case report suggests that one stage bilateral THA is a feasible treatment option for young adults with Larsen syndrome and secondary arthritis of the hip.
INTRODUCTION
Larsen syndrome is a rare genetic disorder caused by congenital weakness of the connective tissues that may present with a wide variety of musculoskeletal symptoms. It is named after L.J. Larsen who initially described the disease in 1950 as the combination of skeletal deformities with multiple joint dislocations and characteristic craniofacial abnormalities [1].
The classic form of Larsen syndrome is categorized amongst the FLNB-related disorders that occur due to mutations of the Filamin B gene (FLNB) gene [2]. These mutations may occur spontaneously [3] or can be inherited as an autosomal dominant trait [2]. The autosomal recessive form of Larsen syndrome is associated with mutations in the carbohydrate sulfotransferase 3 (CHST3) gene; alterations of this gene have also been identified in patients with other skeletal malformations such as humero-spinal dysostosis and spondyloepiphyseal dysplasia Omani type [4]. The incidence of Larson Syndrome is estimated at 1 in 100,000 live births [2, 5].
Characteristic findings of the disorder include short stature, dislocations of the large joints [1, 6] (hips, knees, elbows, frequently bilateral) and various other skeletal malformations of the hands (spatulate), feet (equinovarus, hind foot valgus) and spine (instability, hypodontia, kyphosis, scoliosis) [7-10]. Distinctive facial features include frontal bossing, midface hypoplasia with depression of the nasal bridge, and ocular hypertelorism [1, 11]. Other abnormalities include cardiovascular [12] (mitral valve prolapse, atrial or ventricular septum defects, aortic dilatation/ dissection/aneurysm) and respiratory system (broncho-/tracheomalacia) [13]. As a result the life expectancy is variable with a high rate of early deaths occurring in up to 40% of patients [10], mostly secondary to cardiovascular events, respiratory complications or instability of the cervical spine [10]. Interdisciplinary treatment of patients that survive into adolescence or adulthood is challenging due to the variety of conditions and associated complications [10].
The current report describes two siblings with Larsen Syndrome presenting with severe bilateral end-stage arthritis of the hip. To the authors´ best knowledge, this is the first report of total hip arthroplasties performed in patients with Larsen Syndrome.
CASE REPORT
The current case report presents 2 siblings (32 year-old female, 30 year-old male) with severe bilateral hip arthritis and a history of Larsen Syndrome.
Because of the severe bilateral disease and functional impairment the decision was made to proceed with a one stage bilateral total hip replacement. Both patients underwent detailed, multidisciplinary preoperative medical assessment and radiological imaging including CT- based planning.
The 32 year old female patient (height 1.33 m, weight 65 kg, BMI 36.7 kg/m2) reported a long standing history of bilateral hip and knee problems with predominately right hip pain and stiffness that had become progressively worse over the last years. The past surgical history included previous bilateral knee surgeries for bilateral patella dislocation, as well as bilateral open reductions for high hip dislocation at the age of 4. At age 10, the patient underwent a right pelvic osteotomy for recurrent dislocation of the right hip. The patient reported activity related pain of 9/10 on a visual analog scale (VAS) during walking. The walking distance was limited to approximately 150 m with permanent support of a cane.
Physical examination demonstrated a slow, antalgic gait with a marked leg length discrepancy of 32 mm with shortening of the right side. The range of motion was reduced to 75° of flexion on the right and 90° of flexion on the left. The patient had bilateral flexion contractures of 30° on the right and 20° on the left. There was no internal and external rotation. Both knees were stable and showed a flexion contracture of 20°. Knee flexion was limited to less than 90°. Both lower extremities were neurovascularly intact.
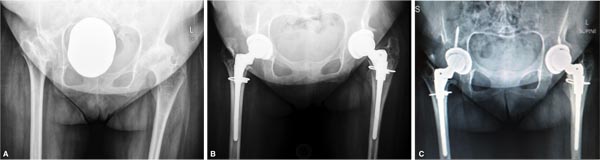
AP pelvis and lateral hip radiographs showed end stage secondary arthritis with residual dysplasia and status post pelvic osteotomy of the right hip (Figs. 1 and 2). Preoperative radiographs also revealed multisegmental osteochondrosis and spondylarthrosis with dysplastic alterations of the cervical vertebrae without evidence of atlantoaxial or subaxial instability in the 32-year-old female.
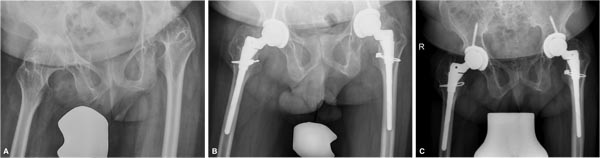
The 30-year-old male patient (height 1.25 m, weight 48 kg, BMI 30.7 kg/m2) reported a long-standing history of bilateral hip and knee problems with progressive hip pain and stiffness. He was born with bilateral dislocations of the patellae and had undergone initial bilateral knee surgery at the age of 2 years. Due to recurrent dislocations on the right side multiple revisions were performed with the last surgery being performed at the age of 11 years. No previous hip surgery was reported. The patient reported activity related hip pain of 8/10 on a visual analog scale (VAS) during walking. The walking distance was limited to approximately 100 m with support of a cane.
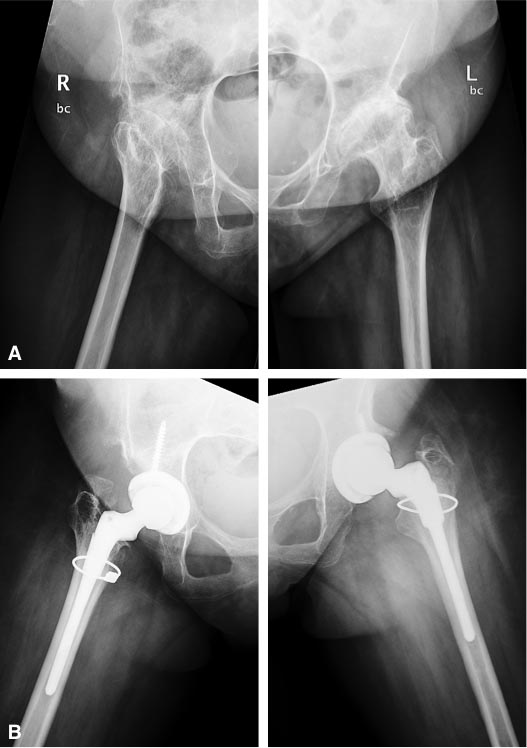
Physical examination demonstrated a slow, antalgic gait with 5 mm shortening of the right leg. Bilateral hip flexion was limited to 80° on the right and 45° on the left and the patient presented with bilateral flexion contractures of 50° and 30°, respectively. There was no internal and external rotation. Both knees were stable and the right knee showed a flexion contracture of 10°. Both lower extremities were neurovascularly intact. AP pelvis and lateral hip radiographs showed end-stage secondary arthritis with signs of acetabular retroversion (Figs. 3 and 4). The 30-year-old male patient presented with additional marked hypodontia on the C-spine films.
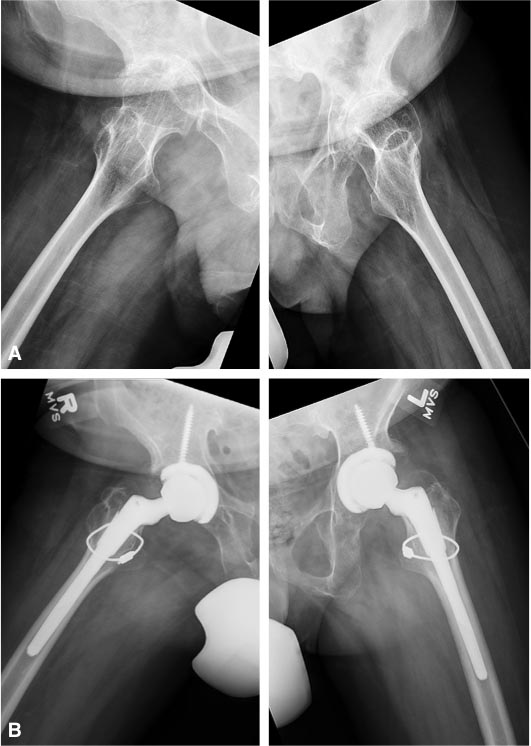
Both patients underwent cardiac echocardiogram to rule out cardiovascular abnormalities. Both patients were indicated for combined spinal epidural anesthesia to minimize the reported risk of malignant hyperthermia [14] and postoperative cardiac [15] or respiratory complications [16] in patients with Larsen syndrome. Because of the small size and resulting blood volume autologous blood donation was not recommended.
The decision was made to protect the cervical spine of both patients during surgery using a rigid cervical collar. Both patients displayed marked degenerative thoraco-lumbar scoliosis with resulting pelvic obliquity.
Because of the small size of the patients, the likelihood of version abnormalities and the possibility of a subtrochanteric shortening osteotomy a cementless primary press-fit cup (Pinnacle, DePuy, Warsaw, IN) and a cementless modular stem system were chosen (S-ROM, DePuy, Warsaw, IN).
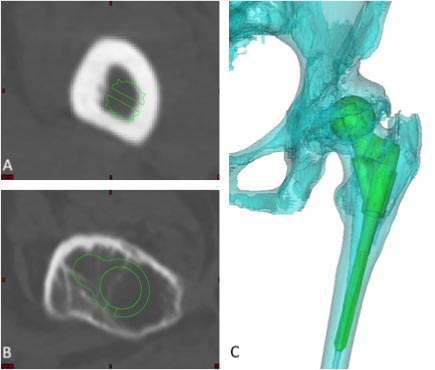
In the female patient, the left femoral canal appeared very narrow so that both an of-the- shelf implant and a custom made femoral stem were taken into consideration. On the radiographic preoperative templates, the smallest S-ROM stem (6 mm stem diameter) still appeared rather large and an additional CT scan with three dimensional (3D) reconstructions was obtained. A 3D simulation of the S-ROM size 6 and 7 mm stem demonstrated an adequate fit (Fig. 5).
In both patients a previously described, posterior approach in a lateral decubitus position with combined spinal-epidural anesthesia was used [17]. 10 mg of Tranexamic acid per kg body weight was administered preoperatively and 3 hours after the procedure to reduce blood loss in both cases.
In all 4 hips, good press fit of the cementless acetabular component in an anatomic position was achieved and one screw was inserted to augment fixation (Figs. 1-4). Both patients had poor bone stock and presented with significant bone loss in the proximal femur with large cysts in the greater trochanter. Before stem preparation, a prophylactic cerclages wire was placed proximal to the lesser trochanter to reduce the risk of intraoperative fracture. Bone grafting of the greater trochanter was performed in all 4 hips utilizing autologous cortico-cancellous chips obtained from the femoral head with additional 3-7 mm Orthovita chips (Stryker Mahwah, NJ). The S-ROM stem allows to adjust femoral version and we applied postoperative hip precautions for 4 weeks to reduce the risk of dislocation. Since the two patients were very stiff, it did not appear that the risk of postoperative dislocation is significantly increased in these patients.
No perioperative complication was encountered and both patients were mobilized with 50% partial weight-bearing on a walker for 4 weeks. The patients were discharged to an inpatient rehabilitation facility on postoperative day 6. Postoperative radiographs demonstrated well-fixed acetabular and femoral components in adequate position with high-density bone grafting around the femoral components.
At 2-year follow-up, both patients reported a remarkable improvement of function and quality of life (Table 1). Both patients were able to participate in moderate activities like walking beyond 1 kilometer and unlimited housework. The gait pattern in both patients improved significantly. Although the female patient continued to limb, she reported no restriction in regard to walking distance. Both patients were pain free, and able to use public transportation. Radiographic follow-up showed graft incorporation without implant loosening (Figs. 1 and 3).
| Preoperative | Postoperative | |
|---|---|---|
| Oxford Hip Score | 21 (range 12-24) | 39 (range 38-41) |
| WOMAC | 51 (range 48-54) | 14 (range 13-15) |
| VF-12 | 68.3 (± 0.4) | 101.9 (± 6.9) |
| MCS | 41.0 (±0.3) | 53.3 (± 6.7) |
| PCS | 27.4 (± 0.2) | 48.6 (± 0.2) |
DISCUSSION
The present case report highlights two major findings in young adults with Larsen syndrome and bilateral secondary arthritis of the hip [1]: the importance of an interdisciplinary preoperative work-up and [2] the technical difficulties of surgery.
In patients with Larsen syndrome undergoing joint replacement, concomitant cardiovascular and respiratory comorbidities need to be excluded preoperatively.
It is important to consider that Larsen syndrome presents with a wide range of musculoskeletal deformities like dislocations of the large joints and malformations of the spine. Because of the described risk of odontoid hypoplasia the authors recommend preoperative imaging of the cervical spine and prophylactic stabilization of the cervical spine using a soft or rigid collar during surgery. When possible, a spinal/epidural anaesthesia avoids hyperextension of the cervical spine during intubation and allows for intraoperative hypotension to reduce blood loss. The anesthesiologist should be comfortable with fiber optic intubation in case this is necessary.
There were several intraoperative findings, which made the implantation of components technically challenging: When performing the acetabular and femoral preparation the combination of soft tissue contractures and poor bone stock increased the risk of fracture. Because of the shortened femoral neck as well as the combination of acetabular retroversion and femoral anteversion the sciatic nerve was located close to the greater trochanter. Acetabular component fixation and positioning was more difficult because of the decreased lateral bone coverage [18] with poor bone stock and retroversion of the acetabulum. Careful templating and preoperative CT scans with 3 dimensional reconstruction and planning of the femoral component might be necessary to make sure custom made femoral components are not needed. CT-based preoperative planning [19] proved to be very helpful in the current cases. Our findings confirm the observation of a high inter-individual variability of the bony deformities in Larsen syndrome even among members of the same family [20].
In the literature, there is limited evidence of therapeutic strategies in patients with Larsen syndrome and most reported cases present the findings of skeletal abnormalities in childhood or adolescence. Although developmental dysplasia can be diagnosed during the first days of life utilizing ultrasound [21] early non-operative treatment is often limited because of concomitant knee dislocations. Since patients often do not respond to serial manipulation and casting of the dislocated knees most patients require open reduction and soft tissue releases which will ultimately lead to further stiffness and poor overall function [22]. We are only aware of one case report of a total joint arthroplasty in a patient with Larsen syndrome. Mueller et al. reported a successful case of bilateral total knee arthroplasty performed in a 59-year-old woman with bilateral congenital knee dislocation [23]. To prevent neurovascular damage, continuous joint distraction was performed preoperatively using unilateral external fixation. Our report confirms the finding of marked joint stiffness with severe joint degeneration of the hips and knees following open surgical procedures in childhood and adolescence in both patients.
In summary, the present case report highlights the importance of an interdisciplinary approach in patients with Larsen syndrome. These patients are at increased risk for cervical instability as well as cardiovascular and respiratory comorbidities. Three-dimensional CT-based planning may be required to make sure that standard implants are feasible. Hip reconstruction is technically challenging as a result of stiffness, bony deformities, previous surgeries and poor bone stock. The current report suggests that one stage bilateral THA can restore function and eliminate hip pain in young adults with Larsen syndrome and secondary end-stage of arthritis the hip.
CONFLICT OF INTEREST
We certify that we have not signed any agreement with commercial interest related to this study, which would in any way limit publication of any and all data generated for the study or to delay publication for any reason.
ACKNOWLEDGEMENTS
Dr. Friedrich Boettner reports personal fees from Smith & Nephew, personal fees and royalties from Ortho Development Corporation and personal fees form Depuy, outside the submitted work.


